Introduction
The thyroid gland is an endocrine organ situated at the lower front of the neck and has a unique butterfly-like shape. The thyroid gland produces thyroid hormones, primarily thyroxine (T4) and triiodothyronine (T3), with their production and secretion being tightly regulated by the hypothalamic-pituitary-thyroid axis. This axis ensures precise control over the levels of these hormones in the body. Thyroid hormones are crucial in regulating body processes such as metabolism, energy generation, and mood.[1] Please see StatPearls' companion resource, "Histology, Thyroid Gland," for more information.
Thyroid function tests (TFTs) are a series of evaluations used to assess the performance of the thyroid gland, particularly when conditions such as hypothyroidism or hyperthyroidism are suspected. These tests primarily involve measuring the levels of thyroid-stimulating hormone (TSH), T3, and T4. TFTs are among the most commonly requested biochemical blood tests due to their high sensitivity, specificity, and established clinical utility. They are used for both diagnosis and monitoring of treatments, including hormone suppression and replacement therapy for thyroid disorders. Additionally, these tests are often performed for conditions such as excessive weight gain, cognitive slowing, atrial fibrillation, and anxiety disorders, which may be associated with excess thyroid hormone imbalances.[2]
Etiology and Epidemiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology and Epidemiology
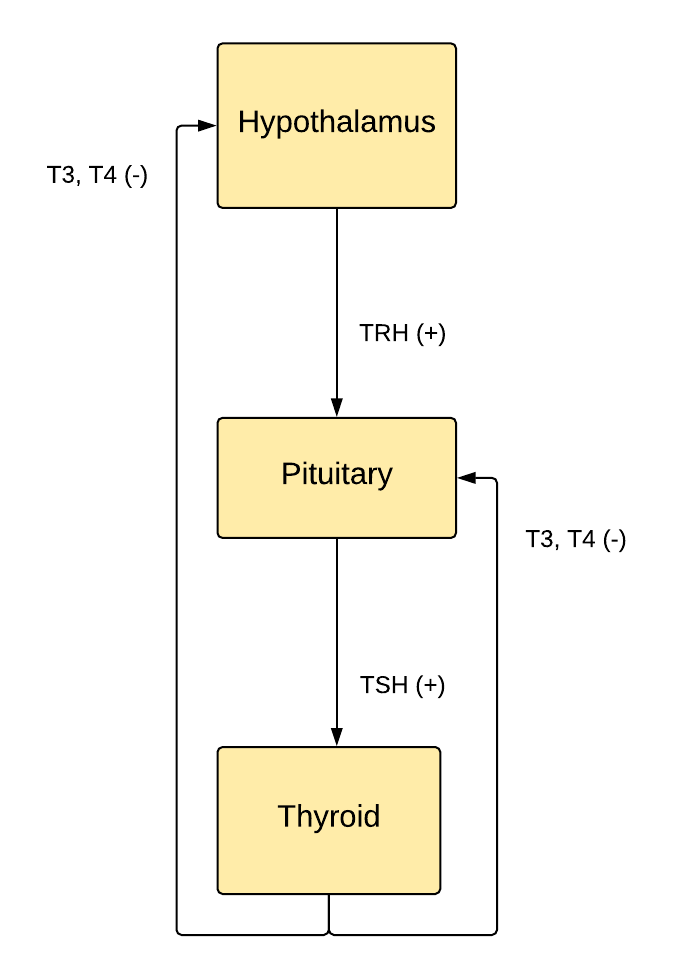
Understanding the causes of thyroid disease requires knowledge of the normal physiology of the hypothalamic-pituitary-thyroid axis. Thyrotropin-releasing hormone (TRH) from the hypothalamus stimulates the pituitary gland to release TSH, which in turn prompts the thyroid gland to produce T3 and T4. Approximately 10% to 15% of thyroid hormone released is T3, whereas 85% to 90% is T4. In circulation, approximately 99.5% of both thyroid hormones are protein-bound to thyroxine-binding globulin (TBG), albumin, and transthyretin (pre-albumin). Thyroid hormones enter cells through specific membrane transport proteins, which also have a role in the release of these hormones from the thyroid gland (see Image. Hypothalamic-Pituitary-Thyroid Axis).
Deiodinases (DIOs) are crucial in regulating thyroid hormones. Type 1 DIO facilitates the peripheral conversion of T4 into its active form, T3. Type 2 DIO converts T4 to T3 within the hypothalamus and pituitary, contributing to feedback regulation. In contrast, type 3 DIO inactivates thyroid hormones by converting T4 to reverse T3 (rT3) and T3 to 3,5-diiodo-L-thyronine (T2), both of which are inactive forms. T3 is the biologically active form, which exerts its effects by binding to thyroid hormone receptors on target tissues.[3]
Thyroid disorders can result from primary thyroid gland disease or secondary hypothalamic-pituitary dysfunction. These disorders may result in either the overproduction of thyroid hormones (hyperthyroidism) or the underproduction of thyroid hormones (hypothyroidism). Additionally, TSH resistance due to genetic mutations of the TSH receptor on the thyroid gland, drug-induced thyroid disease, pregnancy-related thyroid disorders, and nonthyroidal illness (which can occur after acute or chronic diseases, including acute psychiatric disorders) may also contribute to thyroid dysfunction (see Table below).[4] Please see StatPearls' companion resource, "Thyroid Disease and Pregnancy," for more information.
Table. Etiology of Thyroid Disorders
| Thyroid Disorders | Etiologies |
| Thyroiditis [5] |
|
| Autoimmune thyroid disease [6] |
|
| Hypothyroidism [7] |
Primary
Secondary Hypothalamic or pituitary disease: Invasive tumor or lesion, intracranial surgery, drugs, irradiation, head injury, intracranial infarction, lymphocytic hypophysitis, infiltration by sarcoid/hemochromatosis/histiocytosis X, tuberculosis, syphilis, mycoses, genetic (TSH beta and receptor mutations), and idiopathic. |
| Hyperthyroidism [8] |
Primary
Secondary
Other Causes
|
| Thyroid cancer [9] |
Multifactorial, which includes risks related to many factors such as sex, age, previous radiation exposure, and personal or family history. |
Specimen Requirements and Procedure
The preferred sample for measuring free T3 (FT3) and free T4 (FT4) levels is serum, which should be separated from the clot promptly after collection. Whole blood samples remain stable at 15 ºC to 35 ºC for up to 24 hours. Serum samples are stable at room temperature for 1 week, but refrigeration at 4 ºC is recommended if analysis is delayed beyond 24 hours. If not analyzed within 6 days, serum samples should be frozen at -20 ºC or below. Repeated freeze-thaw cycles and vortexing, which can denature proteins, should be avoided.[10][11] TSH analysis can be performed on either serum or plasma samples. While plasma samples may show clinically insignificant higher values, no significant difference has been noted between samples collected using serum-separator gel tubes and those collected without gel.[12]
TSH exhibits diurnal variation, with peak values occurring at night, typically around midnight, and the lowest levels in the morning. This circadian rhythm is disrupted during illness, and in central hypothyroidism, the usual increase in TSH levels at night is absent. Sleep deprivation is associated with higher TSH levels.[13][14] Although TSH secretion remains relatively constant throughout the day, reference intervals are based on levels measured during these times. However, valuable clinical information can still be obtained from blood specimens drawn from ambulatory patients at various times throughout the day.[15] Age and gender generally do not significantly affect reference intervals, except at the extremes of age.[16]
Diagnostic Tests
Biochemical analysis is essential for diagnosing and managing thyroid dysfunction. The primary diagnostic tests for assessing thyroid function include TSH, T3, and T4. Additional tests that aid in evaluating thyroid disease include thyroglobulin (Tg), thyroglobulin antibody (Tg-Ab), anti-thyroid peroxidase antibody (aTPOAb), thyrotropin receptor antibody (TRAb), and calcitonin.[17]
TSH analysis, performed using third-generation immunoassays, is highly sensitive and is the recommended screening test for suspected thyroid disease. Elevated TSH levels suggest hypothyroidism, whereas low levels indicate hyperthyroidism. However, relying solely on TSH analysis in cases of subclinical thyroid disease, hospitalized patients with secondary hypothyroidism, or during the initial phases of medical therapy for hyper- or hypothyroidism can be misleading; concomitant analysis of FT3 and FT4 is essential.[17]
FT3 and FT4 levels are more commonly measured than total hormone levels because they are not affected by altered TBG levels, which can occur during pregnancy, acute illness, or with the use of medications such as estrogen, tamoxifen, opiates, androgens, and glucocorticoids. However, the picomolar concentration of these hormones in the blood makes their measurement more challenging.[17]
Testing Procedures
TSH is primarily measured using chemiluminescent immunoassay, where chemiluminescent-labeled antibodies bind to TSH in the patient's blood, which is then captured by a fixed solid-phase antibody. This method has led to the development of third-generation TSH assays, which have a coefficient of variation of less than 20% at values as low as mIU/L. In contrast, first-generation assays could only distinguish between normal and hypothyroid patients, while second-generation assays could detect TSH levels below the reference interval but were still unable to differentiate primary hyperthyroidism from other causes of low TSH.[18]
FT4 and FT3 measurements can be either direct or indirect. Direct methods involve physically separating free hormone from bound hormone, using techniques such as equilibrium dialysis or ultrafiltration. Indirect methods estimate free hormone levels using hormone-binding proteins. Immunoassays, which are indirect measurements of free hormones, are widely used and can be categorized as 1-step or 2-step, depending on whether a wash step is performed before tracer insertion.[19]
Total T3 and T4 are present in nanomolar amounts in the blood, making them easier to detect and useful for confirming free hormone results. Mass spectrometry is the gold standard and recommended method for measurement; however, competitive immunoassays using displacing agents to release T3 or T4 from serum binding sites are still widely used. These immunoassays show poor correlation among themselves and when compared to mass spectrometry.[20][21][22]
Interfering Factors
Immunoassays can be affected by anti-animal (heterophile) antibodies in the patient's sample.[23][24] These antibodies can bind to the assay antibodies or even to the analyte of interest, resulting in either false-negative or false-positive results. TSH, FT4, FT3, and Tg are all susceptible to this type of interference.[25][26][27]
FT4 and FT3 levels can also be influenced by disturbances in binding proteins during analysis. This includes inherited or acquired alterations in binding proteins, such as familial dysalbuminemic hyperthyroxinemia (FDH), which results in an altered free-to-total hormone ratio.[28] Additionally, substances such as certain drugs, non-esterified fatty acids, and heparin can cause hormones to dissociate from their binding proteins.[29][30] Autoantibodies may also interfere with both FT3 and FT4 and total T3 and T4 measurements.[31] Although the use of grossly hemolyzed or lipemic samples is not recommended, moderate hemolysis or lipemia generally does not interfere with most assay methods.[32]
Results, Reporting, and Critical Findings
The diagnosis of hypo- or hyperthyroidism, or normal thyroid function, is typically straightforward with TSH and FT4 results alone.[33] However, occasionally, results may be atypical or inconsistent with the clinical presentation due to factors such as medications, physiological changes (eg, pregnancy), rare acquired or genetic hypothalamic-pituitary disorders (such as TSHomas and thyroid hormone resistance [THR]), or concurrent illnesses.[3][34]
TSH responds to changes in FT4 levels in a log-linear manner; therefore, serial monitoring of both should take this relationship into account.[35] Changes in TSH levels may occur before FT4 in primary thyroid disorders, leading to a subclinical presentation where FT4 remains normal but TSH is abnormal. TSH is adequate in most patients to rule out primary thyroid disease if the results are normal. However, it may not be accurate in cases where the patient has recently been treated for thyrotoxicosis, is on medications that transiently decrease TSH levels (eg, glucocorticoids), has non-thyroidal illness, secondary hypothyroidism, THR, a TSHoma, or disorders in the transport or metabolism of thyroid hormone.[36]
Additional Tests
FT3 analysis may be necessary if TSH is low with a normal FT4 to diagnose T3 toxicosis, or for patients on T4 treatment who show high FT4 levels but a normal TSH. If results suggest subclinical hypothyroidism, an antithyroid antibody analysis should be performed for prognosis. In cases where Graves disease is suspected, TRAb testing is highly sensitive and specific for confirming the diagnosis.[3][37]
Discordant Thyroid-Stimulating Hormone and Free Thyroxine
High FT4 with a normal TSH can occur in patients on T4 treatment due to decreased deiodination. This pattern may also be seen in conditions such as FDH, THR, and TSHoma.[3][37]
Critical Results
Thyroid emergencies, such as thyrotoxic storm and myxedema coma, require immediate attention. Laboratory findings alone cannot confirm these conditions; they must be interpreted in conjunction with the clinical presentation to establish a diagnosis.[38]
A diagnosis of myxedema coma can be made using a diagnostic scoring system, which considers the patient's body temperature, central nervous system effects, gastrointestinal symptoms, presence or absence of a precipitating event, cardiovascular dysfunction, and metabolic disturbances. This system is used in conjunction with laboratory findings, including high, normal, or low TSH levels and low or undetectable FT4 levels.[39]
A thyrotoxic storm results in markedly elevated T4 and T3 levels with low TSH. The diagnosis can be made using a diagnostic scoring system, which considers the patient's body temperature, central nervous system effects, gastrointestinal symptoms, and cardiovascular dysfunction. Please see StatPearls' companion resource, "Thyroid Storm," for more information.
Clinical Significance
Thyroid disorders present with symptoms and signs that are often nonspecific, making biochemical confirmation essential. Although TSH is commonly used for screening thyroid disease, it is important to recognize its limitations in cases of central hypothyroidism, subclinical thyroid disease, hospitalized patients, treatment monitoring, and when certain medications are being used.[17]
Subclinical Thyroid Disease
Asymptomatic individuals with normal thyroid hormone levels but low or high TSH levels are classified as having subclinical hyperthyroidism or subclinical hypothyroidism, respectively. Treatment should be considered for subclinical hyperthyroidism in geriatric patients or those with atrial fibrillation or osteoporosis. Subclinical hypothyroidism may require treatment if the TSH is greater than 10 mIU/L, if aTPOAb levels are elevated, or if there are certain comorbidities, such as hypercholesterolemia or infertility.[17]
Hyperthyroidism
Hyperthyroidism is most commonly caused by the overproduction of thyroid hormone in the thyroid gland, and it is biochemically characterized by elevated T4 and/or T3 levels with a suppressed TSH. A positive TRAb confirms Graves disease as the underlying cause. In cases of pituitary tumor-related hyperthyroidism, TSH levels are typically elevated, along with increased sex hormone–binding globulin (released peripherally in response to thyroid hormone), alpha-subunit, and an elevated alpha-subunit-to-TSH ratio.
High or inappropriately normal TSH levels may also indicate resistance to thyroid hormone, which is an autosomal dominant disorder characterized by a reduced FT4:FT3 ratio. TRH stimulation in these cases typically causes a normal or exaggerated response. Additionally, conditions such as hydatidiform mole or embryonal carcinoma can lead to increased thyroid hormone production due to elevated hCG levels. As a result, patients with these conditions will present with low TSH, high T4 and T3, and elevated hCG levels. Patients with T3 toxicosis typically present with elevated T3, normal T4, and low TSH levels. In contrast, hyperthyroidism caused by oral T3 administration is characterized by low T4 levels. Treatment of hyperthyroidism should be monitored using both TSH and FT4, as TSH levels may take several months to normalize, even with adequate treatment.[17][40]
Hypothyroidism
Hypothyroidism is primarily caused by thyroid gland hypofunction, which is reflected in biochemical results as low T3 and T4 levels with elevated TSH. In contrast, pituitary (central) hypothyroidism presents with normal or low TSH levels in association with low T3 and T4 levels.[40]
Other Considerations
- Elevated T4 and T3 levels may occur in patients with increased TBG levels, whereas isolated increased T4 levels are observed in patients with FDH and familial elevation of thyroid-binding prealbumin.[41]
- Hyperestrogenic conditions (eg, pregnancy, estrogen-containing drugs, and hydatidiform mole), liver disease, lymphocytoma, acute intermittent porphyria, drugs (such as heroin, clofibrate, methadone, and 5-fluorouracil), and acute psychosis are associated with increased TBG, leading to elevated total T3 and T4 levels. Conversely, patients with low TBG—whether congenital or drug-induced (e.g., androgens)—exhibit decreased total T4 and T3 levels with normal TSH.[42]
- Euthyroid hyperthyroxinemia syndrome is characterized by elevated T4 and T3 levels with a normal or mildly increased TSH, often due to peripheral THR.[43]
- In severely ill patients, nonthyroidal illness syndrome typically presents with low T3 and variable T4 levels, which may occasionally be elevated. Progression to true thyrotoxicosis is extremely rare. Low T4 and T3 levels often indicate a poor prognosis in these patients, resulting from decreased T4 and TBG production. Dopamine administration can inhibit the TSH response to thyroid-releasing hormone, while furosemide inhibits T4 binding. If these patients survive, TSH levels typically increase during recovery. Please see StatPearls' companion resource, "Euthyroid Sick Syndrome," for more information.
- Acute myocardial infarction is also associated with biochemical changes in T3 and T4 levels, with low T3 levels peaking 3 days post-infarct and high T4 levels peaking 6 to 7 days post-infarct.[44]
- The impact of drugs on thyroid function extends beyond merely altering TBG levels. For example, propranolol blocks the conversion of T4 to T3, resulting in elevated T4 and reduced T3 levels. Amiodarone and iodine-containing contrast agents, such as ipodate and iopanoate, also impair the peripheral conversion of T4 to T3 while exerting additional, less-understood effects on the thyroid gland itself. Medications such as heparin, salicylates, diazepam, fenclofenac, and phenylbutazone displace T4 from binding proteins, leading to decreased total T4 levels, with low total T3 levels also observed in patients taking salicylates or fenclofenac.[45][46]
- Diphenylhydantoin displaces T4 from binding proteins and also enhances T4 clearance by inducing liver enzymes.[47] Sulfonylureas and sulfonamides interfere with thyroid hormone synthesis (organification), and sulfonylureas further displace T4 from TBG, potentially leading to hypothyroidism.[48] Drugs such as carbamazepine and phenobarbital increase T4 clearance through enzyme induction.[49] Sodium nitroprusside inhibits thyroid function, reducing T4 levels. Heavy cigarette smoking mildly lowers total T4 and T3 levels and FT4, while phenothiazine use is associated with reduced total T4.
- Patients with impaired kidney function may have low T3 and T4 levels with normal or high TSH.[50]
- Prolactin-secreting tumors can lead to TSH suppression, resulting in low T4 and inappropriately normal TSH levels, likely due to increased dopamine production. Please see StatPearls' companion resource, "Pituitary Adenoma," for more information.
- Low total T4 and T3 levels and FT3 with high TSH levels may be seen in pseudohypoparathyroidism type 1. Please see StatPearls' companion resource, "Pseudohypoparathyroidism," for more information.
- Low T3 levels, resulting from decreased deiodination of T4, can occur in conditions such as protein-energy malnutrition, poorly controlled diabetes mellitus, use of certain drugs (eg, propylthiouracil, glucocorticoids, iopanoate, sodium ipodate, amiodarone, and colistipal), and in severely ill patients. T3 levels may also be low in hyperparathyroidism and with the use of drugs such as propylthiouracil.[51]
Quality Control and Lab Safety
A quality management system (QMS) is fundamental to ensuring reliable TFT, enabling accurate and consistent measurement of TSH, FT4, and T3. At the core of the QMS is quality control (QC), a vital process that minimizes diagnostic errors and supports informed clinical decisions. This systematic approach integrates policies, procedures, and resources to uphold high standards across all laboratory operations.[52]
QC in thyroid testing comprises 2 key components—internal and external. Internal QC involves analyzing control samples with known values alongside patient samples, with results monitored using Levey-Jennings charts. To identify deviations, laboratories apply Westgard rules, which differentiate between random errors (unpredictable variations, such as pipetting inaccuracies) and systematic errors (consistent biases, such as reagent degradation or instrument drift). These tools offer a clear framework for tracking assay performance over time.[53]
Random errors necessitate immediate corrective actions, such as recalibrating equipment, retraining staff, or reviewing procedural consistency, while preventive measures like automation help minimize human error. In contrast, systematic errors may require reagent replacement, instrument recalibration, or scheduled maintenance to address underlying causes. These proactive measures ensure test results remain reliable and reproducible.[54]
External QC, also known as proficiency testing, provides an additional layer of validation by requiring laboratories to analyze blind samples from an independent organization. Comparing results with peer laboratories helps identify biases or systemic issues that may not be detected through internal QC alone. This external benchmarking enhances confidence in the laboratory’s performance and ensures alignment with industry standards.[55]
Beyond formal QC measures, laboratories monitor patient data trends as an extra layer of safeguard. For example, unexpected shifts in TSH distributions among patients could indicate assay issues or instrument malfunctions. This real-world data, alongside control sample analysis, provides a broader perspective on testing reliability and helps identify subtle issues early.[56] To maintain regulatory compliance and prepare for audits, laboratories meticulously document all QC activities, including control sample outcomes, corrective actions, and instrument maintenance logs. Regular reagent validation and routine equipment servicing further enhance test reliability. Detailed record-keeping ensures traceability and supports continuous improvement in laboratory processes.[57]
Advanced technologies, such as automated analyzers with built-in QC checks, improve efficiency by detecting deviations in real time. However, skilled oversight remains essential. Ongoing training for laboratory personnel reinforces protocol adherence, minimizes errors, and fosters a culture of quality and accountability within the QMS framework.[58]
Safety in laboratories conducting TFTs is crucial due to the risks associated with handling blood samples, reagents, and potentially hazardous equipment. Staff must wear personal protective equipment, such as gloves, lab coats, and face shields, when necessary to protect against biohazards like bloodborne pathogens, including HIV and hepatitis, present in patient samples. Proper venipuncture techniques are essential, with needles disposed of in sharps containers to prevent needlestick injuries. Samples should be accurately labeled and stored at appropriate temperatures, such as being refrigerated overnight if testing is delayed, to maintain their integrity.[59]
Reagents used for assays such as TSH, FT4, or antibody tests must be handled carefully according to manufacturer instructions, securely stored, and, if volatile, used in fume hoods. Analyzers require regular maintenance, calibration, and operation by trained personnel to prevent malfunctions or electrical hazards. Biohazardous waste (eg, blood tubes) and chemical waste must be properly segregated and disposed of in accordance with local regulations to prevent contamination. Laboratories should also maintain spill kits, eyewash stations, and first-aid supplies, ensuring staff are trained to manage exposures or accidents effectively.[60]
Enhancing Healthcare Team Outcomes
Biochemical TFTs are essential for diagnosing and monitoring thyroid disease, which is highly prevalent and often presents with nonspecific symptoms. Thyroid disorders can arise from primary thyroid gland dysfunction or secondary hypothalamic-pituitary disease, leading to either excessive thyroid hormone production (hyperthyroidism) or insufficient production (hypothyroidism). Additional causes include TSH resistance due to genetic aberrations of the TSH receptor, drug-induced thyroid dysfunction, pregnancy-related thyroid disorders, and nonthyroidal illness following acute or chronic conditions, including psychiatric disorders. While TFTs primarily measure TSH, T3, and T4 levels, further testing is often required to determine the underlying etiology.
Given the wide range of potential causes for thyroid disease, accurate diagnosis and management require clinical evaluation by general practitioners, internal medicine specialists, endocrinologists, and obstetricians. Additionally, due to the unusual results that may arise in critically ill patients, intensivists and cardiologists must also be aware of the limitations of TFTs in these cases. Many medications can affect thyroid hormones, making it crucial for pharmacists to understand their role in thyroid disease. The final diagnosis often depends on imaging studies, which is why it is also an important area of focus for radiologists and nuclear medicine physicians.
Effective communication among healthcare team members is crucial. Physicians and nurses must quickly recognize the symptoms of thyroid disease, while laboratory staff should ensure proper collection, storage, and testing procedures. Both internal and external QC measures are essential to guarantee accurate results. Open, clear communication promotes reliable laboratory outcomes, prevents errors, and ensures a coordinated and efficient response.
Media
References
Brochmann H, Bjøro T, Gaarder PI, Hanson F, Frey HM. Prevalence of thyroid dysfunction in elderly subjects. A randomized study in a Norwegian rural community (Naerøy). Acta endocrinologica. 1988 Jan:117(1):7-12 [PubMed PMID: 3381628]
Level 1 (high-level) evidenceFeingold KR, Ahmed SF, Anawalt B, Blackman MR, Boyce A, Chrousos G, Corpas E, de Herder WW, Dhatariya K, Dungan K, Hofland J, Kalra S, Kaltsas G, Kapoor N, Koch C, Kopp P, Korbonits M, Kovacs CS, Kuohung W, Laferrère B, Levy M, McGee EA, McLachlan R, Muzumdar R, Purnell J, Rey R, Sahay R, Shah AS, Singer F, Sperling MA, Stratakis CA, Trence DL, Wilson DP, Spencer CA. Assay of Thyroid Hormones and Related Substances. Endotext. 2000:(): [PubMed PMID: 25905337]
Koulouri O, Moran C, Halsall D, Chatterjee K, Gurnell M. Pitfalls in the measurement and interpretation of thyroid function tests. Best practice & research. Clinical endocrinology & metabolism. 2013 Dec:27(6):745-62. doi: 10.1016/j.beem.2013.10.003. Epub 2013 Oct 17 [PubMed PMID: 24275187]
Rivas AM, Lado-Abeal J. Thyroid hormone resistance and its management. Proceedings (Baylor University. Medical Center). 2016 Apr:29(2):209-11 [PubMed PMID: 27034574]
Pearce EN, Farwell AP, Braverman LE. Thyroiditis. The New England journal of medicine. 2003 Jun 26:348(26):2646-55 [PubMed PMID: 12826640]
Antonelli A, Ferrari SM, Corrado A, Di Domenicantonio A, Fallahi P. Autoimmune thyroid disorders. Autoimmunity reviews. 2015 Feb:14(2):174-80. doi: 10.1016/j.autrev.2014.10.016. Epub 2014 Oct 25 [PubMed PMID: 25461470]
Roberts CG, Ladenson PW. Hypothyroidism. Lancet (London, England). 2004 Mar 6:363(9411):793-803 [PubMed PMID: 15016491]
Franklyn JA, Boelaert K. Thyrotoxicosis. Lancet (London, England). 2012 Mar 24:379(9821):1155-66. doi: 10.1016/S0140-6736(11)60782-4. Epub 2012 Mar 5 [PubMed PMID: 22394559]
Haugen BR, Alexander EK, Bible KC, Doherty GM, Mandel SJ, Nikiforov YE, Pacini F, Randolph GW, Sawka AM, Schlumberger M, Schuff KG, Sherman SI, Sosa JA, Steward DL, Tuttle RM, Wartofsky L. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid : official journal of the American Thyroid Association. 2016 Jan:26(1):1-133. doi: 10.1089/thy.2015.0020. Epub [PubMed PMID: 26462967]
Tanner M, Kent N, Smith B, Fletcher S, Lewer M. Stability of common biochemical analytes in serum gel tubes subjected to various storage temperatures and times pre-centrifugation. Annals of clinical biochemistry. 2008 Jul:45(Pt 4):375-9. doi: 10.1258/acb.2007.007183. Epub [PubMed PMID: 18583622]
Mardell R, Gamlen TR. Discrepant results for free thyroxin by radioimmunoassay and dialysis procedures explained. Clinical chemistry. 1982 Sep:28(9):1989 [PubMed PMID: 7127824]
Ercan M, Fırat Oğuz E, Akbulut ED, Yilmaz M, Turhan T. Comparison of the effect of gel used in two different serum separator tubes for thyroid function tests. Journal of clinical laboratory analysis. 2018 Jul:32(6):e22427. doi: 10.1002/jcla.22427. Epub 2018 Mar 7 [PubMed PMID: 29512190]
Keffer JH. Preanalytical considerations in testing thyroid function. Clinical chemistry. 1996 Jan:42(1):125-34 [PubMed PMID: 8565214]
Philippe J, Dibner C. Thyroid circadian timing: roles in physiology and thyroid malignancies. Journal of biological rhythms. 2015 Apr:30(2):76-83. doi: 10.1177/0748730414557634. Epub 2014 Nov 19 [PubMed PMID: 25411240]
Soldin OP, Chung SH, Colie C. The Use of TSH in Determining Thyroid Disease: How Does It Impact the Practice of Medicine in Pregnancy? Journal of thyroid research. 2013:2013():148157. doi: 10.1155/2013/148157. Epub 2013 May 9 [PubMed PMID: 23762775]
Fisher DA. Physiological variations in thyroid hormones: physiological and pathophysiological considerations. Clinical chemistry. 1996 Jan:42(1):135-9 [PubMed PMID: 8565215]
Soh SB, Aw TC. Laboratory Testing in Thyroid Conditions - Pitfalls and Clinical Utility. Annals of laboratory medicine. 2019 Jan:39(1):3-14. doi: 10.3343/alm.2019.39.1.3. Epub [PubMed PMID: 30215224]
Spencer CA, Schwarzbein D, Guttler RB, LoPresti JS, Nicoloff JT. Thyrotropin (TSH)-releasing hormone stimulation test responses employing third and fourth generation TSH assays. The Journal of clinical endocrinology and metabolism. 1993 Feb:76(2):494-8 [PubMed PMID: 8432796]
Van Houcke SK, Van Uytfanghe K, Shimizu E, Tani W, Umemoto M, Thienpont LM. IFCC international conventional reference procedure for the measurement of free thyroxine in serum: International Federation of Clinical Chemistry and Laboratory Medicine (IFCC) Working Group for Standardization of Thyroid Function Tests (WG-STFT)(1). Clinical chemistry and laboratory medicine. 2011 Aug:49(8):1275-1281. doi: 10.1515/CCLM.2011.639. Epub 2011 Jun 16 [PubMed PMID: 21675941]
Soukhova N, Soldin OP, Soldin SJ. Isotope dilution tandem mass spectrometric method for T4/T3. Clinica chimica acta; international journal of clinical chemistry. 2004 May:343(1-2):185-90 [PubMed PMID: 15115693]
Thienpont LM, Van Uytfanghe K, Marriott J, Stokes P, Siekmann L, Kessler A, Bunk D, Tai S. Feasibility study of the use of frozen human sera in split-sample comparison of immunoassays with candidate reference measurement procedures for total thyroxine and total triiodothyronine measurements. Clinical chemistry. 2005 Dec:51(12):2303-11 [PubMed PMID: 16223886]
Level 2 (mid-level) evidenceThienpont LM, Van Uytfanghe K, Beastall G, Faix JD, Ieiri T, Miller WG, Nelson JC, Ronin C, Ross HA, Thijssen JH, Toussaint B, IFCC Working Group on Standardization of Thyroid Function Tests. Report of the IFCC Working Group for Standardization of Thyroid Function Tests; part 3: total thyroxine and total triiodothyronine. Clinical chemistry. 2010 Jun:56(6):921-9. doi: 10.1373/clinchem.2009.140228. Epub 2010 Apr 15 [PubMed PMID: 20395622]
Ross HA, Menheere PP, Endocrinology Section of SKML (Dutch Foundation for Quality Assessment in Clinical Laboratories), Thomas CM, Mudde AH, Kouwenberg M, Wolffenbuttel BH. Interference from heterophilic antibodies in seven current TSH assays. Annals of clinical biochemistry. 2008 Nov:45(Pt 6):616. doi: 10.1258/acb.2008.008066. Epub 2008 Sep 9 [PubMed PMID: 18782812]
Level 2 (mid-level) evidenceZouwail SA, O'Toole AM, Clark PM, Begley JP. Influence of thyroid hormone autoantibodies on 7 thyroid hormone assays. Clinical chemistry. 2008 May:54(5):927-8. doi: 10.1373/clinchem.2007.099770. Epub [PubMed PMID: 18443182]
Sapin R, Gasser F, Schlienger JL. Familial dysalbuminemic hyperthyroxinemia and thyroid hormone autoantibodies: interference in current free thyroid hormone assays. Hormone research. 1996:45(3-5):139-41 [PubMed PMID: 8964572]
Ohba K, Noh JY, Unno T, Satoh T, Iwahara K, Matsushita A, Sasaki S, Oki Y, Nakamura H. Falsely elevated thyroid hormone levels caused by anti-ruthenium interference in the Elecsys assay resembling the syndrome of inappropriate secretion of thyrotropin. Endocrine journal. 2012:59(8):663-7 [PubMed PMID: 22673200]
Loh TP, Kao SL, Halsall DJ, Toh SA, Chan E, Ho SC, Tai ES, Khoo CM. Macro-thyrotropin: a case report and review of literature. The Journal of clinical endocrinology and metabolism. 2012 Jun:97(6):1823-8. doi: 10.1210/jc.2011-3490. Epub 2012 Mar 30 [PubMed PMID: 22466337]
Level 3 (low-level) evidenceCartwright D, O'Shea P, Rajanayagam O, Agostini M, Barker P, Moran C, Macchia E, Pinchera A, John R, Agha A, Ross HA, Chatterjee VK, Halsall DJ. Familial dysalbuminemic hyperthyroxinemia: a persistent diagnostic challenge. Clinical chemistry. 2009 May:55(5):1044-6. doi: 10.1373/clinchem.2008.120303. Epub 2009 Mar 12 [PubMed PMID: 19282355]
Stockigt JR, Lim CF. Medications that distort in vitro tests of thyroid function, with particular reference to estimates of serum free thyroxine. Best practice & research. Clinical endocrinology & metabolism. 2009 Dec:23(6):753-67. doi: 10.1016/j.beem.2009.06.004. Epub [PubMed PMID: 19942151]
Stevenson HP, Archbold GP, Johnston P, Young IS, Sheridan B. Misleading serum free thyroxine results during low molecular weight heparin treatment. Clinical chemistry. 1998 May:44(5):1002-7 [PubMed PMID: 9590373]
Després N, Grant AM. Antibody interference in thyroid assays: a potential for clinical misinformation. Clinical chemistry. 1998 Mar:44(3):440-54 [PubMed PMID: 9510847]
Krasowski MD. Educational Case: Hemolysis and Lipemia Interference With Laboratory Testing. Academic pathology. 2019 Jan-Dec:6():2374289519888754. doi: 10.1177/2374289519888754. Epub 2019 Nov 22 [PubMed PMID: 31803827]
Level 3 (low-level) evidenceClark PM, Holder RL, Haque SM, Hobbs FD, Roberts LM, Franklyn JA. The relationship between serum TSH and free T4 in older people. Journal of clinical pathology. 2012 May:65(5):463-5. doi: 10.1136/jclinpath-2011-200433. Epub 2012 Jan 28 [PubMed PMID: 22287691]
Koulouri O, Gurnell M. How to interpret thyroid function tests. Clinical medicine (London, England). 2013 Jun:13(3):282-6. doi: 10.7861/clinmedicine.13-3-282. Epub [PubMed PMID: 23760704]
Hoermann R, Eckl W, Hoermann C, Larisch R. Complex relationship between free thyroxine and TSH in the regulation of thyroid function. European journal of endocrinology. 2010 Jun:162(6):1123-9. doi: 10.1530/EJE-10-0106. Epub 2010 Mar 18 [PubMed PMID: 20299491]
Surks MI, Ortiz E, Daniels GH, Sawin CT, Col NF, Cobin RH, Franklyn JA, Hershman JM, Burman KD, Denke MA, Gorman C, Cooper RS, Weissman NJ. Subclinical thyroid disease: scientific review and guidelines for diagnosis and management. JAMA. 2004 Jan 14:291(2):228-38 [PubMed PMID: 14722150]
Gurnell M, Halsall DJ, Chatterjee VK. What should be done when thyroid function tests do not make sense? Clinical endocrinology. 2011 Jun:74(6):673-8. doi: 10.1111/j.1365-2265.2011.04023.x. Epub [PubMed PMID: 21521292]
Ylli D, Klubo-Gwiezdzinska J, Wartofsky L. Thyroid emergencies. Polish archives of internal medicine. 2019 Aug 29:129(7-8):526-534. doi: 10.20452/pamw.14876. Epub 2019 Jun 25 [PubMed PMID: 31237256]
Popoveniuc G, Chandra T, Sud A, Sharma M, Blackman MR, Burman KD, Mete M, Desale S, Wartofsky L. A diagnostic scoring system for myxedema coma. Endocrine practice : official journal of the American College of Endocrinology and the American Association of Clinical Endocrinologists. 2014 Aug:20(8):808-17. doi: 10.4158/EP13460.OR. Epub [PubMed PMID: 24518183]
Walker HK, Hall WD, Hurst JW, Dunlap DB. Thyroid Function Tests. Clinical Methods: The History, Physical, and Laboratory Examinations. 1990:(): [PubMed PMID: 21250093]
Ting MJM, Zhang R, Lim EM, Ward BK, Wilson SG, Walsh JP. Familial Dysalbuminemic Hyperthyroxinemia as a Cause for Discordant Thyroid Function Tests. Journal of the Endocrine Society. 2021 Apr 1:5(4):bvab012. doi: 10.1210/jendso/bvab012. Epub 2021 Feb 1 [PubMed PMID: 33728390]
Díaz S, Cárdenas H, Brandeis A, Miranda P, Salvatierra AM, Croxatto HB. Relative contributions of anovulation and luteal phase defect to the reduced pregnancy rate of breastfeeding women. Fertility and sterility. 1992 Sep:58(3):498-503 [PubMed PMID: 1521642]
Olateju TO, Vanderpump MP. Thyroid hormone resistance. Annals of clinical biochemistry. 2006 Nov:43(Pt 6):431-40 [PubMed PMID: 17132274]
Rasool R, Unar A, Jafar TH, Chanihoon GQ, Mubeen B. A Role of Thyroid Hormones in Acute Myocardial Infarction: An Update. Current cardiology reviews. 2023:19(1):e280422204209. doi: 10.2174/1573403X18666220428121431. Epub [PubMed PMID: 35657286]
Nademanee K, Piwonka RW, Singh BN, Hershman JM. Amiodarone and thyroid function. Progress in cardiovascular diseases. 1989 May-Jun:31(6):427-37 [PubMed PMID: 2652189]
Wiersinga WM. Propranolol and thyroid hormone metabolism. Thyroid : official journal of the American Thyroid Association. 1991 Summer:1(3):273-7 [PubMed PMID: 1688102]
Heyma P, Larkins RG, Perry-Keene D, Peter CT, Ross D, Sloman JG. Thyroid hormone levels and protein binding in patients on long-term diphenylhydantoin treatment. Clinical endocrinology. 1977 May:6(5):369-76 [PubMed PMID: 872445]
Hershman JM, Craane TJ, Colwell JA. Effect of sulfonylurea drugs on the binding of triiodothyronine and thyroxine to thyroxine-binding globulin. The Journal of clinical endocrinology and metabolism. 1968 Nov:28(11):1605-10 [PubMed PMID: 4972270]
Rootwelt K, Ganes T, Johannessen SI. Effect of carbamazepine, phenytoin and phenobarbitone on serum levels of thyroid hormones and thyrotropin in humans. Scandinavian journal of clinical and laboratory investigation. 1978 Dec:38(8):731-6 [PubMed PMID: 105401]
Mohamedali M, Reddy Maddika S, Vyas A, Iyer V, Cheriyath P. Thyroid disorders and chronic kidney disease. International journal of nephrology. 2014:2014():520281. doi: 10.1155/2014/520281. Epub 2014 Apr 13 [PubMed PMID: 24829799]
Heinen E, Herrmann J, Mosny D, Moreno F, Teschke R, Krüskemper HL. Inhibition of peripheral deiodination of 3, 5, 3'-triiodothyronine: an adverse effect of propylthiouracil in the treatment of T3-thyrotoxicosis. Journal of endocrinological investigation. 1981 Jul-Sep:4(3):331-4 [PubMed PMID: 7320437]
Zhang S, Wang W, Zhao H, He F, Zhong K, Yuan S, Wang Z. Status of internal quality control for thyroid hormones immunoassays from 2011 to 2016 in China. Journal of clinical laboratory analysis. 2018 Jan:32(1):. doi: 10.1002/jcla.22154. Epub 2017 Feb 15 [PubMed PMID: 28205257]
Level 2 (mid-level) evidenceHu LT, Wang ZG. Internal quality control practice of thyroid disease related tests and imprecision analysis in China. Clinical laboratory. 2014:60(2):301-8 [PubMed PMID: 24660545]
Level 2 (mid-level) evidenceKinns H, Pitkin S, Housley D, Freedman DB. Internal quality control: best practice. Journal of clinical pathology. 2013 Dec:66(12):1027-32. doi: 10.1136/jclinpath-2013-201661. Epub 2013 Sep 26 [PubMed PMID: 24072731]
Level 2 (mid-level) evidenceJames D, Ames D, Lopez B, Still R, Simpson W, Twomey P. External quality assessment: best practice. Journal of clinical pathology. 2014 Aug:67(8):651-5. doi: 10.1136/jclinpath-2013-201621. Epub 2014 Mar 12 [PubMed PMID: 24621574]
Level 2 (mid-level) evidenceZhang Y, Wang HL, Xie YH, He DH, Zhou CQ, Kong LR. Practical application of the patient data-based quality control method: the potassium example. Biochemia medica. 2024 Feb 15:34(1):010901. doi: 10.11613/BM.2024.010901. Epub [PubMed PMID: 38361737]
Level 2 (mid-level) evidenceValenstein PN, Stankovic AK, Souers RJ, Schneider F, Wagar EA. Document control practices in 120 clinical laboratories. Archives of pathology & laboratory medicine. 2009 Jun:133(6):942-9 [PubMed PMID: 19492888]
Wadhwa V, Rai S, Thukral T, Chopra M. Laboratory quality management system: road to accreditation and beyond. Indian journal of medical microbiology. 2012 Apr-Jun:30(2):131-40. doi: 10.4103/0255-0857.96647. Epub [PubMed PMID: 22664426]
Level 2 (mid-level) evidenceLunn G, Lawler Chemical Resistance Of Gloves G. Laboratory safety. Current protocols in protein science. 2002 Aug:Appendix 2():A.2A.1-A.2A.35. doi: 10.1002/0471140864.psa02as28. Epub [PubMed PMID: 18429218]
Tait FN, Mburu C, Gikunju J. Occupational safety and health status of medical laboratories in Kajiado County, Kenya. The Pan African medical journal. 2018:29():65. doi: 10.11604/pamj.2018.29.65.12578. Epub 2018 Jan 23 [PubMed PMID: 29875946]