Introduction
BRASH syndrome—an acronym for bradycardia, renal failure, atrioventricular nodal blockade, shock, and hyperkalemia syndrome—describes a self-perpetuating cycle of severe bradycardia and hyperkalemia that occurs when atrioventricular nodal blockers are used. Although the use of the acronym BRASH is a more recent development, the connection between these medications and renal failure has been described in the literature for years.
Since their development in the 1960s, atrioventricular nodal-blocking medications, such as β-blockers and calcium channel blockers, have been used to treat conditions such as coronary artery disease, hypertension, and tachyarrhythmias, including atrial fibrillation. These medications act on the atrioventricular node to attenuate conduction, leading to decreased heart rates. Similar to all pharmaceuticals, atrioventricular nodal blockers are associated with adverse reactions. The most common adverse effects include fatigue, dizziness, sleep disturbance, and dyspnea. However, BRASH syndrome is a frequently overlooked complication that can lead to significant morbidity and mortality if not properly recognized and treated.[1]
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
BRASH syndrome is a distinct entity that is becoming more prevalent as a cause of profound bradycardia due to the compounding comorbidities of the aging population and the advent of new pharmaceuticals targeting cardiac output at the atrioventricular node. The synergistic effect of atrioventricular nodal blockade and renal failure causes a vicious cycle of profound bradycardia and hyperkalemia. Some experts hypothesize that BRASH syndrome occurs more often than reported but is frequently unrecognized, leading to misdiagnosis and mismanagement.[2]
Epidemiology
As BRASH syndrome is a recently described condition, little is known regarding its epidemiology; however, case reports indicate a higher prevalence in older individuals with both underlying cardiac and renal impairment. The risk of developing this complication increases further when the patient is on medications that attenuate the atrioventricular node. Angiotensin-converting enzyme inhibitors and angiotensin receptor blockers increase the risk of developing acute kidney injury and hyperkalemia.[3]
Pathophysiology
The pathophysiology of BRASH syndrome involves the synergistic effects of atrioventricular nodal blockers directly causing bradycardia and indirectly affecting renal perfusion, leading to hyperkalemia and worsening bradycardia (see Image. Pathophysiology of BRASH Syndrome). A direct atrioventricular node blockade results in reduced cardiac output and renal perfusion, ultimately leading to an acute kidney injury. As kidney function worsens, potassium clearance decreases, resulting in hyperkalemia. Severe hyperkalemia alone can cause bradycardia. However, due to the collective effects of atrioventricular nodal blockade and reduced renal clearance, profound bradycardia can occur even in the setting of mild hyperkalemia. Lipid-soluble β-blockers, such as metoprolol and carvedilol, and the majority of calcium channel blockers are primarily excreted by the kidneys; therefore, these medications impose a greater risk as their concentrations inversely increase in the renal tubules with decreasing glomerular filtration rate (GFR).[4] If left undiagnosed, this cycle can progress to multi-system organ failure.
Innocuous events, such as a recent illness, medication change, or simple dehydration, often initiate this vicious cycle. Dehydration is one of the most common causes, as hypovolemia leads to a pre-renal acute kidney injury.[5] A study found a temporal association between the development of bradyarrhythmias and hyperkalemia in warm summer months, suggesting dehydration as the root cause.[6] Other common inciting events include the up-titration of medications that alter cardiac output, the addition of nephrotoxic agents, or potassium-sparing diuretics to the medication regimen.
History and Physical
The clinical presentation of BRASH syndrome may vary widely, ranging from asymptomatic bradycardia to cardiogenic shock requiring vasopressors and hemodialysis. Despite these variations, all patients present with profound bradycardia. As this syndrome is often precipitated by decreased renal perfusion, history often includes recent gastrointestinal illness, dehydration, recent medication changes, or other history components identified as risk factors for acute kidney injury. This syndrome is not dose-dependent when considering the role that atrioventricular nodal blockers play.[7]
Evaluation
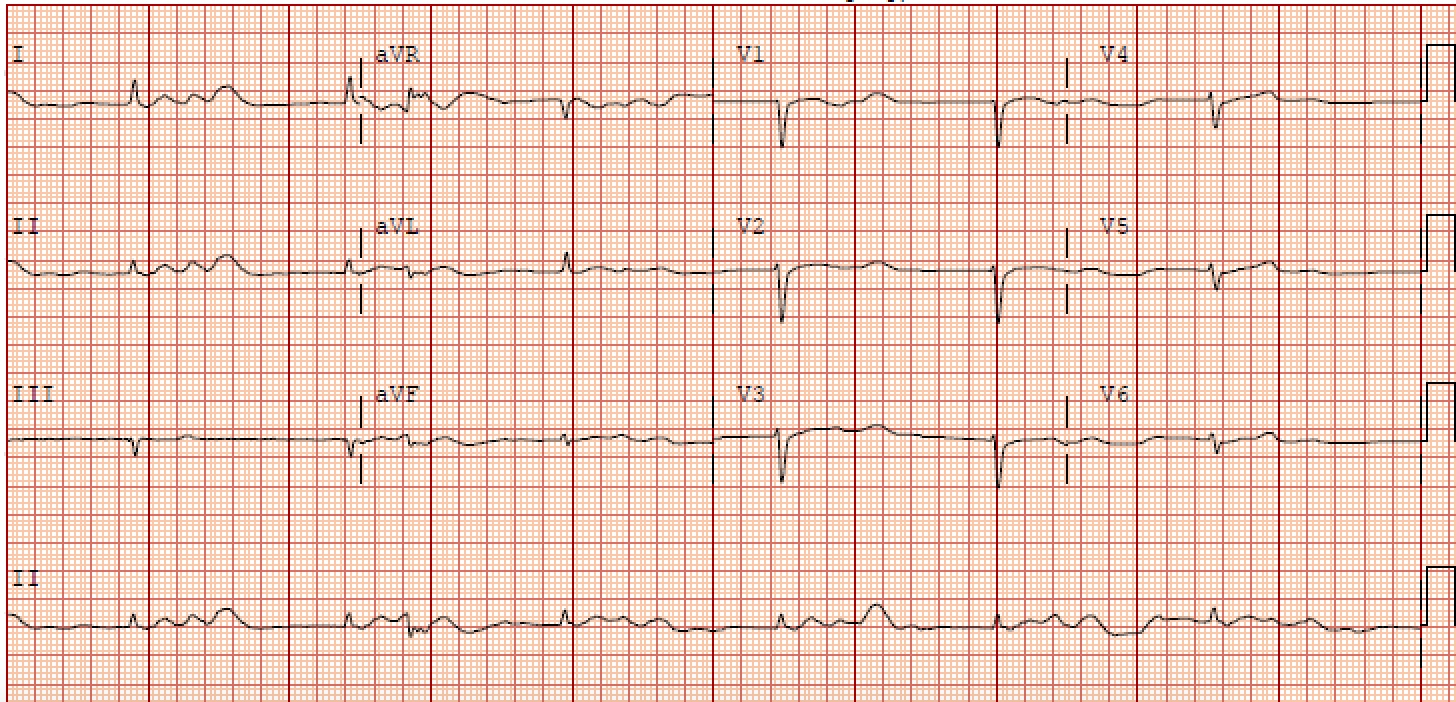
An electrocardiogram (ECG) is crucial in differentiating BRASH syndrome from other causes of bradycardia, such as isolated hyperkalemia, hypothermia, or hypothyroidism (see Image. Electrocardiographic Findings in a Patient with BRASH Syndrome). However, these findings are commonly underwhelming as an ECG does not typically show the stereotypical electrocardiographic changes observed in patients with hyperkalemia. A progression of electrocardiographic changes occurs as serum potassium levels elevate, starting with peaked T-waves and PR interval prolongation, followed by QRS interval widening and subsequent bradycardia with a sinusoidal rhythm.[8] These morphologies are commonly absent in patients with BRASH syndrome.
A comprehensive metabolic panel (CMP) demonstrates some degree of hyperkalemia. Many case reports have documented profound bradycardia associated with even mild hyperkalemia, likely due to the synergistic effect of the concomitant atrioventricular nodal blockage, which is the hallmark of BRASH syndrome. A case report described the development of BRASH syndrome in a patient with a serum potassium of 5.1 mEq/L; however, potassium levels of 5.5 to 7.0 mEq/L are more commonly described.[9] Regardless of hyperkalemia, potassium levels exceeding 5 mEq/L can be cardiotoxic, resulting in decreased membrane excitability and bradycardia.
An acute kidney injury with creatinine elevation is typically observed on a CMP. Although most of these patients have a tenuous renal function at baseline, there is a notable increase in creatinine and blood urea nitrogen levels and a decrease in GFR. A uremic acidosis may also be present in some cases. The remaining laboratory studies vary depending on the patient's presentation. Lastly, a high index of suspicion is essential for diagnosing this condition, as traditional laboratory testing and ECG monitoring may appear innocuous.
Treatment / Management
The BRASH syndrome describes a constellation of symptoms, not an underlying cause. During the initial evaluation, clinicians often focus on the dominant feature of the presentation, such as bradycardia. However, failing to acknowledge the pathophysiology leading to bradycardia often leads to failed resuscitative efforts and unnecessary invasive interventions, such as transcutaneous or transvenous pacing. A concerted effort focusing on aggressive fluid resuscitation, cardiac membrane stabilization, potassium shifting, and improving cardiac output is vital.
Although patients often present with profound bradycardia, aggressive treatment of hyperkalemia should be initiated first by stabilizing the cardiac membrane with either calcium chloride or calcium gluconate. Hypocalcemia increases the cardiotoxic effects of hyperkalemia; prompt intravenous administration of calcium is warranted. Calcium chloride contains 3 times more elemental calcium than calcium gluconate. However, calcium gluconate is more commonly used due to increased patient tolerance when administered peripherally. The standard dose of calcium gluconate is 10 mL of 10% solution or 1 g intravenously, administered over 5 to 10 min. The standard dose of calcium chloride is 1 g, administered over 1 to 2 min.[10]
If bradycardia fails to improve within 5 minutes, calcium can be re-administered. If bradycardia persists despite this, epinephrine can improve both cardiac chronotropy and inotropy, thus leading to increased heart rates and systemic vascular resistance. Improvement can be achieved by administering a push-dose pressor of 10 to 20 μg of a 1:100,000 dilution. Epinephrine also stimulates β2 receptors, leading to a small potassium shift. Isoproterenol is a pure β-agonist with a stronger chronotropic effect compared to epinephrine. However, isoproterenol is not as readily available as epinephrine and, therefore, is not used as frequently. The key difference between BRASH syndrome resuscitation and the standard Advanced Cardiac Life Support approach to bradyarrhythmias is the administration of 0.5 mg of atropine every 3 to 5 minutes. Atropine is often ineffective in these cases as the profound bradycardia is not vagally mediated.[11]
Combination therapies that promote the intracellular shift of potassium or increase the total body clearance of potassium are also important interventions. Insulin reduces serum potassium levels by shifting potassium back into the cell. A bolus of 10 units of regular insulin should be administered, followed by 25 to 50 g of 50% dextrose to help prevent hypoglycemia. In patients with severe kidney injury or failure and euglycemia, it is advisable to administer 25 to 50 g of 50% before and after the insulin bolus to help mitigate the risk of potentially fatal hypoglycemia. Some studies show that it is reasonable to withhold the dextrose when glucose is 250 mg/dL. Nebulized albuterol can also be used to shift potassium back into cells. This treatment is dose-dependent, with the standard dose being 15 to 20 mg. Albuterol is a β2 agonist that shifts potassium intracellularly and produces a positive inotropic effect.[12][13][14] (B3)
As hypovolemia and dehydration are the most common causes of BRASH syndrome, performing a focused bedside cardiac ultrasound to assess fluid status is essential. Balanced crystalloids are appropriate in patients with an initial pH of 7.35 to 7.45. Large volume resuscitation with 0.9% normal saline should be avoided, as it can cause hyperchloremic acidosis, potentially worsening prognosis.[15]
A non-anion gap metabolic acidosis, specifically uremia acidosis, is common in patients with BRASH syndrome. The proposed fluid for these scenarios is isotonic 150 mEq/L sodium bicarbonate in D5W. The BICAR-ICU trial studied sodium bicarbonate as the fluid of choice in 389 patients admitted to the intensive care unit and its role in all-cause mortality. Despite suggesting a decrease in mortality with bicarbonate use for resuscitation, the primary endpoint was not statistically significant. However, the study results did show an improvement in morbidity as it decreased the need for emergent dialysis from 52% to 35% (P=0.0009).[16] (A1)
A definitive treatment for hyperkalemia requires the removal of potassium from the body. Kaliuresis describes the process by which potassium is excreted through the urine. This process is generally achieved by administering loop diuretics, thiazides, or acetazolamide. Successful kaliuresis often requires the use of high-dose diuretics to concentrate the medication within the nephron. Mild-to-moderate hyperkalemia may be successfully treated with loop diuretic monotherapy, such as furosemide 60 to 100 mg intravenously or bumetanide 2 mg intravenously. More severe hyperkalemia often requires the addition of a thiazide diuretic. Combination therapy has been shown to improve diuresis in patients resistant to high-dose loop diuretics. For patients with anuria, emergent dialysis is warranted for the definitive removal of potassium.[17] (B3)
If hypotension persists despite adequate fluid resuscitation and the above interventions, vasopressors such as epinephrine or isoproterenol should be initiated. Norepinephrine and dobutamine can be considered to improve cardiac contractility in patients with BRASH syndrome who do not respond to other therapies.[18](A1)
Differential Diagnosis
The differential diagnosis for patients presenting with bradycardia and shock is broad. BRASH syndrome shares many clinical signs and symptoms with atrioventricular nodal toxicity from overdose and pure hyperkalemia. These conditions must be carefully considered, as their definitive management strategies may differ. A fingerstick glucose level is a useful diagnostic tool. β-blocker overdoses often result in hypoglycemia, whereas calcium channel overdose often causes hyperglycemia. Significant potassium derangement is rare in these overdoses.[19][20] Although hyperkalemia alone can cause bradycardia, it is not typically observed until potassium levels exceed 7.0 mEq/L. ECG changes associated with mild hyperkalemia (5.5-6.5 mEq/L) include peaked T-waves. Flatting of P-waves with PR interval prolongation is noted with moderate hypokalemia (6.5-7.0 mEq/L). QRS prolongation and bradycardia are typically observed only when the potassium level exceeds 7.0 mEq/L. This stepwise progression of ECG changes is often absent in patients with BRASH syndrome.[21]
Prognosis
Most cases of BRASH syndrome respond well to the interventions described in this topic; however, more invasive therapies, such as hemodialysis or transvenous pacing, may be required. Prompt recognition of this syndrome improves prognosis and decreases the likelihood of needing these advanced measures. The severity of acute kidney injury and hyperkalemia is the primary factor influencing patient outcomes.
Complications
Complications of BRASH syndrome can range from renal failure necessitating the initiation of hemodialysis to cardiogenic shock requiring vasopressor initiation or transvenous pacing. If left untreated, these complications ultimately result in cardiac arrest.
Deterrence and Patient Education
Patient education should focus on recognizing warning signs for arrhythmia, including lightheadedness, syncope, or signs suggestive of acute renal failure, such as fluid overload or decreased urine output. Atrioventricular nodal blockers are commonly prescribed for various conditions. Therefore, patients should be educated on this syndrome as a potential adverse reaction.
Enhancing Healthcare Team Outcomes
Medications affecting the atrioventricular node are some of the most commonly prescribed in the United States; therefore, this potentially life-threatening complication must be acknowledged by all members of the healthcare team. Clinicians should be aware of the risk factors associated with an aging population taking multiple medications, fostering a heightened index of suspicion for uncommon complications, such as BRASH syndrome.
Nursing staff must also be made aware of these potentially harmful adverse effects as they can alert clinicians to medication combinations that may increase the patient's risk. Pharmacists have an important role in reviewing patients' medications and new prescriptions, avoiding duplicate therapy, and alerting patients to the dangers of polypharmacy. Early identification and diagnosis by any of these healthcare team members leads to the development of appropriate management plans that, in turn, improve patient outcomes.
Media
(Click Image to Enlarge)
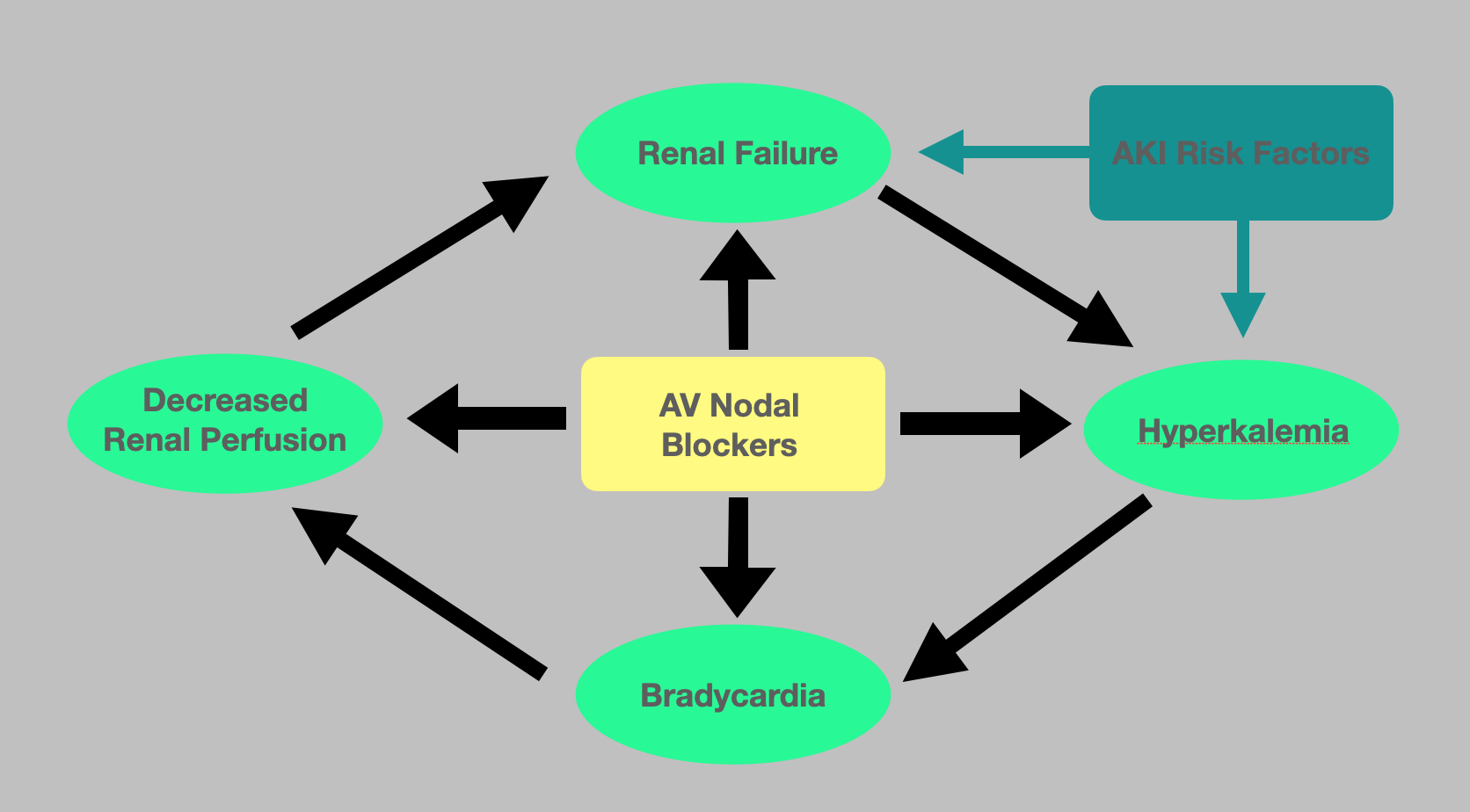
Pathophysiology of BRASH Syndrome. The pathophysiology of BRASH syndrome involves the synergistic effects of atrioventricular nodal blockers directly causing bradycardia and indirectly affecting renal perfusion, leading to hyperkalemia and worsening bradycardia. AKI, acute kidney injury; AV, atrioventricular.
Contributed by K Lizyness, DO
(Click Image to Enlarge)
References
Arif AW, Khan MS, Masri A, Mba B, Talha Ayub M, Doukky R. BRASH Syndrome with Hyperkalemia: An Under-Recognized Clinical Condition. Methodist DeBakey cardiovascular journal. 2020 Jul-Sep:16(3):241-244. doi: 10.14797/mdcj-16-3-241. Epub [PubMed PMID: 33133361]
Srivastava S, Kemnic T, Hildebrandt KR. BRASH syndrome. BMJ case reports. 2020 Feb 23:13(2):. doi: 10.1136/bcr-2019-233825. Epub 2020 Feb 23 [PubMed PMID: 32094236]
Level 3 (low-level) evidenceFarkas JD, Long B, Koyfman A, Menson K. BRASH Syndrome: Bradycardia, Renal Failure, AV Blockade, Shock, and Hyperkalemia. The Journal of emergency medicine. 2020 Aug:59(2):216-223. doi: 10.1016/j.jemermed.2020.05.001. Epub 2020 Jun 18 [PubMed PMID: 32565167]
Bowman BN, Nawarskas JJ, Anderson JR. Treating Diuretic Resistance: An Overview. Cardiology in review. 2016 Sep-Oct:24(5):256-60. doi: 10.1097/CRD.0000000000000116. Epub [PubMed PMID: 27465540]
Level 3 (low-level) evidenceJolly SR, Keaton N, Movahed A, Rose GC, Reeves WC. Effect of hyperkalemia on experimental myocardial depression by verapamil. American heart journal. 1991 Feb:121(2 Pt 1):517-23 [PubMed PMID: 1990757]
Level 3 (low-level) evidencePalmisano P, Accogli M, Zaccaria M, Vergari A, De Masi Gde L, Negro L, De Blasi S. Relationship between seasonal weather changes, risk of dehydration, and incidence of severe bradyarrhythmias requiring urgent temporary transvenous cardiac pacing in an elderly population. International journal of biometeorology. 2014 Sep:58(7):1513-20. doi: 10.1007/s00484-013-0755-7. Epub 2013 Oct 22 [PubMed PMID: 24146304]
Ravioli S, Woitok BK, Lindner G. BRASH syndrome - fact or fiction? A first analysis of the prevalence and relevance of a newly described syndrome. European journal of emergency medicine : official journal of the European Society for Emergency Medicine. 2021 Apr 1:28(2):153-155. doi: 10.1097/MEJ.0000000000000762. Epub [PubMed PMID: 33674517]
Weiss JN, Qu Z, Shivkumar K. Electrophysiology of Hypokalemia and Hyperkalemia. Circulation. Arrhythmia and electrophysiology. 2017 Mar:10(3):. doi: 10.1161/CIRCEP.116.004667. Epub [PubMed PMID: 28314851]
Hegazi MO, Aldabie G, Al-Mutairi S, El Sayed A. Junctional bradycardia with verapamil in renal failure--care required even with mild hyperkalaemia. Journal of clinical pharmacy and therapeutics. 2012 Dec:37(6):726-8. doi: 10.1111/j.1365-2710.2012.01352.x. Epub 2012 May 9 [PubMed PMID: 22568727]
Level 3 (low-level) evidenceChakraborty A, Patel P, Can AS. Calcium Gluconate. StatPearls. 2025 Jan:(): [PubMed PMID: 32491395]
Sidhu S, Marine JE. Evaluating and managing bradycardia. Trends in cardiovascular medicine. 2020 Jul:30(5):265-272. doi: 10.1016/j.tcm.2019.07.001. Epub 2019 Jul 9 [PubMed PMID: 31311698]
Dépret F, Peacock WF, Liu KD, Rafique Z, Rossignol P, Legrand M. Management of hyperkalemia in the acutely ill patient. Annals of intensive care. 2019 Feb 28:9(1):32. doi: 10.1186/s13613-019-0509-8. Epub 2019 Feb 28 [PubMed PMID: 30820692]
Palmer BF, Carrero JJ, Clegg DJ, Colbert GB, Emmett M, Fishbane S, Hain DJ, Lerma E, Onuigbo M, Rastogi A, Roger SD, Spinowitz BS, Weir MR. Clinical Management of Hyperkalemia. Mayo Clinic proceedings. 2021 Mar:96(3):744-762. doi: 10.1016/j.mayocp.2020.06.014. Epub 2020 Nov 5 [PubMed PMID: 33160639]
Aziz EF, Javed F, Korniyenko A, Pratap B, Cordova JP, Alviar CL, Herzog E. Mild hyperkalemia and low eGFR a tedious recipe for cardiac disaster in the elderly: an unusual reversible cause of syncope and heart block. Heart international. 2011:6(2):e12. doi: 10.4081/hi.2011.e12. Epub 2011 Oct 26 [PubMed PMID: 22049311]
Level 3 (low-level) evidencePrough DS, Bidani A. Hyperchloremic metabolic acidosis is a predictable consequence of intraoperative infusion of 0.9% saline. Anesthesiology. 1999 May:90(5):1247-9 [PubMed PMID: 10319767]
Jaber S, Paugam C, Futier E, Lefrant JY, Lasocki S, Lescot T, Pottecher J, Demoule A, Ferrandière M, Asehnoune K, Dellamonica J, Velly L, Abback PS, de Jong A, Brunot V, Belafia F, Roquilly A, Chanques G, Muller L, Constantin JM, Bertet H, Klouche K, Molinari N, Jung B, BICAR-ICU Study Group. Sodium bicarbonate therapy for patients with severe metabolic acidaemia in the intensive care unit (BICAR-ICU): a multicentre, open-label, randomised controlled, phase 3 trial. Lancet (London, England). 2018 Jul 7:392(10141):31-40. doi: 10.1016/S0140-6736(18)31080-8. Epub 2018 Jun 14 [PubMed PMID: 29910040]
Level 1 (high-level) evidenceJentzer JC, DeWald TA, Hernandez AF. Combination of loop diuretics with thiazide-type diuretics in heart failure. Journal of the American College of Cardiology. 2010 Nov 2:56(19):1527-34. doi: 10.1016/j.jacc.2010.06.034. Epub [PubMed PMID: 21029871]
Level 3 (low-level) evidenceLevy B, Perez P, Perny J, Thivilier C, Gerard A. Comparison of norepinephrine-dobutamine to epinephrine for hemodynamics, lactate metabolism, and organ function variables in cardiogenic shock. A prospective, randomized pilot study. Critical care medicine. 2011 Mar:39(3):450-5. doi: 10.1097/CCM.0b013e3181ffe0eb. Epub [PubMed PMID: 21037469]
Level 1 (high-level) evidenceLauterbach M. Clinical toxicology of beta-blocker overdose in adults. Basic & clinical pharmacology & toxicology. 2019 Aug:125(2):178-186. doi: 10.1111/bcpt.13231. Epub 2019 Apr 15 [PubMed PMID: 30916882]
Graudins A, Lee HM, Druda D. Calcium channel antagonist and beta-blocker overdose: antidotes and adjunct therapies. British journal of clinical pharmacology. 2016 Mar:81(3):453-61. doi: 10.1111/bcp.12763. Epub 2015 Oct 30 [PubMed PMID: 26344579]
Littmann L, Gibbs MA. Electrocardiographic manifestations of severe hyperkalemia. Journal of electrocardiology. 2018 Sep-Oct:51(5):814-817. doi: 10.1016/j.jelectrocard.2018.06.018. Epub 2018 Jul 4 [PubMed PMID: 30177318]