Epidemiologic and Etiologic Considerations of Obesity
Epidemiologic and Etiologic Considerations of Obesity
Introduction
For much of the 20th century, cigarette smoking was the most common preventable cause of morbidity and mortality. However, a decrease in tobacco use and a simultaneous increase in obesity has been observed.[1][2] In simple terms, obesity occurs when caloric intake exceeds energy expenditure.[3] This chronic and often progressive condition is debilitating and has far-reaching implications extending beyond the afflicted individual and into society.[4] The causes of obesity involve genetics, socioeconomic level, diet, physical activity, medications, comorbid conditions, and culture.[5]
Genome-wide databases have identified more than 500 specific gene loci related to the development of obesity. Those genes can have profound lifelong implications (eg, Prader-Willi syndrome and Bardet-Biedl syndrome).[6] Researchers have discovered racial disparities in the development of obesity.[7] Various prenatal and childhood factors and normal aging are associated with the development of adult obesity. In addition, those with sedentary lifestyles tend to make poor choices in food selection and adopt a daily caloric intake that does not match their energy expenditure. As a result, this caloric excess is the primary driver for developing obesity.[8]
Health professionals should be aware of various medications known to cause obesity. In addition, various comorbid conditions affecting the hypothalamic-pituitary axis (HPA) can contribute to obesity onset. Recently, investigators have become interested in the role of the gut microbiome in the development of obesity, particularly in the proportion of 2 bacterial strains, Bacteroides and Firmicutes, in the gut microbiome and the role of endocrine-disrupting chemicals.[9][10][11] Further, the development of chronic obesity produces a cascade of downstream molecular targets, leading to the development of dyslipidemia, hypertension, and diabetes. This unique cluster of conditions in conjunction with obesity has been coined metabolic syndrome and substantially increases morbidity and mortality.[12] The microbiome can contribute to obesity by modifying metabolic pathways, including the endocannabinoid system, which impacts energy use and can change the sensation of satiety. Some gut bacteria can also digest fiber and starch through fermentation and hydrolysis.[13]
The above-reviewed factors are quintessential to understanding how to treat this chronic disease. Previously, healthcare professionals have struggled with understanding these known causes of obesity. These deficiencies have led to missed opportunities and failure to educate patients on up-to-date treatment options for chronic obesity.[14]
Issues of Concern
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Issues of Concern
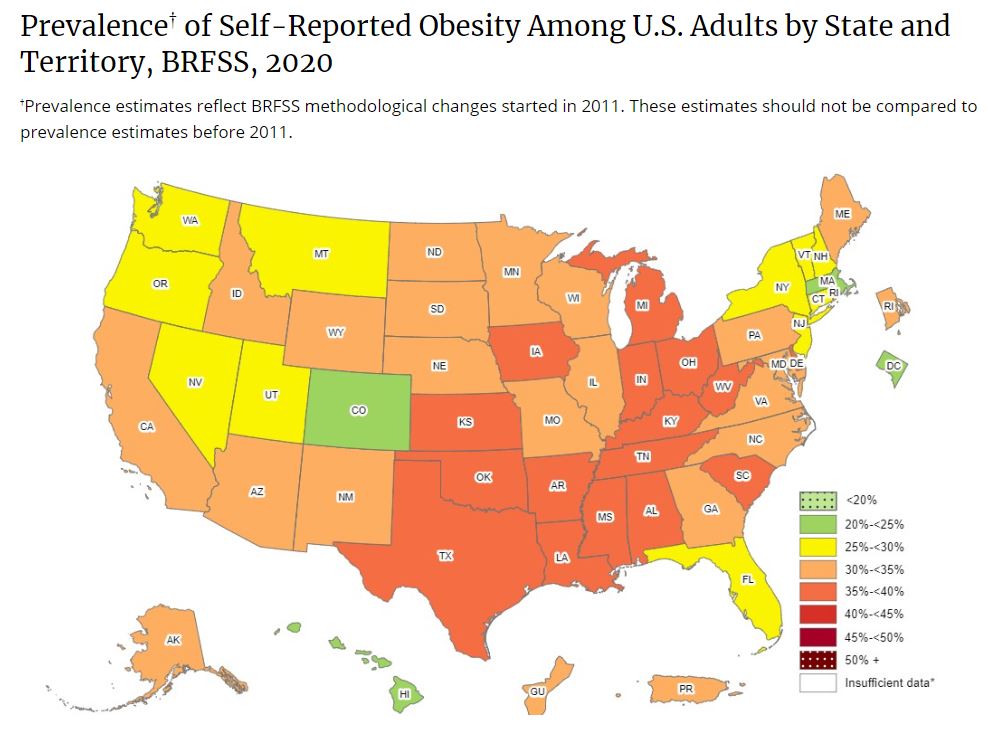
Historically, malnourishment was considered the most medical issue in society. Fortunately, social programs, charitable organizations, improved technology, and increased availability of prepackaged foods in resource-rich societies have made nutrition more accessible. Notwithstanding, the relative increased availability has ushered in a fundamental shift in population health, replacing caloric deficit with caloric excess.[15] Currently, increasing rates of obesity, characterized by higher body mass index (BMI), expanding waistlines, and a growing prevalence of related chronic medical conditions, are widely observed (see Image. Prevalence of Self-Reported Obesity). Obesity is considered a multifactorial, progressive, and chronic systemic medical problem that can often be complex and relatively difficult to treat.
From a clinical perspective, patients are often aware of their need to lose weight and have likely received advice from clinicians to "take charge" of their weight. Unfortunately, few nonsurgical strategies have been established to ensure successful and sustainable weight loss. As fundamentally habitual beings, humans require interventions to disrupt the reward-feedback loop associated with eating, which is paramount to controlling dietary behaviors. Therefore, breaking the reward-feedback loop associated with eating is paramount to controlling dietary habits.[16] Achieving this control requires understanding the complex behavioral elements of obesity. Clinicians must begin at the molecular level by discussing how food becomes storable energy and how storable energy in excess becomes fat.
Molecular and Physiological Processes Associated With Fat Storage and Energy Utilization
Food consumption undergoes a rigorous digestion process that breaks down food into nutrients essential for bodily functions. At its core, the fundamental building blocks for all cells in our body rely on fats, carbohydrates, and proteins, which we collectively call macronutrients (ie, macros). Digestion occurs in 3 phases: the cephalic, gastric, and intestinal phases, each involving complex interactions of stimulatory and inhibitory hormones to ensure proper food degradation.
In the cephalic phase, the sight, smell, or thought of food triggers an innate biological reflex. The oral cavity is the location for food's mechanical and early enzymatic breakdown. The cephalic phase activates salivary glands to produce saliva, which contains lingual lipase and amylase, beginning the process of digestion. This reflex response is triggered by the cerebral cortex acting on the hypothalamus and medulla oblongata, sending signals to the vagus nerve to stimulate gastric juices. This signaling action will activate and prime the gastrointestinal tract in preparation for the meal. The gastric phase begins when masticated food enters the stomach.
A feedback loop activates when stretch receptors in the stomach stimulate a vasovagal reflex, triggering the vagus nerve to increase gastric secretory activity. The gastric lumen is an intricate set of epithelial cells with specific secretory activity. These cells protect the stomach lining and assist in secreting enzymes to aid digestion. The intestinal phase arrives when partially digested foods mixed with gastric juices enter the small intestine, where the systemic absorption of macronutrients begins.[17][18][19]
The outlined digestion process breaks down carbohydrates, starting in the mouth and moving to their absorbable form, monosaccharides, in the small intestine. Fats become reduced to fatty acid chains and monoglycerides by bile salts secreted from the liver and lipase from the pancreas. Finally, protein is broken down into amino acids by pepsin released from the stomach and other protein-degrading enzymes released from the pancreas.[20]
If the individual is fed, the metabolic pathway shifts toward energy storage; in a fasting state, it shifts toward energy consumption through macronutrient oxidation. Through differing metabolic pathways, monosaccharides, fatty acids, and amino acids can produce acetyl-coenzyme A, which can enter the Krebs cycle and eventually generate energy through oxidative phosphorylation via the electron transport chain of adenosine triphosphate.
Glycogen, primarily stored in the liver, can readily raise serum glucose through glycogenolysis. However, glycogen stores are depleted within 12 to 18 hours while fasting. Due to this limitation, Glycogen, stored primarily in the liver, can raise serum glucose through glycogenolysis but is depleted within 12 to 18 hours of fasting. The liver compensates by synthesizing glucose from noncarbohydrate precursors in gluconeogenesis. This process utilizes precursors, eg, glycerol (from fatty acids), lactate (from anaerobic metabolism), and amino acids like glutamine and alanine.[21]
Hormones, including leptin and ghrelin, tightly regulate the digestion process. Leptin, stored in fat cells, suppresses food intake and is considered anorexigenic, while ghrelin, stored in gastrointestinal tract cells, stimulates hunger and is deemed orexigenic. Ghrelin is thought to initiate the cephalic phase of digestion.
However, research has uncovered paradoxes in hormone function in obese individuals. For example, obesity is often associated with elevated leptin levels and reduced ghrelin levels, suggesting leptin resistance, though the exact mechanisms remain unclear.[22] A deeper understanding of the above molecular and physiological responses is essential to treating this chronic disease properly.
Genetic Factors of Obesity
Obesity arises from a complex interplay of genetic, environmental, behavioral, physiological, and sociocultural factors. Studies on twins, adoptees, and families indicate that genetic factors significantly influence body mass index (BMI), with heritability estimates ranging from 40% to 70%.[6][23] Adolescents with 1 obese parent have a 3- to 4-fold higher chance of becoming obese.[24] The risk of obesity is more than 10 times higher if a person has 2 obese biological parents.[6] Unfortunately, even with the extensive composite data of obesity-related gene loci, clinical testing based solely on variations in gene loci currently cannot accurately predict obesity using genes alone.[25][26][25]
The single nucleotide polymorphisms in the fat mass and obesity-associated FTO gene located on chromosome 16 contribute to the development of diabetes indirectly.[23] In another specific condition, genomic imprinting can produce de novo deletions in paternally derived 15q11-q13, a condition known as Prader-Willi syndrome. In addition to a characteristic physical appearance, patients with Prader-Willi syndrome have hyperphagia, which can lead to chronic energy intake/expenditure.[27]
Another gene commonly implicated in monogenic childhood obesity is MC4R, which encodes the melanocortin-4 receptor. The MC4R gene is associated with hyperinsulinemia and increased weight gain in infancy.[28] Leptin, leptin receptor deficiencies, and proopiomelanocortin deficiency are the other genetic causes of obesity. Monogenetic obesity is often severe, manifests at a young age, and is frequently accompanied by other symptoms.
Socioeconomic and Environmental Factors of Obesity
Epidemiological studies have examined how geographical, social, and cultural factors contribute to obesity. These factors often profoundly shape dietary habits and behaviors, leading to unhealthy dietary patterns in certain populations. For instance, individuals with lower awareness of proper nutritional practices may be more susceptible to obesity.[29][30]
In addition, healthy food options are often cost-prohibitive in urban locations. Due to recent global events, the cost of groceries has markedly increased, worsening this disparity in healthy food availability. Low- and middle-income families frequently turn to less costly, calorie-dense, and nutrient-poor options to make ends meet during difficult financial times. In combination with increased stress, socioeconomic factors will play a pivotal role in likely worsening the obesity pandemic across the globe. The worsening of obesity will exacerbate the high rate of chronic, obesity-related conditions, increasing the burden on our already stressed healthcare system.
Advertising that promotes obesity-inducing foods targets specific racial and socioeconomic groups differently. An isolated example of this effect was observed with outdoor food and beverage advertisement density targeting Sacramento, Los Angeles, New Orleans, Philadelphia, Austin, and New York. Geographical areas with low-income Black and Latino populations were found to have a higher density of unhealthy food and beverage advertisements, placing those populations at a significant risk for developing obesity.[31][32][33][34]
The financial barrier to healthy food further exacerbates disparities in obesity prevalence across different communities. For example, Black adults have the highest obesity prevalence in the U.S. at 50%, while non-Hispanic Asian adults have the lowest at 17%.[35] These disparities are closely tied to social determinants of health, eg, limited access to physical activity opportunities, wholesome dietary options, and general health education.
Obesity hypoventilation syndrome (Pickwickian syndrome) results from diminished ventilatory drive and capacity related to obesity. This syndrome is defined as the presence of awake alveolar hypoventilation characterized by daytime hypercapnia (arterial carbon dioxide pressure level >45 mm Hg). The prevalence of obesity hypoventilation syndrome is largely unknown, but based on results from previous studies, it is estimated to be between 20% and 30% of patients who are obese.[36] Please see StatPearls' companion reference, "Obesity Hypoventilation Syndrome," for further discussion of this process.
Prenatal and Childhood Factors
Childhood and infancy experiences may predispose patients to adult obesity. High maternal body mass index, high gestational weight gain, gestational diabetes, and maternal type 2 diabetes may all predispose to adult obesity and metabolic disorders in offspring. By modifying gene expression, maternal obesity may influence the brain regions involved in body weight regulation.[37]
These epigenetic modifications, which may result from an increase in maternal nutrition supply to the developing fetus, promote increased hunger and fat accumulation in children. Children with obesity are 5 times more likely to be obese in adulthood.[38] This lends credibility to starting obesity-related prevention strategies earlier to reduce obesity prevalence in adulthood.
Another area for improvement is by adding healthier school lunch options. Results from a study using nonlinear regression models found that children who attended public schools had a higher BMI regardless of socioeconomic status. Furthermore, those children eligible for free or reduced-cost lunch and breakfast had a higher BMI.[39] Even though additional data is required to determine the impact of limiting the availability of nutrient-poor or high-sugar goods in schools on obesity, some study results have shown a net-positive result.[40][41][42]
Weight and Adulthood Factors of Obesity
Between the ages of 20 and 65, most adults acquire weight steadily. Therefore, the probability of becoming overweight (BMI 25 kg/m2) or obese (BMI 30 kg/m2) during one's lifetime is substantial. The energy imbalance responsible for the rise in obesity over the previous 30 years is estimated to be 100 kcal/day, illustrating that even a small daily positive energy balance may lead to clinically considerable weight gain over time. Weight rises until age 65. After that, the average weight loss is 0.65 kg/year.[43][44] This loss partly results from a loss in muscle mass (sarcopenia), whereas fat mass increases throughout old age, resulting in a reduced correlation between BMI and fat mass.[45] Aging reduces resting and active energy expenditure and may also affect taste and smell, thus reducing food intake.
Dietary Factors of Obesity
Over the last 70 years, a major shift in the global food environment has occurred, with more people having access to processed, high-calorie foods. The United States Department of Agriculture (USDA) statistics on dietary energy supply show that the average daily calorie consumption has risen from 2398 kcal/day/person in the 1970s to 2895 kcal/day/person in the 2000s. This increase in caloric consumption alone can explain the rise in obesity rates seen over this period.
Additionally, the USDA statistics show that Americans consume less fruit, vegetables, and dairy than is advised while consuming more fat, sugar, meats, and grains. The "Western diet" is a colloquial term used to describe a diet high in processed foods (eg, refined sugars and red meats) that may be excessively salty and sugary. This diet has been adopted primarily in the United States but has recently begun spreading into parts of Europe and Asia. In general, this diet type is associated with chronic inflammatory states, which are precursors to several chronic diseases (eg, diabetes, atherosclerosis, and chronic kidney disease).[46] Eaton et al described a "discordance hypothesis" that postulates that the Western diet is the primary driver of increasing BMIs due to a mismatch between calories consumed and burned.[47] Hunter-gatherers in our primitive human species would expend tremendous calories hunting, transporting, and preparing food. However, in our society, very little energy is expended procuring high-calorie foods, contributing to our natural tendency to store adipose tissue. Evidence suggesting that a predominantly plant-based diet is associated with weight loss in obese individuals is limited.[48][49]
Physical Activity Factors of Obesity
Physical activity accounts for the greatest variation in total energy expenditure. Reduced regular physical activity and increased sedentary behavior have been linked to an increased risk of obesity.[50] The Amish community, with a low obesity rate, walks 14,000 to 18,000 steps per day, while the average American takes about 5000 to 6000 steps per day.[51]
Furthermore, occupational and physical activity has decreased in the United States during the last half-century. Half of the American workforce was fairly active in 1960, but by 2010, over 70% were either sedentary or reported little physical activity. This decreased work-related energy expenditure by 140 kcal/day for men and 120 kcal/day for women, further explaining the increasing prevalence of obesity. Adults with physical or mental disabilities are more likely to be obese; those with reduced lower-extremity mobility are at the most significant risk.
Sleep Factors of Obesity
Results from epidemiological studies demonstrate substantial and persistent relationships between reduced nocturnal sleep duration and night shift employment with the development of obesity and other metabolic disorders.[52][53] Night shift employees who spend more time sedentary and less time physically active have increased in numbers recently, resulting in decreased energy expenditure and an increase in the likelihood of obesity. Insufficient sleep also activates brain regions involved with food reward, increasing food consumption, particularly high-fat meals. Thus, adequate sleep and sleep patterns are crucial for lowering adiposity and other metabolic ailments.
Medical Factors of Obesity
Multiple other comorbid conditions are associated with obesity. Hypothyroidism causes weight gain by reducing basal metabolic rate, thus leading to increased adiposity; however, the weight gain is usually modest and improves with treatment. Further, the increased levels of glucocorticoids in Cushing disease stimulate 11-beta-hydroxysteroid dehydrogenase type 1 in visceral fat and increase its lipogenic capability, leading to adiposity. Cushing disease is characterized by progressive central adiposity involving the trunk and abdomen, fat accumulation on the face and neck, and muscle wasting in the extremities. Cushing syndrome may result from the excess (iatrogenically or an overdose) intake of steroids over a long period.
The use of intensive insulin therapy and home glucose monitoring often manages diabetes. Tight control of blood glucose is critical in preventing the vascular complications of the condition. However, insulin therapy itself is associated with weight gain.[54][55] Some studies have suggested that metformin may result in weight loss in patients with type 2 diabetes. However, the evidence to establish this association is limited.[56]
Hypothalamic obesity is uncommon and may result from damage (eg, tumor, irradiation, surgery, or elevated intracranial pressure) to the ventromedial or paraventricular area of the hypothalamus or the amygdala, which regulates metabolic information about nutrient storage and food availability. Damage to the ventromedial hypothalamus results in hyperphagia and decreased energy expenditure, leading to obesity. Hypothalamic obesity can be associated with additional symptoms, including headache, nausea, or blurring of vision.[57]
Clinical Significance
Chronic obesity reverberates across many different areas of Western society. Since 1970, obesity incidence has nearly doubled in the United States, reflecting more than two-thirds of Americans now being overweight. In 2008, the estimated annual cost of obesity was approximately 147 billion dollars, representing almost 10% of healthcare-related spending.[58]
In 2021, that number substantially increased to approximately 260.6 billion dollars.[59] We now know that truncal (abdominal) obesity and increased adipose stores are primary risk factors for developing atherosclerosis, which increases morbidity and mortality from cardiovascular complications. An international classification of obesity exists, with an associated likelihood of morbidity (see Table. Classification of Overweight and Obesity Based on Body Mass Index).[60]
Table 1. Classification of Overweight and Obesity Based on Body Mass Index
|
Class |
Body Mass Index (kg/m2) |
Probability of associated comorbidities |
|
Normal Range |
18.5–24.99 |
Average |
|
Preobese |
25.00-29.99 |
Above-average |
|
Class I Obesity |
30.0–34.99 |
Moderate |
|
Class II Obesity |
35.0-39.99 |
High |
|
Class III Obesity |
>40 |
Very high |
Truncal obesity is a significant risk factor for insulin resistance, which is the primary pathophysiological mechanism underlying the development of type 2 diabetes. Research spanning over a century has explored the complex relationships between obesity, insulin resistance, and related metabolic disorders. In 1988, Dr. Gerald Reaven coined the term "metabolic syndrome," also known as dysmetabolic syndrome X, to describe a cluster of clinical features that increase the risk of cardiovascular disease and diabetes. These features include elevated blood pressure, blood sugar, and serum triglycerides with low high-density lipoprotein (HDL) cholesterol levels and central obesity.[61]
Although being obese is not necessarily a direct causative factor for developing metabolic syndrome, weight loss can be the most beneficial lifestyle modification a person can make to reduce the risk of complications from metabolic syndrome.[62] The clinical classification of metabolic syndrome is debated, and 5 known professional organizations have published several evidence-based criteria to aid clinicians in arriving at this diagnosis. However, all 5 organizations have included truncal obesity as a potential risk factor.[63][64] Patients with obesity have elevated cytokines, including tumor necrosis factor-α and interleukin 1-β, contributing to metabolic syndrome.[9][65] Chronic obesity is also a risk factor for the development of metabolic dysfunction-associated steatotic liver disease (MASLD, formerly nonalcoholic fatty liver disease or NAFLD), leading to an increased risk of all-cause mortality.[63][66]
Obesogenic Factors
Understanding the multifactorial implications of obesity is paramount to accurately diagnosing and treating the condition. In evaluating a patient who demonstrates features of obesity, the first step is to determine whether the cause is predominately lifestyle factors or the result of a secondary etiology (eg, medication use). Conditions for which obesity is a risk factor include:
- Coronary artery disease and stoke
- Hypertension
- Diabetes
- Obstructive sleep apnea
- Malignancy
- Osteoarthritis
- Depression
- Gout [67]
Furthermore, clinicians should determine if the patient is taking a medication known to cause obesity. Certain atypical antipsychotics like olanzapine, quetiapine, and risperidone have been shown to cause a net weight gain, with the most substantial weight gain observed with olanzapine.[68] The anticonvulsant and mood stabilizer gabapentin is associated with a 2.2 kg (approximately 5 lb) weight gain within 2 months. Additionally, clinicians should exercise caution with hypoglycemic agents, particularly in the sulfonylurea class, due to known weight gain associated with tolbutamide (2.8 kg/6 lbs) and glimepiride (2.1 kg/5 lbs). Thiazolidinedione-type hypoglycemic agents have also shown a statistically significant weight gain, particularly pioglitazone (2.6 kg/5.5 lbs).[69]
According to data from the United States Centers for Disease Control and Prevention, from 2015 to 2018, approximately 13.2% of adults were on some form of antidepressant medication, with women predominately receiving prescriptions within this class (17.7%).[70] However, prescribers should be aware of the antidepressants amitryptiline and mirtazapine, which are associated with a 1.8 kg (4 lbs) and 1.5 kg (3.5 lbs) weight gain, respectively. Furthermore, glucocorticoids are commonly prescribed in clinical practice for their potent anti-inflammatory effects, which can be useful for treating a broad range of acute and chronic conditions. Unfortunately, glucocorticoids' mechanism of action typically leads to an approximate 4% to 8% rise in weight gain.[69][71]
Other Issues
By emphasizing diet and lifestyle factors related to obesity, clinicians may encourage patients to follow medical advice and view obesity as a serious but manageable health condition.[72]
Large portion sizes significantly contribute to increased energy intake and obesity. Clinicians, coaches, and nutritionists can educate patients on proper portion sizes, calorie content, glycemic index of foods, and unique calorie needs based on physical activity levels. They may also dispel misconceptions about fad diets and teach strategies to avoid hunger-induced binge eating. Comprehensive weight management counseling should focus on reducing calorie intake and promoting healthier food choices. Patients who consume more low-fat, nutrient-dense foods, eg, fruits, vegetables, legumes, and whole grains, are more likely to meet their nutritional needs. Fresh vegetables, in particular, have high water content, adding weight and volume to meals, which enhances satiety without increasing calorie intake.
Diet and exercise combined remain the most effective treatments for obesity. Physical therapists can encourage nonexercise activity thermogenesis, eg, standing while using the phone or taking stairs instead of elevators, which may result in a 2000 kcal/day difference between active and inactive adults.[73] Walking is another accessible exercise option, and therapists can guide patients toward achievable goals, including adding 1 minute to a 10-minute walk, climbing an additional flight of stairs, or increasing their daily step count by 100. Physical therapists play a crucial role in designing low-impact exercises for patients with musculoskeletal disorders, eg, seated leg movements, to improve mobility and increase activity levels. Obese individuals are particularly prone to knee issues, making targeted interventions essential for enhancing their physical activity.
Mental health disorders can contribute to weight gain and obesity, as eating is often used as a coping mechanism for depression, stress, or anxiety. Research indicates that depression, in particular, can lead to weight gain through poor lifestyle choices, eg, inactivity due to low motivation or the consumption of high-calorie comfort foods. Psychiatric disorders and obesity frequently form a self-perpetuating cycle, where depression and anxiety exacerbate weight gain, which in turn worsens mental health symptoms.
Enhancing Healthcare Team Outcomes
Effectively managing obesity requires an interprofessional, patient-centered approach that encourages collaboration among advanced clinicians, nurses, pharmacists, dietitians, physical therapists, social workers, and psychologists. Advanced clinicians provide medical expertise and motivation, while dietitians create tailored nutritional plans, and physical therapists guide patients in safe, effective physical activity.
Psychologists address underlying mental health conditions, offering strategies to support emotional well-being and behavior change. Social workers help patients access essential resources, eg, mobility aids like walkers or wheelchairs, financial assistance for medications, or gym memberships. Nurses ensure continuity of care by maintaining regular contact, educating patients, and monitoring adherence to treatment plans. This coordinated team approach fosters clear communication among healthcare clinicians, enhances patient outcomes, improves safety, and supports long-term weight management strategies.
Nursing, Allied Health, and Interprofessional Team Interventions
The roles of nursing, allied health, and interprofessional healthcare teams are vital to patient-centered care. As highlighted above, the strategies for reversing obesity in patients require routine follow-up and consistent evaluation by healthcare professionals. Once the plan is implemented and approved by the patient's clinician, support staff can perform routine follow-ups and monitor patients regularly. An example of such a monitoring program might be to check hemoglobin A1c every 3 months, lipid panel every 6 months, and assess patients' barriers to weight loss. Support staff can communicate the results to the patient's clinician to modify the plan and ensure the patient remains on track.
Patients with chronic obesity can often have an unhealthy relationship with food. Cognitive-behavioral therapy can be a valuable tool for helping patients reconcile feelings that may lead to overeating. Various technological applications exist to assist patients with direct access to a therapist.
Applications that measure daily body weight and count calories are primarily effective because they draw users' attention to lifestyle-related details that influence weight gain and loss. For most, cataloging daily caloric intake can be burdensome and arduous, so it is crucial to follow up with patients during their weight-loss journey consistently. Follow-up is the quintessential element of ensuring adherence to a weight-loss plan. Therefore, it is essential to set expectations that this will require consistent virtual or in-person meetings to ensure continued compliance when beginning the weight-loss journey. If the patient is unwilling to agree to follow-up, the patient will likely fail and revert to previous poor lifestyle choices.
Nursing, Allied Health, and Interprofessional Team Monitoring
Telehealth can be a valuable contemporary strategy for managing new patients to a lifestyle modification regimen.[74] Quick face-to-face meetings with weight reporting can be a helpful tool for patients to benchmark their progress and find new motivation.
Media
(Click Image to Enlarge)
References
Flegal KM. The effects of changes in smoking prevalence on obesity prevalence in the United States. American journal of public health. 2007 Aug:97(8):1510-4 [PubMed PMID: 17600266]
Level 2 (mid-level) evidenceHo FK, Celis-Morales C, Petermann-Rocha F, Parra-Soto SL, Lewsey J, Mackay D, Pell JP. Changes over 15 years in the contribution of adiposity and smoking to deaths in England and Scotland. BMC public health. 2021 Feb 11:21(1):169. doi: 10.1186/s12889-021-10167-3. Epub 2021 Feb 11 [PubMed PMID: 33568116]
Schwartz MW, Seeley RJ, Zeltser LM, Drewnowski A, Ravussin E, Redman LM, Leibel RL. Obesity Pathogenesis: An Endocrine Society Scientific Statement. Endocrine reviews. 2017 Aug 1:38(4):267-296. doi: 10.1210/er.2017-00111. Epub [PubMed PMID: 28898979]
Cawley J, Meyerhoefer C. The medical care costs of obesity: an instrumental variables approach. Journal of health economics. 2012 Jan:31(1):219-30. doi: 10.1016/j.jhealeco.2011.10.003. Epub 2011 Oct 20 [PubMed PMID: 22094013]
Grundy SM. Multifactorial causation of obesity: implications for prevention. The American journal of clinical nutrition. 1998 Mar:67(3 Suppl):563S-72S. doi: 10.1093/ajcn/67.3.563S. Epub [PubMed PMID: 9497171]
Loos RJ. The genetics of adiposity. Current opinion in genetics & development. 2018 Jun:50():86-95. doi: 10.1016/j.gde.2018.02.009. Epub 2018 Mar 9 [PubMed PMID: 29529423]
Level 3 (low-level) evidenceFouad MN, Waugaman KJ, Dutton GR. The Complex Contributors to Obesity-Related Health Disparities: Introduction to the Special Issue. American journal of preventive medicine. 2022 Jul:63(1 Suppl 1):S1-S5. doi: 10.1016/j.amepre.2022.03.022. Epub [PubMed PMID: 35725135]
Swinburn B, Sacks G, Ravussin E. Increased food energy supply is more than sufficient to explain the US epidemic of obesity. The American journal of clinical nutrition. 2009 Dec:90(6):1453-6. doi: 10.3945/ajcn.2009.28595. Epub 2009 Oct 14 [PubMed PMID: 19828708]
Hardin BI, Keyes D. Enterohormonal and Microbiota Pathophysiology of Obesity. StatPearls. 2025 Jan:(): [PubMed PMID: 35201732]
Maruvada P, Leone V, Kaplan LM, Chang EB. The Human Microbiome and Obesity: Moving beyond Associations. Cell host & microbe. 2017 Nov 8:22(5):589-599. doi: 10.1016/j.chom.2017.10.005. Epub [PubMed PMID: 29120742]
Gore AC, Chappell VA, Fenton SE, Flaws JA, Nadal A, Prins GS, Toppari J, Zoeller RT. EDC-2: The Endocrine Society's Second Scientific Statement on Endocrine-Disrupting Chemicals. Endocrine reviews. 2015 Dec:36(6):E1-E150. doi: 10.1210/er.2015-1010. Epub 2015 Nov 6 [PubMed PMID: 26544531]
Kahn R, Buse J, Ferrannini E, Stern M, American Diabetes Association, European Association for the Study of Diabetes. The metabolic syndrome: time for a critical appraisal: joint statement from the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes care. 2005 Sep:28(9):2289-304 [PubMed PMID: 16123508]
Sasidharan Pillai S, Gagnon CA, Foster C, Ashraf AP. Exploring the Gut Microbiota: Key Insights Into Its Role in Obesity, Metabolic Syndrome, and Type 2 Diabetes. The Journal of clinical endocrinology and metabolism. 2024 Oct 15:109(11):2709-2719. doi: 10.1210/clinem/dgae499. Epub [PubMed PMID: 39040013]
Phelan SM, Burgess DJ, Yeazel MW, Hellerstedt WL, Griffin JM, van Ryn M. Impact of weight bias and stigma on quality of care and outcomes for patients with obesity. Obesity reviews : an official journal of the International Association for the Study of Obesity. 2015 Apr:16(4):319-26. doi: 10.1111/obr.12266. Epub 2015 Mar 5 [PubMed PMID: 25752756]
Level 2 (mid-level) evidenceEknoyan G. A history of obesity, or how what was good became ugly and then bad. Advances in chronic kidney disease. 2006 Oct:13(4):421-7 [PubMed PMID: 17045228]
Level 3 (low-level) evidenceLean M, Lara J, Hill JO. ABC of obesity. Strategies for preventing obesity. BMJ (Clinical research ed.). 2006 Nov 4:333(7575):959-62 [PubMed PMID: 17082548]
Zafra MA, Molina F, Puerto A. The neural/cephalic phase reflexes in the physiology of nutrition. Neuroscience and biobehavioral reviews. 2006:30(7):1032-44 [PubMed PMID: 16678262]
Patricia JJ, Dhamoon AS. Physiology, Digestion. StatPearls. 2025 Jan:(): [PubMed PMID: 31334962]
Forssell H. Gastric mucosal defence mechanisms: a brief review. Scandinavian journal of gastroenterology. Supplement. 1988:155():23-8 [PubMed PMID: 3072665]
Goodman BE. Insights into digestion and absorption of major nutrients in humans. Advances in physiology education. 2010 Jun:34(2):44-53. doi: 10.1152/advan.00094.2009. Epub [PubMed PMID: 20522896]
Level 3 (low-level) evidenceMelkonian EA, Asuka E, Schury MP. Physiology, Gluconeogenesis. StatPearls. 2024 Jan:(): [PubMed PMID: 31082163]
Klok MD, Jakobsdottir S, Drent ML. The role of leptin and ghrelin in the regulation of food intake and body weight in humans: a review. Obesity reviews : an official journal of the International Association for the Study of Obesity. 2007 Jan:8(1):21-34 [PubMed PMID: 17212793]
Frayling TM, Timpson NJ, Weedon MN, Zeggini E, Freathy RM, Lindgren CM, Perry JR, Elliott KS, Lango H, Rayner NW, Shields B, Harries LW, Barrett JC, Ellard S, Groves CJ, Knight B, Patch AM, Ness AR, Ebrahim S, Lawlor DA, Ring SM, Ben-Shlomo Y, Jarvelin MR, Sovio U, Bennett AJ, Melzer D, Ferrucci L, Loos RJ, Barroso I, Wareham NJ, Karpe F, Owen KR, Cardon LR, Walker M, Hitman GA, Palmer CN, Doney AS, Morris AD, Smith GD, Hattersley AT, McCarthy MI. A common variant in the FTO gene is associated with body mass index and predisposes to childhood and adult obesity. Science (New York, N.Y.). 2007 May 11:316(5826):889-94 [PubMed PMID: 17434869]
Level 2 (mid-level) evidenceWhitaker KL, Jarvis MJ, Beeken RJ, Boniface D, Wardle J. Comparing maternal and paternal intergenerational transmission of obesity risk in a large population-based sample. The American journal of clinical nutrition. 2010 Jun:91(6):1560-7. doi: 10.3945/ajcn.2009.28838. Epub 2010 Apr 7 [PubMed PMID: 20375189]
Level 2 (mid-level) evidenceZhang Y, Kutateladze TG. Diet and the epigenome. Nature communications. 2018 Aug 28:9(1):3375. doi: 10.1038/s41467-018-05778-1. Epub 2018 Aug 28 [PubMed PMID: 30154441]
Bordoni L, Agostinho de Sousa J, Zhuo J, von Meyenn F. Evaluating the connection between diet quality, EpiNutrient intake and epigenetic age: an observational study. The American journal of clinical nutrition. 2024 Nov:120(5):1143-1155. doi: 10.1016/j.ajcnut.2024.08.033. Epub 2024 Oct 11 [PubMed PMID: 39510725]
Level 2 (mid-level) evidenceButler MG. Prader-Willi Syndrome: Obesity due to Genomic Imprinting. Current genomics. 2011 May:12(3):204-15. doi: 10.2174/138920211795677877. Epub [PubMed PMID: 22043168]
Doulla M, McIntyre AD, Hegele RA, Gallego PH. A novel MC4R mutation associated with childhood-onset obesity: A case report. Paediatrics & child health. 2014 Dec:19(10):515-8 [PubMed PMID: 25587224]
Level 3 (low-level) evidenceOgden CL, Fakhouri TH, Carroll MD, Hales CM, Fryar CD, Li X, Freedman DS. Prevalence of Obesity Among Adults, by Household Income and Education - United States, 2011-2014. MMWR. Morbidity and mortality weekly report. 2017 Dec 22:66(50):1369-1373. doi: 10.15585/mmwr.mm6650a1. Epub 2017 Dec 22 [PubMed PMID: 29267260]
Caprio S, Daniels SR, Drewnowski A, Kaufman FR, Palinkas LA, Rosenbloom AL, Schwimmer JB. Influence of race, ethnicity, and culture on childhood obesity: implications for prevention and treatment: a consensus statement of Shaping America's Health and the Obesity Society. Diabetes care. 2008 Nov:31(11):2211-21. doi: 10.2337/dc08-9024. Epub [PubMed PMID: 18955718]
Level 3 (low-level) evidenceCassady DL, Liaw K, Miller LM. Disparities in Obesity-Related Outdoor Advertising by Neighborhood Income and Race. Journal of urban health : bulletin of the New York Academy of Medicine. 2015 Oct:92(5):835-42. doi: 10.1007/s11524-015-9980-1. Epub [PubMed PMID: 26337182]
Lesser LI, Zimmerman FJ, Cohen DA. Outdoor advertising, obesity, and soda consumption: a cross-sectional study. BMC public health. 2013 Jan 10:13():20. doi: 10.1186/1471-2458-13-20. Epub 2013 Jan 10 [PubMed PMID: 23305548]
Level 2 (mid-level) evidenceKwate NO, Lee TH. Ghettoizing outdoor advertising: disadvantage and ad panel density in black neighborhoods. Journal of urban health : bulletin of the New York Academy of Medicine. 2007 Jan:84(1):21-31 [PubMed PMID: 17146710]
Lowery BC, Sloane DC. The prevalence of harmful content on outdoor advertising in Los Angeles: land use, community characteristics, and the spatial inequality of a public health nuisance. American journal of public health. 2014 Apr:104(4):658-64. doi: 10.2105/AJPH.2013.301694. Epub 2014 Feb 13 [PubMed PMID: 24524512]
Level 2 (mid-level) evidenceHales CM, Carroll MD, Fryar CD, Ogden CL. Prevalence of Obesity and Severe Obesity Among Adults: United States, 2017-2018. NCHS data brief. 2020 Feb:(360):1-8 [PubMed PMID: 32487284]
Ghimire P, Sankari A, Antoine MH, Bollu PC, Kaul P. Obesity-Hypoventilation Syndrome. StatPearls. 2025 Jan:(): [PubMed PMID: 31194373]
Şanlı E, Kabaran S. Maternal Obesity, Maternal Overnutrition and Fetal Programming: Effects of Epigenetic Mechanisms on the Development of Metabolic Disorders. Current genomics. 2019 Sep:20(6):419-427. doi: 10.2174/1389202920666191030092225. Epub [PubMed PMID: 32476999]
Simmonds M, Llewellyn A, Owen CG, Woolacott N. Predicting adult obesity from childhood obesity: a systematic review and meta-analysis. Obesity reviews : an official journal of the International Association for the Study of Obesity. 2016 Feb:17(2):95-107. doi: 10.1111/obr.12334. Epub 2015 Dec 23 [PubMed PMID: 26696565]
Level 1 (high-level) evidenceLi J, Hooker NH. Childhood obesity and schools: evidence from the national survey of children's health. The Journal of school health. 2010 Feb:80(2):96-103. doi: 10.1111/j.1746-1561.2009.00471.x. Epub [PubMed PMID: 20236408]
Level 3 (low-level) evidenceTaber DR, Chriqui JF, Perna FM, Powell LM, Chaloupka FJ. Weight status among adolescents in States that govern competitive food nutrition content. Pediatrics. 2012 Sep:130(3):437-44. doi: 10.1542/peds.2011-3353. Epub 2012 Aug 13 [PubMed PMID: 22891223]
Fox MK, Gordon A, Nogales R, Wilson A. Availability and consumption of competitive foods in US public schools. Journal of the American Dietetic Association. 2009 Feb:109(2 Suppl):S57-66. doi: 10.1016/j.jada.2008.10.063. Epub [PubMed PMID: 19166673]
Level 2 (mid-level) evidenceSmith R, Kelly B, Yeatman H, Boyland E. Food Marketing Influences Children's Attitudes, Preferences and Consumption: A Systematic Critical Review. Nutrients. 2019 Apr 18:11(4):. doi: 10.3390/nu11040875. Epub 2019 Apr 18 [PubMed PMID: 31003489]
Level 1 (high-level) evidenceHill JO, Wyatt HR, Reed GW, Peters JC. Obesity and the environment: where do we go from here? Science (New York, N.Y.). 2003 Feb 7:299(5608):853-5 [PubMed PMID: 12574618]
Yang YC, Walsh CE, Johnson MP, Belsky DW, Reason M, Curran P, Aiello AE, Chanti-Ketterl M, Harris KM. Life-course trajectories of body mass index from adolescence to old age: Racial and educational disparities. Proceedings of the National Academy of Sciences of the United States of America. 2021 Apr 27:118(17):. doi: 10.1073/pnas.2020167118. Epub [PubMed PMID: 33875595]
Batsis JA, Villareal DT. Sarcopenic obesity in older adults: aetiology, epidemiology and treatment strategies. Nature reviews. Endocrinology. 2018 Sep:14(9):513-537. doi: 10.1038/s41574-018-0062-9. Epub [PubMed PMID: 30065268]
Okręglicka K. Health effects of changes in the structure of dietary macronutrients intake in western societies. Roczniki Panstwowego Zakladu Higieny. 2015:66(2):97-105 [PubMed PMID: 26024397]
Eaton SB, Eaton SB 3rd, Konner MJ, Shostak M. An evolutionary perspective enhances understanding of human nutritional requirements. The Journal of nutrition. 1996 Jun:126(6):1732-40 [PubMed PMID: 8648449]
Level 3 (low-level) evidenceKeyes D, Patel NI, Correa KA. Overview of Lifestyle Medicine. StatPearls. 2025 Jan:(): [PubMed PMID: 36943990]
Level 3 (low-level) evidenceSchlesinger S, Neuenschwander M, Schwedhelm C, Hoffmann G, Bechthold A, Boeing H, Schwingshackl L. Food Groups and Risk of Overweight, Obesity, and Weight Gain: A Systematic Review and Dose-Response Meta-Analysis of Prospective Studies. Advances in nutrition (Bethesda, Md.). 2019 Mar 1:10(2):205-218. doi: 10.1093/advances/nmy092. Epub [PubMed PMID: 30801613]
Level 1 (high-level) evidencePiercy KL, Troiano RP, Ballard RM, Carlson SA, Fulton JE, Galuska DA, George SM, Olson RD. The Physical Activity Guidelines for Americans. JAMA. 2018 Nov 20:320(19):2020-2028. doi: 10.1001/jama.2018.14854. Epub [PubMed PMID: 30418471]
Roberts WC. The Amish, body weight, and exercise. The American journal of cardiology. 2004 Nov 1:94(9):1221 [PubMed PMID: 15518631]
McHill AW, Wright KP Jr. Role of sleep and circadian disruption on energy expenditure and in metabolic predisposition to human obesity and metabolic disease. Obesity reviews : an official journal of the International Association for the Study of Obesity. 2017 Feb:18 Suppl 1():15-24. doi: 10.1111/obr.12503. Epub [PubMed PMID: 28164449]
Tse LA, Wang C, Rangarajan S, Liu Z, Teo K, Yusufali A, Avezum Á, Wielgosz A, Rosengren A, Kruger IM, Chifamba J, Calik KBT, Yeates K, Zatonska K, AlHabib KF, Yusoff K, Kaur M, Ismail N, Seron P, Lopez-Jaramillo P, Poirier P, Gupta R, Khatib R, Kelishadi R, Lear SA, Choudhury T, Mohan V, Li W, Yusuf S. Timing and Length of Nocturnal Sleep and Daytime Napping and Associations With Obesity Types in High-, Middle-, and Low-Income Countries. JAMA network open. 2021 Jun 1:4(6):e2113775. doi: 10.1001/jamanetworkopen.2021.13775. Epub 2021 Jun 1 [PubMed PMID: 34190997]
Heller S. Weight gain during insulin therapy in patients with type 2 diabetes mellitus. Diabetes research and clinical practice. 2004 Sep:65 Suppl 1():S23-7 [PubMed PMID: 15315867]
Purnell JQ, Zinman B, Brunzell JD, DCCT/EDIC Research Group. The effect of excess weight gain with intensive diabetes mellitus treatment on cardiovascular disease risk factors and atherosclerosis in type 1 diabetes mellitus: results from the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Study (DCCT/EDIC) study. Circulation. 2013 Jan 15:127(2):180-7. doi: 10.1161/CIRCULATIONAHA.111.077487. Epub 2012 Dec 4 [PubMed PMID: 23212717]
Level 1 (high-level) evidenceApolzan JW, Venditti EM, Edelstein SL, Knowler WC, Dabelea D, Boyko EJ, Pi-Sunyer X, Kalyani RR, Franks PW, Srikanthan P, Gadde KM, Diabetes Prevention Program Research Group. Long-Term Weight Loss With Metformin or Lifestyle Intervention in the Diabetes Prevention Program Outcomes Study. Annals of internal medicine. 2019 May 21:170(10):682-690. doi: 10.7326/M18-1605. Epub 2019 Apr 23 [PubMed PMID: 31009939]
Holmer H, Pozarek G, Wirfält E, Popovic V, Ekman B, Björk J, Erfurth EM. Reduced energy expenditure and impaired feeding-related signals but not high energy intake reinforces hypothalamic obesity in adults with childhood onset craniopharyngioma. The Journal of clinical endocrinology and metabolism. 2010 Dec:95(12):5395-402. doi: 10.1210/jc.2010-0993. Epub 2010 Sep 8 [PubMed PMID: 20826582]
Hammond RA, Levine R. The economic impact of obesity in the United States. Diabetes, metabolic syndrome and obesity : targets and therapy. 2010 Aug 30:3():285-95. doi: 10.2147/DMSOTT.S7384. Epub 2010 Aug 30 [PubMed PMID: 21437097]
Cawley J, Biener A, Meyerhoefer C, Ding Y, Zvenyach T, Smolarz BG, Ramasamy A. Direct medical costs of obesity in the United States and the most populous states. Journal of managed care & specialty pharmacy. 2021 Mar:27(3):354-366. doi: 10.18553/jmcp.2021.20410. Epub 2021 Jan 20 [PubMed PMID: 33470881]
. Obesity: preventing and managing the global epidemic. Report of a WHO consultation. World Health Organization technical report series. 2000:894():i-xii, 1-253 [PubMed PMID: 11234459]
Reaven GM. Banting lecture 1988. Role of insulin resistance in human disease. Diabetes. 1988 Dec:37(12):1595-607 [PubMed PMID: 3056758]
James PT, Rigby N, Leach R, International Obesity Task Force. The obesity epidemic, metabolic syndrome and future prevention strategies. European journal of cardiovascular prevention and rehabilitation : official journal of the European Society of Cardiology, Working Groups on Epidemiology & Prevention and Cardiac Rehabilitation and Exercise Physiology. 2004 Feb:11(1):3-8 [PubMed PMID: 15167200]
Quah YV Jr, Poh BK, Ismail MN. Metabolic Syndrome Based on IDF Criteria in a Sample of Normal Weight and Obese School Children. Malaysian journal of nutrition. 2010 Aug:16(2):207-17 [PubMed PMID: 22691926]
Parikh RM, Mohan V. Changing definitions of metabolic syndrome. Indian journal of endocrinology and metabolism. 2012 Jan:16(1):7-12. doi: 10.4103/2230-8210.91175. Epub [PubMed PMID: 22276247]
Sanz Y, Moya-Pérez A. Microbiota, inflammation and obesity. Advances in experimental medicine and biology. 2014:817():291-317. doi: 10.1007/978-1-4939-0897-4_14. Epub [PubMed PMID: 24997040]
Level 3 (low-level) evidencePolyzos SA, Kountouras J, Mantzoros CS. Obesity and nonalcoholic fatty liver disease: From pathophysiology to therapeutics. Metabolism: clinical and experimental. 2019 Mar:92():82-97. doi: 10.1016/j.metabol.2018.11.014. Epub 2018 Nov 29 [PubMed PMID: 30502373]
Pi-Sunyer X. The medical risks of obesity. Postgraduate medicine. 2009 Nov:121(6):21-33. doi: 10.3810/pgm.2009.11.2074. Epub [PubMed PMID: 19940414]
Allison DB, Mentore JL, Heo M, Chandler LP, Cappelleri JC, Infante MC, Weiden PJ. Antipsychotic-induced weight gain: a comprehensive research synthesis. The American journal of psychiatry. 1999 Nov:156(11):1686-96 [PubMed PMID: 10553730]
Domecq JP, Prutsky G, Leppin A, Sonbol MB, Altayar O, Undavalli C, Wang Z, Elraiyah T, Brito JP, Mauck KF, Lababidi MH, Prokop LJ, Asi N, Wei J, Fidahussein S, Montori VM, Murad MH. Clinical review: Drugs commonly associated with weight change: a systematic review and meta-analysis. The Journal of clinical endocrinology and metabolism. 2015 Feb:100(2):363-70. doi: 10.1210/jc.2014-3421. Epub 2015 Jan 15 [PubMed PMID: 25590213]
Level 1 (high-level) evidenceBrody DJ, Gu Q. Antidepressant Use Among Adults: United States, 2015-2018. NCHS data brief. 2020 Sep:(377):1-8 [PubMed PMID: 33054926]
Akalestou E, Genser L, Rutter GA. Glucocorticoid Metabolism in Obesity and Following Weight Loss. Frontiers in endocrinology. 2020:11():59. doi: 10.3389/fendo.2020.00059. Epub 2020 Feb 20 [PubMed PMID: 32153504]
Eckel RH. Preventive cardiology by lifestyle intervention: opportunity and/or challenge? Presidential address at the 2005 American Heart Association Scientific Sessions. Circulation. 2006 Jun 6:113(22):2657-61 [PubMed PMID: 16754814]
Levine JA. Nonexercise activity thermogenesis--liberating the life-force. Journal of internal medicine. 2007 Sep:262(3):273-87 [PubMed PMID: 17697152]
Hinchliffe N, Capehorn MS, Bewick M, Feenie J. The Potential Role of Digital Health in Obesity Care. Advances in therapy. 2022 Oct:39(10):4397-4412. doi: 10.1007/s12325-022-02265-4. Epub 2022 Aug 4 [PubMed PMID: 35925469]
Level 3 (low-level) evidence