Anesthesia for Patients With Myasthenia Gravis
Anesthesia for Patients With Myasthenia Gravis
Introduction
Neuromuscular diseases significantly contribute to both morbidity and mortality. Myasthenia gravis, an autoimmune disorder characterized by skeletal muscle weakness and fatigue, results from the destruction of post-synaptic nicotinic acetylcholine receptors at the neuromuscular junction, impairing normal muscle function.[1]
Patients with myasthenia gravis typically experience worsening muscle weakness as the day progresses or after physical exercise or exertion, with symptoms improving after rest. Deep tendon reflexes remain intact, and autonomic dysfunction is uncommon. However, as the disease advances, respiratory muscle involvement becomes a significant concern, increasing the risk of perioperative respiratory compromise.
The complex interplay between disease progression and the effects of commonly used treatments—such as acetylcholinesterase inhibitors, corticosteroids, and immunosuppressants—presents unique challenges for anesthesia management. These factors necessitate a carefully tailored approach to anesthetic planning, neuromuscular blockade, and postoperative monitoring to ensure optimal patient safety and outcomes. Lambert-Eaton syndrome, various myasthenic syndromes, and other neuromuscular disorders are not discussed in detail here.
Function
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Function
Myasthenia gravis poses several challenges to anesthesiologists. A thorough understanding of the disease process and pharmacological issues pertaining to treatment is necessary. More than 85% of patients with myasthenia gravis have nicotinic acetylcholine receptor antibodies. However, it is important to note that neuronal nicotinic receptors remain unaffected. This finding indicates that the disease specifically impacts skeletal muscle.
Disease prevalence is estimated to be approximately 1 in 7500 patients. Disease incidence is most common in women in their 20s to 30s, whereas men show a bimodal age pattern, with a peak incidence in their 30s and 60s. Exacerbations and remissions mark the course of the disease. Many triggers of myasthenia gravis exist, including stress, surgery, pregnancy, and drugs, such as antibiotics, rheumatologic agents, and cardiovascular drugs, that can precipitate or exacerbate symptoms.[2]
Classification of myasthenia gravis is primarily based on whether the patient has ocular symptoms only or ocular symptoms with non-ocular weakness. Besides ptosis and diplopia, bulbar involvement is common, affecting pharyngeal and laryngeal muscles. This muscular dysfunction can lead to issues with speech, dysphagia, and an increased risk of pulmonary aspiration. Patients often have more proximal muscle groups and respiratory involvement as the disease progresses. Patients with myasthenia gravis are at an increased risk of postoperative respiratory failure. Myasthenic crisis, an exacerbation of the disease, can result in significant diaphragmatic weakness requiring prolonged mechanical ventilation.
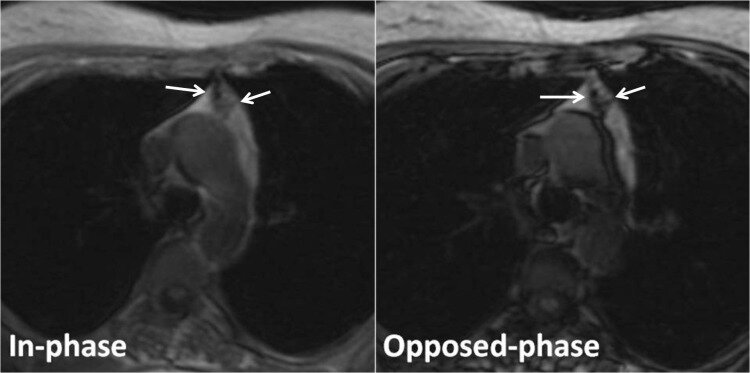
Other autoimmune conditions, such as systemic lupus erythematosus, rheumatoid arthritis, thyroid dysfunction, and diabetes mellitus, are present in 10% of patients. Thymomas are also common, and more than half of patients with myasthenia gravis show some level of thymic follicular hyperplasia (see Image. Hyperplasia in a 40-Year-Old Woman With Myasthenia Gravis). Thymectomy for patients with myasthenia gravis is a relatively common procedure to help relieve symptoms and provide remission for many patients.[3][4]
Issues of Concern
The preoperative evaluation should focus on which muscle groups are affected, the recent course of the disease, current pharmacologic therapy, and any comorbid conditions. Patients with pulmonary or bulbar involvement are at an increased risk of aspiration, and premedication with a proton pump inhibitor, histamine-2 blocker, or prokinetic agent, such as metoclopramide, can be helpful. Avoiding calcium channel blockers and magnesium helps maintain muscle contraction integrity. The mainstay of treatment is acetylcholinesterase inhibitors, primarily pyridostigmine, to help increase acetylcholine at the neuromuscular junction.
Patients should be instructed to continue their myasthenia gravis medications through the preoperative period. Postoperatively, the regimen should be restarted, especially for more generalized and severe diseases, as patients are more likely to decline if treatment is withheld. The acetylcholinesterase inhibitor can be given parenterally, although these should be lowered to one-thirtieth of the oral dose. Excessive administration of these medications can lead to a cholinergic crisis characterized by increased weakness with expected muscarinic effects of miosis, lacrimation, salivation, bradycardia, urination, and defecation. If there is uncertainty about the cause of such an episode, an edrophonium test, also known as the Tensilon test, can be used to differentiate between a myasthenic crisis and a cholinergic crisis.
In a myasthenic crisis, muscle strength increases following the administration of edrophonium. If the Tensilon test is contraindicated, an ice-pack test can also be performed.[5] For moderate-to-severe disease, immunomodulators such as azathioprine, cyclosporine, and corticosteroids are introduced. Patients with respiratory and oropharyngeal weakness or those displaying increased symptoms should be given plasmapheresis or intravenous immune globulin, especially if the patient suffers an acute exacerbation. A discussion with the patient should occur regarding the possible need for prolonged intubation after the procedure. Other drugs that potentiate weakness in these patients include cardiovascular and antiarrhythmic agents, such as beta-blockers, lidocaine, and procainamide, and antibiotics, such as aminoglycosides.
The Levinthal scoring system involves a set of weighted risk factors that predict the increased likelihood of postoperative mechanical ventilation for patients with myasthenia gravis. This system was developed for patients undergoing transcervical thymectomy. However, these risk factors are now widely accepted for use in patients with myasthenia gravis undergoing other procedures requiring general anesthesia with paralysis. These factors can serve as a valuable guide for clinicians during the perioperative period.
Risk factors associated with an increased likelihood for postoperative mechanical ventilation include a duration of disease >6 years, coexisting pulmonary disease, pyridostigmine dose >750 mg/d, and a preoperative vital capacity <2.9 L. More recent studies have demonstrated that a history of previous myasthenic crises and the presence of anti-acetylcholine antibodies were positive indicators of postoperative mechanical ventilation.[6][7]
Lambert-Eaton myasthenic syndrome is often discussed alongside myasthenia gravis but is a distinct disorder with a different pathophysiology. In Lambert-Eaton syndrome, muscle weakness results from antibodies targeting presynaptic calcium channels at the neuromuscular junction. Patients often exhibit improved muscle strength with repeated use, decreased reflexes, and a higher prevalence in males than females. Proximal muscles are affected more in Lambert-Eaton myasthenic syndrome than in myasthenia gravis. The condition is associated with small-cell lung cancer, and patients are often resistant to nondepolarizing neuromuscular blockers.[8]
Clinical Significance
Patients with myasthenia gravis are susceptible to the depressive respiratory effects of benzodiazepines and opiates. Caution must be exercised when administering these medications. Due to the reduced availability of acetylcholine, these patients tend to be resistant to depolarizing neuromuscular blocking agents, such as succinylcholine, and highly sensitive to nondepolarizing neuromuscular blocking agents, such as rocuronium. Therefore, careful dosing and monitoring are essential during the induction and maintenance of general anesthesia when using paralytics.[2]
Depending on the procedure, volatile anesthetics may provide sufficient muscle relaxation and potentially eliminate the need for neuromuscular blocking agents. If needed, a shorter-acting neuromuscular blocking agent is preferred. Physiological stress can exacerbate myasthenia gravis, and general anesthesia during surgery is a known source of physiological stress. Multimodal anesthesia aids in maintaining adequate levels of analgesia and anesthesia for these patients. This approach can include an epidural to help manage the sympathetic nervous system's response to surgery and anesthesia, thereby reducing the overall stress response.[9]
No single specific volatile agent is superior to others, and most are considered safe overall. Patients with neuromuscular disorders, such as myasthenia gravis, often show a pattern of restrictive lung disease as respiratory muscles become involved. The anesthesia provider should remember this fact when providing positive pressure ventilation.
A complete reversal of rocuronium or vecuronium is possible with sugammadex while avoiding the potential drawbacks of neostigmine in this patient population. Several reports show variability in reversing neuromuscular blockade even with the use of sugammadex.[10] Therefore, the healthcare team must remain vigilant in monitoring the patient for any possible weakness. Awake extubation with confirmation of good ventilatory function is ideal in patients with myasthenia gravis who underwent general anesthesia using paralytic agents. Regardless, the extubation criteria are similar for all patients. Any other factors concerning the need for a slower emergence with intensive care unit (ICU) support must be weighed by the anesthesiologist and surgical team. Patients undergoing thymectomy may have shorter ICU stays and fewer postoperative pulmonary complications when extubated <6 hours from the end of surgery.[11]
Myasthenic crisis is a life-threatening condition in patients with myasthenia gravis, resulting in respiratory insufficiency requiring ventilation. This condition typically occurs within the first 2 to 3 years of disease onset and is triggered by factors such as respiratory infections. Older patients are more susceptible to myasthenic crisis, and it can be the first presentation of myasthenia gravis in some cases. Myasthenic crisis is characterized by respiratory and upper airway muscle weakness, leading to reduced lung volume and respiratory obstruction. The exact molecular mechanisms underlying myasthenic crisis are not well understood. Common triggers of myasthenic crisis include respiratory infections, medication changes, surgery, and emotional distress. Risk factors for myasthenic crisis include a history of previous myasthenic crisis, oropharyngeal weakness, severe myasthenia gravis symptoms, muscle-specific kinase antibody positivity, or thymoma. Most cases of myasthenic crisis are not sudden and can be prevented with timely intervention.
Treatment involves airway management, removal of triggers, and the use of plasmapheresis or intravenous immune globulin. Most patients can be weaned from mechanical ventilation within a month, and the overall outcomes of myasthenic crisis are generally favorable. The decision to intubate a patient with a myasthenic crisis is based on clinical evaluation rather than specific parameters, and mechanical ventilation settings should be adjusted based on the patient's condition. Weaning from mechanical ventilation should proceed cautiously, and tracheostomy may be considered in cases of prolonged ventilation. Noninvasive ventilation can be used both before and after intubation to support the respiratory system. This therapy has been successful in preventing intubation in some cases but may fail in others. A high-flow nasal cannula can also be considered in certain situations.[12][13]
Aging leads to a decline in organ function and tolerance to anesthesia and surgery, making older individuals more susceptible to complications. Patients with myasthenia gravis, especially older individuals, are at risk of postoperative myasthenia gravis crisis, and surgery for myasthenia gravis has a high mortality rate. Thymectomy is a treatment option for older patients with myasthenia gravis, especially those with specific associated conditions.
A study conducted on 564 patients with myasthenia gravis who underwent thymectomy categorized individuals into 2 groups—those aged 65 or older (84 cases) and those younger than 65 (481 cases). Perioperative information, such as gender, age, thymoma presence, Osserman classification, lung function, and postoperative complications, was recorded. The study found that surgery was associated with higher risks and complications in older patients with myasthenia gravis. Factors such as age, thymoma presence, lung function, and surgical approach were linked to postoperative complications. The incidence and severity of complications were higher in the older group, particularly for postoperative myasthenic crisis. The study also emphasized the importance of preoperative evaluation and management of comorbidities in older patients, recommending measures to control myasthenia gravis symptoms, adjust medication dosages, and address respiratory complications. Findings suggest that with thorough evaluation, preparation, and close monitoring, older individuals with myasthenia gravis can safely undergo thymectomy. However, surgical indications for each patient should be assessed individually, considering the surgical risk, myasthenia gravis improvement time, and life expectancy.[14][15]
Other Issues
Managing a parturient with myasthenia gravis requires special considerations. These patients are prone to disease exacerbations, with increased muscle weakness often observed from the third trimester through the early postpartum period. Like other individuals with myasthenia gravis, they face an elevated risk of respiratory compromise. Among anesthesia options, epidural anesthesia is considered relatively safe and may be preferred over spinal or general anesthesia.
Maternal antibodies cross the placenta, potentially causing neonatal myasthenia gravis, which can present as feeding difficulties and respiratory dysfunction. Although this condition is typically transient, lasting only a few weeks, some neonates may require intubation and mechanical ventilation for respiratory support.[16] Symptoms may not appear until up to 2 days after birth, making close postnatal monitoring essential.
Enhancing Healthcare Team Outcomes
The treatment of patients with myasthenia gravis who are undergoing surgery requires a coordinated team effort involving the surgeon, anesthesia provider, pharmacists, and nursing staff. Team members should discuss the patient's current health and disease status thoroughly. Before any procedure, the patient's disease should be optimized, beginning with the patient's primary care provider or neurologist.
Informed consent should include a discussion of the increased likelihood of postoperative mechanical ventilation and ICU admission. Postanesthesia care unit staff is crucial for monitoring for signs and symptoms of cholinergic or myasthenic crisis postoperatively. In addition, active management of airway secretions is essential to reduce aspiration events postoperatively. A collaborative model with timely and accurate communication ensures the safest and best possible perioperative care for patients with myasthenia gravis.
Skills
Managing anesthesia in patients with myasthenia gravis requires specialized knowledge and expertise. Management, treatment, and prevention of perioperative adverse events associated with anesthesia in these patients are essential. Managing myasthenia gravis is crucial for healthcare providers involved in perioperative care. Therefore, myasthenia gravis management crosses over the breadth and depth of healthcare situations and settings in the perioperative environment.
Strategy
Maintaining continuous closed-loop communication is ideal among all members of the perioperative care team regarding the needs, techniques, and potential management issues associated with myasthenia gravis. Effective collaboration, interdisciplinary teamwork, and a culture of integrity and quality improvement are essential to a successful strategy.
Ethics
Obtaining thorough informed consent from either patients or their authorized decision-makers is essential before administering anesthesia to patients with myasthenia gravis. All team members should feel empowered to voice any concerns to the team or patient, fostering transparency, stakeholder engagement, and early identification of potential issues.
Responsibilities
All team members must communicate their concerns, responsibilities, and activities with all other team members contemporaneously and as indicated throughout the perioperative period, based on their professional discretion. These team members are responsible for maintaining professional interactions, respecting different opinions, and engaging in shared decision-making with all stakeholders involved in patient care and welfare.
Interprofessional Communication
All team members should respect the free flow of information and concerns without creating hostility. Effective monitoring should be diligent and consistent among team members. Clear and closed-loop professional communication between interdisciplinary team members and among all team members is essential.
Care Coordination
All interprofessional team members should prioritize collaboration by ensuring their actions support the work of others and by avoiding behaviors that create additional challenges or increase the workload for their colleagues.
Media
(Click Image to Enlarge)
References
Bubuioc AM, Kudebayeva A, Turuspekova S, Lisnic V, Leone MA. The epidemiology of myasthenia gravis. Journal of medicine and life. 2021 Jan-Mar:14(1):7-16. doi: 10.25122/jml-2020-0145. Epub [PubMed PMID: 33767779]
Sheikh S, Alvi U, Soliven B, Rezania K. Drugs That Induce or Cause Deterioration of Myasthenia Gravis: An Update. Journal of clinical medicine. 2021 Apr 6:10(7):. doi: 10.3390/jcm10071537. Epub 2021 Apr 6 [PubMed PMID: 33917535]
Al-Bulushi A, Al Salmi I, Al Rahbi F, Farsi AA, Hannawi S. The role of thymectomy in myasthenia gravis: A programmatic approach to thymectomy and perioperative management of myasthenia gravis. Asian journal of surgery. 2021 Jun:44(6):819-828. doi: 10.1016/j.asjsur.2020.12.013. Epub 2021 Feb 9 [PubMed PMID: 33579606]
Wolfe GI, Kaminski HJ, Aban IB, Minisman G, Kuo HC, Marx A, Ströbel P, Mazia C, Oger J, Cea JG, Heckmann JM, Evoli A, Nix W, Ciafaloni E, Antonini G, Witoonpanich R, King JO, Beydoun SR, Chalk CH, Barboi AC, Amato AA, Shaibani AI, Katirji B, Lecky BR, Buckley C, Vincent A, Dias-Tosta E, Yoshikawa H, Waddington-Cruz M, Pulley MT, Rivner MH, Kostera-Pruszczyk A, Pascuzzi RM, Jackson CE, Garcia Ramos GS, Verschuuren JJ, Massey JM, Kissel JT, Werneck LC, Benatar M, Barohn RJ, Tandan R, Mozaffar T, Conwit R, Odenkirchen J, Sonett JR, Jaretzki A 3rd, Newsom-Davis J, Cutter GR, MGTX Study Group. Randomized Trial of Thymectomy in Myasthenia Gravis. The New England journal of medicine. 2016 Aug 11:375(6):511-22. doi: 10.1056/NEJMoa1602489. Epub [PubMed PMID: 27509100]
Level 1 (high-level) evidenceBeloor Suresh A, Asuncion RMD. Myasthenia Gravis. StatPearls. 2025 Jan:(): [PubMed PMID: 32644757]
Chigurupati K, Gadhinglajkar S, Sreedhar R, Nair M, Unnikrishnan M, Pillai M. Criteria for Postoperative Mechanical Ventilation After Thymectomy in Patients With Myasthenia Gravis: A Retrospective Analysis. Journal of cardiothoracic and vascular anesthesia. 2018 Feb:32(1):325-330. doi: 10.1053/j.jvca.2017.06.045. Epub 2017 Jun 27 [PubMed PMID: 29221974]
Level 2 (mid-level) evidenceLeventhal SR, Orkin FK, Hirsh RA. Prediction of the need for postoperative mechanical ventilation in myasthenia gravis. Anesthesiology. 1980 Jul:53(1):26-30 [PubMed PMID: 7386905]
Ivanovski T, Miralles F. Lambert-Eaton Myasthenic syndrome: early diagnosis is key. Degenerative neurological and neuromuscular disease. 2019:9():27-37. doi: 10.2147/DNND.S192588. Epub 2019 May 13 [PubMed PMID: 31191084]
Liu XZ, Wei CW, Wang HY, Ge YH, Chen J, Wang J, Zhang Y. Effects of General-epidural Anaesthesia on Haemodynamics in Patients with Myasthenia Gravis. The West Indian medical journal. 2015 Mar:64(2):99-103. doi: 10.7727/wimj.2013.054. Epub 2015 Jan 28 [PubMed PMID: 26360681]
Fernandes HDS, Ximenes JLS, Nunes DI, Ashmawi HA, Vieira JE. Failure of reversion of neuromuscular block with sugammadex in patient with myasthenia gravis: case report and brief review of literature. BMC anesthesiology. 2019 Aug 17:19(1):160. doi: 10.1186/s12871-019-0829-0. Epub 2019 Aug 17 [PubMed PMID: 31421671]
Level 3 (low-level) evidenceChen L, Xie W, Zheng D, Wang S, Wang G, Sun J, Tai Q, Chen Z. Early extubation after thymectomy is good for the patients with myasthenia gravis. Neurological sciences : official journal of the Italian Neurological Society and of the Italian Society of Clinical Neurophysiology. 2019 Oct:40(10):2125-2132. doi: 10.1007/s10072-019-03941-0. Epub 2019 Jun 10 [PubMed PMID: 31183676]
Claytor B, Cho SM, Li Y. Myasthenic crisis. Muscle & nerve. 2023 Jul:68(1):8-19. doi: 10.1002/mus.27832. Epub 2023 Apr 28 [PubMed PMID: 37114503]
Jiao P, Wu F, Liu Y, Wu J, Sun Y, Tian W, Yu H, Huang C, Li D, Wu Q, Ma C, Tong H. Analysis of influencing factors of postoperative myasthenic crisis in 564 patients with myasthenia gravis in a single center. Thoracic cancer. 2023 Feb:14(5):517-523. doi: 10.1111/1759-7714.14774. Epub 2023 Jan 3 [PubMed PMID: 36594520]
Jiao P, Wu F, Wu J, Sun Y, Tian W, Yu H, Huang C, Li D, Wu Q, Ma C, Tong H. Surgical safety analysis and clinical experience sharing of myasthenia gravis patients aged 65 and over. Thoracic cancer. 2023 Mar:14(8):717-723. doi: 10.1111/1759-7714.14799. Epub 2023 Jan 23 [PubMed PMID: 36691325]
van den Bersselaar LR, Gubbels M, Riazi S, Heytens L, Jungbluth H, Voermans NC, Snoeck MMJ. Mapping the current evidence on the anesthetic management of adult patients with neuromuscular disorders-a scoping review. Canadian journal of anaesthesia = Journal canadien d'anesthesie. 2022 Jun:69(6):756-773. doi: 10.1007/s12630-022-02230-3. Epub 2022 Mar 23 [PubMed PMID: 35322378]
Level 2 (mid-level) evidenceBardhan M, Dogra H, Samanta D. Neonatal Myasthenia Gravis. StatPearls. 2025 Jan:(): [PubMed PMID: 32644361]