Introduction
Aspergilloma and chronic cavitary pulmonary aspergillosis (CCPA) refer to the inert saprophytic colonization of preexisting cavitary spaces in the pulmonary parenchyma, along with their presentation and complications. These conditions fall under the broader category of chronic pulmonary aspergillosis (CPA), which includes several distinct clinical entities, such as aspergilloma, CCPA, chronic fibrosing pulmonary aspergillosis (CFPA), Aspergillus nodules, and subacute invasive aspergillosis.[1] Importantly, the overlap in clinical and radiographic features among CPA entities has led to confusion in interpreting the literature on their natural history and management. This confusion is further attributed to the differences in cumulative clinician experience in managing these conditions. Published case series from regions with high tuberculosis (TB) prevalence may present different diagnostic and management approaches compared to areas with lower prevalence, where aspergilloma is less common.
Management approaches remain controversial due to the existence of small case series in the literature, with a lack of extensive, prospective, randomized studies.[2] Diagnosing and managing these chronic fungal infections is complex and challenging, as radiographic features often overlap with those of other lung diseases.[3] Moreover, as CPA usually develops most often in diseased lungs, it is difficult to determine the extent of radiological distortion caused by the fungus versus the underlying lung disease.
Consensus Definitions of Chronic Pulmonary Aspergillosis
The European Society for Clinical Microbiology and Infectious Diseases, the European Respiratory Society, and the Infectious Diseases Society of America have established consensus definitions for CPA,[1][4] and their diagnostic criteria are mentioned below.
Simple Aspergilloma
A single pulmonary cavity containing a fungal ball, with serological or microbiological evidence of Aspergillus spp. in a non-immunocompromised patient. The patient experiences minor or no symptoms, and there is no radiological progression over at least 3 months of observation.
Chronic Cavitary Pulmonary Aspergillosis
One or more pulmonary cavities (with either thin or thick walls), possibly containing one or more aspergillomas or irregular intraluminal material, along with serological or microbiological evidence of Aspergillus spp. This condition is associated with significant pulmonary and/or systemic symptoms and overt radiological progression, such as new cavities, increasing pericavitary infiltrates, or increasing fibrosis, over at least 3 months of observation.
Chronic Fibrosing Pulmonary Aspergillosis
Severe fibrotic destruction of at least 2 lobes of the lung complicates CCPA, resulting in significant loss of lung function. When severe fibrotic destruction affects a single lobe with a cavity, it is referred to as CCPA affecting that lobe. The fibrosis typically presents as consolidation, but large cavities surrounded by fibrosis may also be observed.
Aspergillus Nodule
An unusual form of CPA, Aspergillus nodules may present as one or more nodules that may or may not cavitate. These nodules can mimic conditions such as tuberculoma, lung carcinoma, coccidioidomycosis, and other diagnoses. A definitive diagnosis requires histological examination. While tissue invasion is not typically observed, necrosis is commonly present.
Subacute Invasive Aspergillosis
Subacute invasive aspergillosis typically occurs in mildly immunocompromised patients over a period of 1 to 3 months. This infection presents with variable radiological features, including cavitation, nodules, and progressive consolidation with abscess formation. Biopsy reveals hyphae invading lung tissue, and microbiological investigations, including positive Aspergillus galactomannan antigens in blood or respiratory fluids, are consistent with invasive aspergillosis.
The above definitions are not mutually exclusive, and progression between disease categories may occur.[5] While not fully understood, the factors contributing to disease progression likely include a combination of immune function, treatment response, and preexisting lung pathology. This activity focuses on pulmonary aspergilloma and CCPA, with the terms aspergilloma, pulmonary mycetoma, and fungus ball used to describe the chronic colonization of Aspergillus species within preexisting lung cavities or bronchiectatic parenchyma.
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
Aspergillus species are ubiquitous saprophytes in the environment. Aspergillus is a filamentous fungus that thrives in moist conditions, such as soil, plants, and decaying vegetation. However, the conditions that promote the dispersion of spores include dry, dusty environments, hay barns, and compost sites. A fumigatus is the most common human pathogen, although A niger, A flavus, and A oryzae have also been reported to cause human disease.
Risk factors for Aspergillus-mediated lung disease (including aspergilloma and chronic cavitary pulmonary aspergillosis) are:
- Pulmonary tuberculosis
- Nontuberculous mycobacterial infection
- Cystic fibrosis
- Chronic bronchiectasis
- Pneumoconiosis
- Post-infarct pulmonary cavity
- Post-radiation pulmonary cavity
- Sarcoidosis
- Bronchial cysts and bullae
- Chronic lung abscess
- Lung malignancy
- Ankylosing spondylitis
- Allergic bronchopulmonary aspergillosis
Chronic Debilitating Conditions Affecting Local Bronchopulmonary Defense
- Malnutrition
- Chronic obstructive pulmonary disease
- Chronic liver disease
Immunosuppression
- Post-solid organ transplant
- Stem cell transplant
- Chemotherapy
- Neutropenia
- Prolonged corticosteroid use
- HIV
- Primary immunodeficiency syndromes
Aspergilloma and CCPA most commonly occur in immunocompetent individuals with preexisting structural lung disease. The worldwide prevalence is highest among those with residual lung cavities due to TB, with approximately 10% of preexisting tuberculous lung cavities developing into aspergillomas.[6][7] In addition to TB, individuals with nontuberculous mycobacteria, chronic obstructive pulmonary disease, and lung cancer are also at high risk. SARS-CoV-2 pneumonitis has recently been identified as a cause of aspergilloma, with several reported cases (see Image. HRCT Chest Scan Showing Aspergilloma With SARS-CoV-2 Pneumonitis Features).[8][9]
Epidemiology
The prevalence of CPA varies significantly across regions. In developed countries such as the United States, the prevalence is lower than 1 case per 100,000 individuals. However, in some African nations, the prevalence can be as high as 42.9 per 100,000 individuals. This difference is largely attributed to the higher prevalence of tuberculous pulmonary cavities in populations from resource-poor regions of the world.[10] The incidence of aspergilloma in patients with CPA is about 25%. The estimated global 5-year period prevalence is 18 cases per 100,000 individuals,[11] which translates to a global burden of 1.2 million patients. Higher incidence and prevalence are reported in Africa, the Western Pacific, and Southeast Asia.[11]
In resource-rich countries with a low prevalence of TB, the primary risk factor for CPA is chronic obstructive pulmonary disease. Isolated aspergilloma without preexisting parenchymal disease is much rarer, reported at 0.13%. Interestingly, although invasive Aspergillus disease is more common in patients with primary or acquired immunodeficiency, the incidence is relatively uncommon in HIV-infected patients.[12]
Pathophysiology
Human lungs are constantly exposed to airborne fungi, which are a common component of the external environmental microbiome. Among all fungal organisms, A fumigatus and A niger are frequent colonizers. Aspergillus spores primarily enter the respiratory tract and external auditory canal. These filamentous organisms rapidly multiply and become clinically significant in patients with pre-existing lung disease or systemic immunodeficiency.
Aspergillomas thrive in poorly drained, avascular cavitary spaces. Within these cavitary airspaces, the organism attaches to the wall with its conidia, germinates, and triggers an inflammatory response. Both aspergilloma and CCPA typically do not invade the surrounding lung tissue. Instead, the organism and inflammatory debris form an amorphous mass known as an aspergilloma. In the early stages, ulceration or an irregular, cobblestoned cavity wall or floor may be the only pathological finding. As the disease progresses, the aspergilloma becomes mobile within the cavity, which can be observed on imaging.
Hemoptysis is the most common clinical manifestation in symptomatic patients, with the bleeding typically originating from a bronchial vessel. This is usually secondary to:
- Direct invasion of the capillaries in the wall lining.
- Endotoxin release from the organism.
- Mechanical irritation of exposed vessels within the cavity.
- A rapidly growing cavity that erodes into the pleural surface and intercostal arteries, leading to massive, often fatal hemoptysis, which is highly challenging to control.
An aggressively growing aspergilloma increases the risk of exposure and erosion into the broader pulmonary arterial system. Triggered by hypoxia, inflammation, and architectural distortion, the opening of the pulmonary and bronchial arterial anastomotic plexus becomes a target for erosion, leading to hemoptysis. As previously noted, clinical features may overlap. Multiple fungal balls can develop, especially in patients with chronic cystic bronchiectasis or multicystic bullous disease. In some cases, aspergilloma may form within the bronchial tree (endobronchial aspergilloma) or the pleural space in chronic empyema.[13]
Histopathology
The organism is characterized by septate hyphae with dichotomous branching and long conidiophores that carry numerous spores at their tips. In histological specimens, aspergilloma appears as a network of inflamed, swollen septate hyphae, fibrin, blood clots, cellular debris, and mucous residues. The central core is often necrotic. Although the lining of a simple aspergilloma may consist of ciliated epithelium, it is more commonly composed of pseudostratified columnar or metaplastic squamous epithelium. Depending on the severity of the inflammation and the progression of the condition, the walls may show signs of chronic granulomatous inflammation, lymphoid follicles, endarteritis, ulceration, and fibrosis.
History and Physical
A general understanding of predisposition is essential for recognizing specific forms of Aspergillus-mediated disease. Individuals with atopy or a tendency toward hypersensitivity are more prone to obstructive bronchopulmonary aspergillosis. Immunocompromised patients are at higher risk for invasive Aspergillus infections, while aspergilloma and other forms of CPA are more common in immunocompetent individuals with preexisting lung disease. However, there is an overlap between these predisposition categories. Patients with aspergilloma can develop a hypersensitivity response similar to allergic bronchopulmonary aspergillosis.[14] Additionally, a chronic aspergilloma that remains unchanged over months to years can progress to CCPA, CFPA, or invasive infection.
Aspergilloma and CCPA can present with a wide range of clinical symptoms, from asymptomatic colonization of a preexisting lung cavity to potentially fatal hemoptysis. Additionally, patients with aspergilloma may exhibit symptoms related to their underlying lung disease, such as bullous emphysema, bronchiectasis, cavitary TB, cystic fibrosis, lung tumors, or sarcoidosis. According to various case series, hemoptysis is the most common clinical manifestation, occurring in 54% to 87.5% of cases.[15][16] Massive hemorrhage occurs in approximately 30% of patients. Notably, the size of an aspergilloma, lesion complexity, underlying lung disease, or prior minor hemoptysis do not reliably predict the risk of massive bleeding.[17][18] Although fever is rare, patients with CCPA may experience cough, chest pain, malaise, and weight loss. If the organism invades the surrounding lung parenchyma, recurrent pneumonia, chronic cough, or pulmonary fibrosis may develop.[19]
Evaluation
A presumptive diagnosis of aspergilloma and CCPA is made based on characteristic radiographic features in patients with known underlying lung disease. Occasionally, it may be discovered as an incidental finding in asymptomatic individuals. Once identified radiographically, additional diagnostic criteria include the presence of Aspergillus organisms in lower airway cultures obtained through bronchoalveolar lavage (BAL), a positive serum or BAL galactomannan test, and the detection of serum Aspergillus-specific immunoglobulin-G (IgG) antibodies.
Patients with a simple aspergilloma are not immunocompromised and typically experience minimal or no symptoms related to the fungal mass. Aspergilloma is often suspected based on findings from chest x-rays or computed tomography (CT) scans in patients being evaluated for symptoms related to underlying pulmonary diseases. For example, aspergillomas may be suspected in individuals with preexisting TB lung cavities, bronchiectasis, tumors, or chronic lung abscesses based on radiographic evidence. Alternatively, hemoptysis resulting from an aspergilloma may also prompt further evaluation. The European Society for Clinical Microbiology and Infectious Diseases, in collaboration with the European Respiratory Society and the Infectious Diseases Society of America, has published comprehensive guidelines for the diagnosis and management of CPA, including aspergilloma and CCPA.[1][4]
X-Ray
A chest x-ray is typically the initial diagnostic test for patients presenting with symptoms. An x-ray may reveal upper lobe–predominant cavitation, wall thickening, scarring, and, in some cases, a rounded mass-like opacity within the cavity.
Computed Tomography Scan
A CT scan provides greater detail and reveals characteristic features, including:
- The appearance of a solid spherical lesion within the cavitary space.
- A halo sign, indicating an inflammatory reaction in the cavity wall.
- An air crescent sign, where the fungus ball is separated from the cavity wall along part or all of its circumference (also seen in lung abscess, hydatid cyst, and granulomatosis with polyangiitis).
- The Monod sign, which is characterized by a shift in the fungus ball’s position within the cavity when the patient changes position during imaging (see Image. CT Scan of the Lung Showing Aspergilloma Mass With Monod Sign).
CT imaging helps define the thickness of the cavity wall, architectural distortion, and inflammation in the surrounding parenchyma and pleura. This also provides insights into the relationship between the aspergilloma and nearby blood vessels, as well as the nature of neovascularization in both the lung parenchyma and parietal pleura. The upper lobe of either lung is the most common location for aspergilloma, although the superior segment of the lower lobe can also be involved.
Radiographic Features
Radiographic features of CCPA may show areas of consolidation along with one or multiple cavities of varying wall thickness, often containing an aspergilloma. Adjacent pleural thickening is commonly observed. Simple aspergilloma can progress to CCPA, and both conditions can evolve into the fibrotic and subacute invasive forms of CPA.[20]
Radiographs provide a presumptive diagnosis of aspergilloma and CCPA. The presence of Aspergillus organisms in sputum is not diagnostic, as the fungi are ubiquitous and can colonize the airway without causing disease. However, in patients with characteristic radiographic findings, identifying Aspergillus organisms in BAL further supports the diagnosis. Conversely, the absence of organisms in the culture does not rule out the diagnosis. The role of molecular tests performed during bronchoscopy remains uncertain.
Aspergillus-specific IgG antibodies in serum provide strong presumptive evidence of CPA when characteristic radiological features are present. However, patients who acquired their infection in regions such as Pakistan, where A flavus and A fumigatus are predominant, may have negative serum serology tests unless species-specific assays are used.[21] The galactomannan antigen assay, targeting a polysaccharide component of the fungal cell wall, demonstrates high specificity. Typically, the sensitivity of the galactomannan antigen is higher in BAL fluid compared to serum. False-positive results may occur in patients receiving piperacillin-tazobactam, while false-negative results can arise with high-dose steroid therapy. IgG antibodies against Aspergillus species, detected by precipitin assay, are positive in over 90% of cases. Aspergillus IgG titers are typically elevated in patients with CPA and gradually decrease (although rarely becoming undetectable) with treatment.[22][23]
Aspergilloma and CCPA can coexist with active lung conditions, such as neoplasms and other infectious diseases.[24][25][26] These additional etiologies should be considered when evaluating patients with suspected aspergilloma and CCPA.
Treatment / Management
The management of CPA lacks standardized guidelines, as several aspects remain controversial due to the unpredictable nature of the infection and limited clinical experience.[13] Most published studies are small case series or anecdotal reports that do not clearly differentiate between the various types of CPA. The primary treatment goals are alleviating symptoms, preventing and minimizing hemoptysis episodes, and preventing progression to pulmonary fibrosis.
Approximately 10% of simple aspergillomas undergo spontaneous regression, while a significant number can lead to massive hemoptysis.[2] The ability to predict the severity of hemoptysis based on factors such as symptomatic versus asymptomatic presentation, aspergilloma size, extent, and associated underlying lung disease is limited.[2] Data suggest that the incidence and severity of hemoptysis are similar between simple and complex aspergillomas, with complex aspergilloma likely encompassing CCPA, although its definition remains vague.[2] An episode of minor hemoptysis predicts subsequent massive hemoptysis in approximately 30% of patients.[27] (B2)
Surgical resection, long considered the gold standard, can be associated with high morbidity and mortality, while antifungal therapy is often considered of limited value. The experience (or lack thereof) of the surgical and medical team can significantly impact the management outcome, which should be considered when evaluating published case series. Although guidelines have been developed, controversy persists regarding management approaches, especially for asymptomatic patients.[1][2][4] The Infectious Diseases Society of America recommends conservative, ongoing observation for asymptomatic patients with a single, stable aspergilloma. If symptoms like hemoptysis develop, resection is advised unless contraindicated. Perioperative or postoperative antifungal therapy should be considered if there is a risk of surgical spillage. In high-risk surgical candidates and those reluctant to undergo surgery, prolonged courses of triazoles are recommended.(A1)
Although surgical resection is considered the mainstay of therapy, it is important to recognize that not all patients with aspergilloma are suitable surgical candidates due to poor general health, low pulmonary reserve, and the presence of extensive disease. Before the development of triazoles and echinocandins, systemic antifungal therapy had a limited role in management. However, more recent experience with triazole and echinocandin therapy has demonstrated therapeutic benefits in patients deemed inoperable.[28] In patients with symptomatic CCPA, oral triazole therapy is now considered the standard of care.[1] Guidelines recommend an initial 4- to 6-month course of oral triazole therapy, with the duration extended for those who demonstrate slow or minimal response. Despite the lack of robust long-term data, lifelong triazole suppression may be worth considering, though no firm recommendations are currently recognized.
An alternative form of antifungal therapy should be considered for patients who do not respond to or are intolerant of oral triazole therapy. Triazole resistance can develop with prolonged administration. Systemic administration of amphotericin B desoxycholate has not shown significant benefits, likely due to its associated toxicities. In small published studies, intravenous (IV) liposomal amphotericin and IV echinocandins have demonstrated clinical benefits and should be considered for patients who do not respond, are intolerant, or have triazole-resistant organisms.[1]
Intracavitary installation of antifungal agents should be considered in nonsurgical candidates who experience recurrent hemoptysis or patients not responding to systemic antifungal therapy. Depending on the cavity size, location, the patient's pulmonary status, and the medical team's experience, intracavitary antifungal installation can be performed via an endobronchial or percutaneous catheter.[29][30][28] These methods should be considered short-term therapeutic options, with potential complications including pneumothorax and cough. Transbronchial removal of aspergilloma, a nonsurgical approach, has recently shown promise in selected patients.[31][32] However, its application and value in management await further evaluation.(B2)
In patients with mild-to-moderate hemoptysis, oral tranexamic acid may be administered to help stabilize clot formation.[33] Antifungal therapy can also prevent the recurrence of hemoptysis. In cases of massive hemoptysis, bronchial artery embolization by interventional radiology has proven successful in 50% to 90% of cases, although recurrence rates remain high.[34] Subsequent antifungal therapy should be initiated following embolization. In some instances, embolization of the internal mammary, subclavian, and lateral thoracic arteries may be necessary to control hemoptysis. However, embolization of arteries near the origins of the spinal and vertebral arteries should be avoided to prevent spinal cord infarcts and paralysis.[35] Recent data from centers with experienced surgical teams report operative mortality between 0.9% and 3.3%, with morbidity rates ranging from 23.6% to 33.3%.[36][37] (A1)
The decision to proceed with surgery should be tailored to the individual, taking into account several factors, including:
- The patient's overall functional status and pulmonary reserve
- The experience of the surgical team
- Presenting symptoms, such as asymptomatic, cough, chest pain, and weight loss
- The presence or absence of hemoptysis and the degree of its severity
- An indeterminate diagnosis and the potential presence of an underlying neoplasm
Surgical approaches for aspergilloma include lobectomy, pneumonectomy, segmentectomy, cavernectomy, and pleurectomy. Video-assisted thoracoscopy can be a valuable option for select patients.[22] Surgical complications may include bronchopleural fistula, pneumonia, empyema, respiratory failure, hemorrhage, and death. Surgical removal of a simple aspergilloma is typically considered curative. However, if there is intraoperative spillage of the cavity contents or if the entire aspergilloma cannot be removed, a postoperative course of antifungal therapy is recommended.[1] However, the duration of antifungal treatment in the postoperative setting is empiric and should be individualized.
Removal of a simple aspergilloma is considered curative. However, complex aspergillomas and CCPA often require prolonged courses of antifungal therapy, and a complete cure may not be achievable. In these cases, the goal is to stabilize the disease and reduce hemoptysis. Follow-up should include chest CT scans every 3 to 6 months initially. If radiographic findings stabilize or improve, the frequency of scans can be reduced.[38] Serial Aspergillus-IgG serology should also be monitored, with a decline in antibody titers indicating a positive response to therapy.
Differential Diagnosis
A space-occupying lesion within a lung cavity may be attributed to various conditions, including:
- Primary lung malignancy
- Metastatic disease
- Aspergilloma
- Hydatid cyst
- Lung abscess
The differential diagnoses of CCPA include:
- Active tuberculosis
- Nontuberculous mycobacteria
- Histoplasmosis
- Coccidioidomycosis
- Actinomycosis
- Neoplasm
Prognosis
The prognosis of aspergilloma and CCPA is difficult to quantify due to their unpredictable course, the presence of associated lung diseases, and the patient's functional status. Spontaneous resolution occurs in approximately 10% of simple aspergilloma cases. The recent availability of triazole and echinocandin antifungal agents in symptomatic patients has shown benefits in about 65% of those treated. Similar benefits have been observed with intracavitary instillation of antifungals. However, the impact of these treatments on long-term prognosis remains unclear, and symptomatic improvements do not necessarily correlate with life expectancy.
The mortality rate for patients experiencing massive hemoptysis from any cause has historically been reported as high as 38%.[39] Whether this figure applies to current management strategies for aspergilloma and CCPA remains uncertain. After successful bronchial artery embolization, the recurrence of hemoptysis occurs in 19% to 55% of cases.[28] The effect of new antifungal agents and nonsurgical management on the prognosis of aspergilloma and CCPA requires further investigation.
Complications
Hemoptysis is the most common and feared complication. Additional complications related to the underlying lung disease are expected. Potential complications may arise from the use of antifungal agents, whether administered systemically or through intracavitary instillation. Intraoperative and postoperative complications, as outlined above, should be carefully considered when assessing preoperative risks for each patient. The natural history of simple aspergilloma and CCPA is poorly understood, with the conditions either remaining relatively stable or progressing to more extensive and invasive lung pathology.
Deterrence and Patient Education
Aspergilloma and CCPA are most prevalent in resource-poor areas, primarily due to their association with preexisting TB lung cavities. These regions often face significant limitations in diagnosis and treatment. Regardless of location, once patients with aspergilloma and CCPA are diagnosed, their management options may be restricted by available resources and the experience of the healthcare team. Therapeutic choices can also be limited by the patient's poor functional and pulmonary status.
If antifungal therapy is chosen as the treatment approach, it typically involves prolonged courses, which can significantly affect cost considerations and require monitoring for potential adverse effects. Additionally, once treatment is initiated, close follow-up will be necessary over many years to assess the therapeutic response. The decision to pursue surgery should consider the experience of the surgical team.[2]
The management of aspergilloma and CCPA has evolved over the past decade with the introduction of new antifungal agents and alternative nonsurgical and surgical therapies. However, due to the relatively low prevalence of the disease, many clinicians have limited experience, particularly in developed regions. This limited experience is further compounded by reliance on somewhat outdated studies in the medical literature. Controversy exists regarding certain therapeutic and follow-up strategies, as well as a lack of clinical studies. These challenges complicate the diagnosis and management of this complex infectious disease.
Enhancing Healthcare Team Outcomes
Aspergilloma and CCPA are disease entities that are classified under the broader category of CPA. These infectious diseases are most commonly accompanied by preexisting lung disorders, with the most frequent association occurring in patients with TB. Diagnosis involves identifying characteristic radiographic features along with microbiological studies and/or immunological evidence of Aspergillus species. Once the diagnosis is confirmed, a thorough evaluation of the patient's functional and pulmonary status, along with the presence or absence of symptoms related to the fungus, must be conducted.
Individualized management options, such as conservative observation, antifungal therapy, or surgical intervention, should be considered. When developing follow-up strategies for both short- and long-term treatment, it is important to account for the financial burden on patients, particularly if the current therapy is ineffective or not tolerated. In cases of complications, especially hemoptysis, immediate medical evaluation and treatment resources must be readily available.
All of the above necessitates a coordinated effort from the healthcare team. Expertise in radiology, infectious diseases, pulmonary and critical care, thoracic surgery, emergency medicine, and interventional radiology is essential. Additionally, pharmacists knowledgeable in administering oral, IV, and intracavitary antifungals are crucial members of the multidisciplinary healthcare team, along with radiology technicians and nurses from inpatient, operating room, and outpatient settings.
The education of the healthcare team is critical in the decision-making process, especially considering the challenges posed by a lack of robust evidence-based studies that form clear guidelines. The educational component should emphasize that, at present, evidence-based data addressing key management questions is limited. Nearly all decisions regarding the diagnosis, management, and follow-up of patients with aspergilloma and CCPA require close communication and coordination among a large, multidisciplinary healthcare team.
Media
(Click Image to Enlarge)
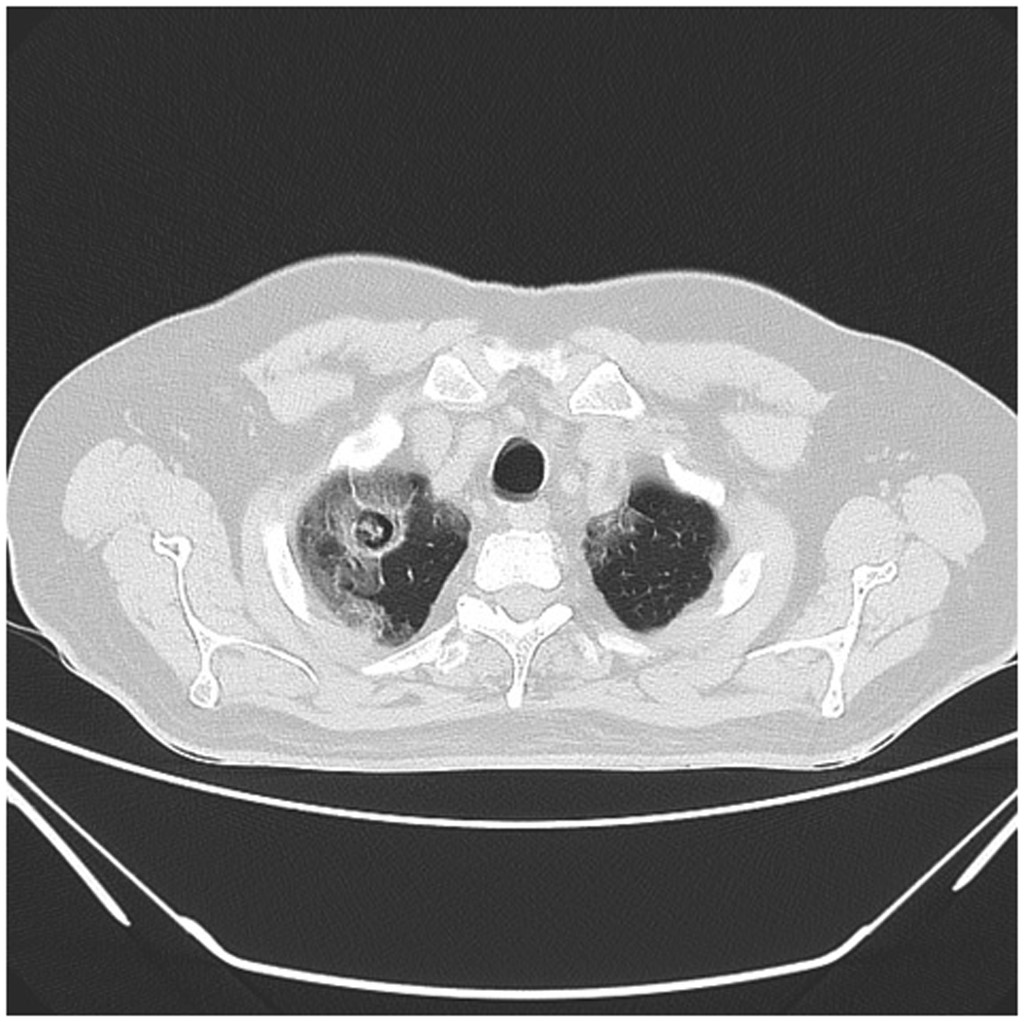
CT Scan of the Lung Showing Aspergilloma Mass With Monod Sign. An axial computed tomography (CT) scan of the lung demonstrates a round, enhancing lesion with a surrounding air shadow in the superior basal segment of the right lower lobe, measuring 2.5 cm × 1.6 cm. This finding is characteristic of the Monod sign and indicates an aspergilloma.
Contributed by S Sarkar, MD, and B Ghewade, MD
(Click Image to Enlarge)
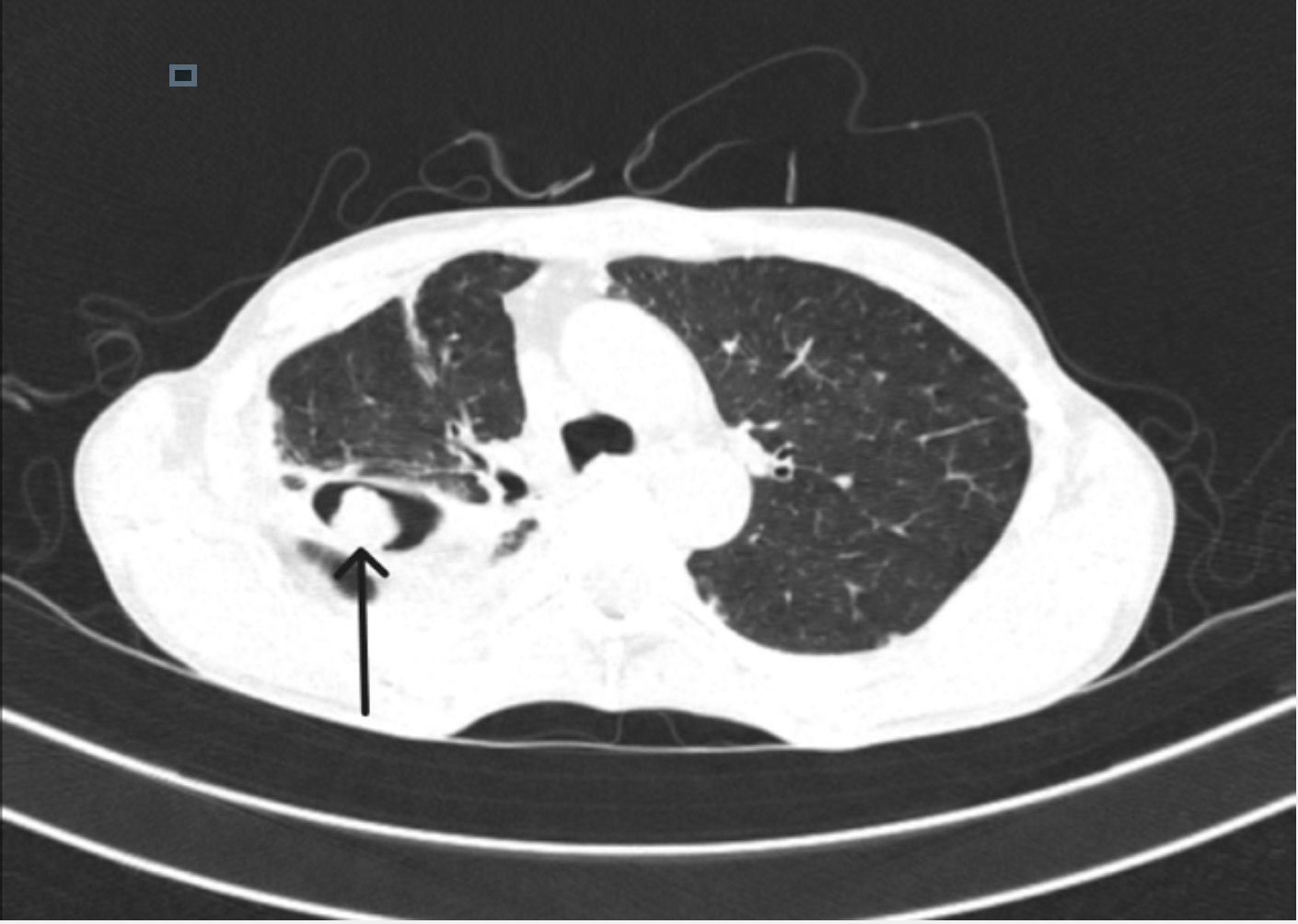
HRCT Chest Scan Showing Aspergilloma With SARS-CoV-2 Pneumonitis Features. A high-resolution computed tomography (HRCT) scan of the chest reveals bilateral peripheral ground-glass opacities and a right upper lobe cavity measuring 2.3 × 2.3 cm and containing an opacity, likely a fungal ball. These findings are consistent with SARS-CoV-2 pneumonitis and indicate an aspergilloma.
Chaurasia S, Thimmappa M, Chowdhury S. Case report: chronic cavitatory pulmonary aspergillosis after COVID-19. Am J Trop Med Hyg. 2021;106(1):105-107. doi: 10.4269/ajtmh.21-0701.
References
Denning DW, Cadranel J, Beigelman-Aubry C, Ader F, Chakrabarti A, Blot S, Ullmann AJ, Dimopoulos G, Lange C, European Society for Clinical Microbiology and Infectious Diseases and European Respiratory Society. Chronic pulmonary aspergillosis: rationale and clinical guidelines for diagnosis and management. The European respiratory journal. 2016 Jan:47(1):45-68. doi: 10.1183/13993003.00583-2015. Epub [PubMed PMID: 26699723]
Muniappan A, Tapias LF, Butala P, Wain JC, Wright CD, Donahue DM, Gaissert HA, Lanuti M, Mathisen DJ. Surgical therapy of pulmonary aspergillomas: a 30-year North American experience. The Annals of thoracic surgery. 2014 Feb:97(2):432-8. doi: 10.1016/j.athoracsur.2013.10.050. Epub 2013 Dec 21 [PubMed PMID: 24365218]
Level 2 (mid-level) evidenceGazzoni FF, Severo LC, Marchiori E, Guimarães MD, Garcia TS, Irion KL, Camargo JJ, Felicetti JC, de Mattos Oliveira F, Hochhegger B. Pulmonary diseases with imaging findings mimicking aspergilloma. Lung. 2014 Jun:192(3):347-57. doi: 10.1007/s00408-014-9568-7. Epub 2014 Mar 11 [PubMed PMID: 24615678]
Patterson TF, Thompson GR 3rd, Denning DW, Fishman JA, Hadley S, Herbrecht R, Kontoyiannis DP, Marr KA, Morrison VA, Nguyen MH, Segal BH, Steinbach WJ, Stevens DA, Walsh TJ, Wingard JR, Young JA, Bennett JE. Practice Guidelines for the Diagnosis and Management of Aspergillosis: 2016 Update by the Infectious Diseases Society of America. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2016 Aug 15:63(4):e1-e60. doi: 10.1093/cid/ciw326. Epub 2016 Jun 29 [PubMed PMID: 27365388]
Level 1 (high-level) evidenceHayes GE, Novak-Frazer L. Chronic Pulmonary Aspergillosis-Where Are We? and Where Are We Going? Journal of fungi (Basel, Switzerland). 2016 Jun 7:2(2):. doi: 10.3390/jof2020018. Epub 2016 Jun 7 [PubMed PMID: 29376935]
Volpe-Chaves CE, Venturini J, B Castilho S, S O Fonseca S, F Nunes T, T Cunha EA, M E Lima G, O Nunes M, P Vicentini A, V L Oliveira SM, Carvalho LR, Thompson L, P Mendes R, M M Paniago A. Prevalence of chronic pulmonary aspergillosis regarding time of tuberculosis diagnosis in Brazil. Mycoses. 2022 Jul:65(7):715-723. doi: 10.1111/myc.13465. Epub 2022 May 15 [PubMed PMID: 35524507]
Ocansey BK, Otoo B, Adjei A, Gbadamosi H, Kotey FCN, Kosmidis C, Afriyie-Mensah JS, Denning DW, Opintan JA. Chronic pulmonary aspergillosis is common among patients with presumed tuberculosis relapse in Ghana. Medical mycology. 2022 Sep 9:60(9):. doi: 10.1093/mmy/myac063. Epub [PubMed PMID: 35953428]
Chaurasia S, Thimmappa M, Chowdhury S. Case Report: Chronic Cavitatory Pulmonary Aspergillosis after COVID-19. The American journal of tropical medicine and hygiene. 2021 Nov 24:106(1):105-107. doi: 10.4269/ajtmh.21-0701. Epub 2021 Nov 24 [PubMed PMID: 34818624]
Level 3 (low-level) evidenceKoehler P, Bassetti M, Chakrabarti A, Chen SCA, Colombo AL, Hoenigl M, Klimko N, Lass-Flörl C, Oladele RO, Vinh DC, Zhu LP, Böll B, Brüggemann R, Gangneux JP, Perfect JR, Patterson TF, Persigehl T, Meis JF, Ostrosky-Zeichner L, White PL, Verweij PE, Cornely OA, European Confederation of Medical Mycology, International Society for Human Animal Mycology, Asia Fungal Working Group, INFOCUS LATAM/ISHAM Working Group, ISHAM Pan Africa Mycology Working Group, European Society for Clinical Microbiology, Infectious Diseases Fungal Infection Study Group, ESCMID Study Group for Infections in Critically Ill Patients, Interregional Association of Clinical Microbiology and Antimicrobial Chemotherapy, Medical Mycology Society of Nigeria, Medical Mycology Society of China Medicine Education Association, Infectious Diseases Working Party of the German Society for Haematology and Medical Oncology, Association of Medical Microbiology, Infectious Disease Canada. Defining and managing COVID-19-associated pulmonary aspergillosis: the 2020 ECMM/ISHAM consensus criteria for research and clinical guidance. The Lancet. Infectious diseases. 2021 Jun:21(6):e149-e162. doi: 10.1016/S1473-3099(20)30847-1. Epub 2020 Dec 14 [PubMed PMID: 33333012]
Level 3 (low-level) evidenceWang F, Zhao L, Chen L, Chen J, Chen S, Ren Y. Risk Factors of Chronic Pulmonary Aspergillosis in Patients with Etiology Positive Pulmonary Tuberculosis. Alternative therapies in health and medicine. 2024 Jan:30(1):83-87 [PubMed PMID: 37820672]
Denning DW, Pleuvry A, Cole DC. Global burden of chronic pulmonary aspergillosis as a sequel to pulmonary tuberculosis. Bulletin of the World Health Organization. 2011 Dec 1:89(12):864-72. doi: 10.2471/BLT.11.089441. Epub 2011 Sep 27 [PubMed PMID: 22271943]
Level 2 (mid-level) evidenceAddrizzo-Harris DJ, Harkin TJ, McGuinness G, Naidich DP, Rom WN. Pulmonary aspergilloma and AIDS. A comparison of HIV-infected and HIV-negative individuals. Chest. 1997 Mar:111(3):612-8 [PubMed PMID: 9118696]
Level 2 (mid-level) evidenceMoodley L, Pillay J, Dheda K. Aspergilloma and the surgeon. Journal of thoracic disease. 2014 Mar:6(3):202-9. doi: 10.3978/j.issn.2072-1439.2013.12.40. Epub [PubMed PMID: 24624284]
Ein ME, Wallace RJ Jr, Williams TW Jr. Allergic bronchopulmonary aspergillosis-like syndrome consequent to aspergilloma. The American review of respiratory disease. 1979 May:119(5):811-20 [PubMed PMID: 378049]
Level 3 (low-level) evidenceBabatasi G, Massetti M, Chapelier A, Fadel E, Macchiarini P, Khayat A, Dartevelle P. Surgical treatment of pulmonary aspergilloma: current outcome. The Journal of thoracic and cardiovascular surgery. 2000 May:119(5):906-12 [PubMed PMID: 10788811]
Level 2 (mid-level) evidencePassera E, Rizzi A, Robustellini M, Rossi G, Della Pona C, Massera F, Rocco G. Pulmonary aspergilloma: clinical aspects and surgical treatment outcome. Thoracic surgery clinics. 2012 Aug:22(3):345-61. doi: 10.1016/j.thorsurg.2012.04.001. Epub [PubMed PMID: 22789598]
Rafferty P, Biggs BA, Crompton GK, Grant IW. What happens to patients with pulmonary aspergilloma? Analysis of 23 cases. Thorax. 1983 Aug:38(8):579-83 [PubMed PMID: 6612648]
Level 2 (mid-level) evidenceJewkes J, Kay PH, Paneth M, Citron KM. Pulmonary aspergilloma: analysis of prognosis in relation to haemoptysis and survey of treatment. Thorax. 1983 Aug:38(8):572-8 [PubMed PMID: 6612647]
Level 3 (low-level) evidenceLipinski JK, Weisbrod GL, Sanders DE. Unusual manifestations of pulmonary aspergillosis. Journal of the Canadian Association of Radiologists. 1978 Dec:29(4):216-20 [PubMed PMID: 363725]
Level 3 (low-level) evidenceKosmidis C, Denning DW. The clinical spectrum of pulmonary aspergillosis. Thorax. 2015 Mar:70(3):270-7. doi: 10.1136/thoraxjnl-2014-206291. Epub 2014 Oct 29 [PubMed PMID: 25354514]
Jabeen K, Farooqi J, Iqbal N, Wahab K, Irfan M. Aspergillus fumigatus and Aspergillus flavus-Specific IgG Cut-Offs for the Diagnosis of Chronic Pulmonary Aspergillosis in Pakistan. Journal of fungi (Basel, Switzerland). 2020 Oct 26:6(4):. doi: 10.3390/jof6040249. Epub 2020 Oct 26 [PubMed PMID: 33114653]
Level 2 (mid-level) evidenceJiang C, Ge T, Jiang G, Zhu Y, Zhang P. Single- versus multi-port video-assisted thoracic surgery for pulmonary aspergilloma: a propensity-matched study. Interdisciplinary cardiovascular and thoracic surgery. 2023 May 4:36(5):. doi: 10.1093/icvts/ivad016. Epub [PubMed PMID: 37141922]
LONGBOTTOM JL, PEPYS J, CLIVE FT. DIAGNOSTIC PRECIPITIN TEST IN ASPERGILLUS PULMONARY MYCETOMA. Lancet (London, England). 1964 Mar 14:1(7333):588-9 [PubMed PMID: 14104489]
Adhikari A, Khanal S, Magar SR, Thapa S, Khati S, Lamichhane R, Marasini K. Aspergillus Coinfection in a Hydatid Cyst Cavity of Lung in an Immunocompetent Host: A Case Report and Review of Literature. Case reports in infectious diseases. 2023:2023():6975041. doi: 10.1155/2023/6975041. Epub 2023 Jul 14 [PubMed PMID: 37485286]
Level 3 (low-level) evidenceHirano T, Yamada M, Igusa R, Tanno A, Numakura T, Sakamoto K, Kikuchi T, Ichinose M. Two cases of endobronchial aspergilloma complicated with primary and metastatic lung cancer: A case report and literature review. Respiratory investigation. 2016 May:54(3):211-5. doi: 10.1016/j.resinv.2015.12.005. Epub 2016 Feb 16 [PubMed PMID: 27108018]
Level 3 (low-level) evidenceKumar AA, Shantha GP, Jeyachandran V, Rajkumar K, Natesan S, Srinivasan D, Joseph LD, Sundaresan M, Rajamanickam D. Multidrug resistant tuberculosis co-existing with aspergilloma and invasive aspergillosis in a 50 year old diabetic woman: a case report. Cases journal. 2008 Nov 8:1(1):303. doi: 10.1186/1757-1626-1-303. Epub 2008 Nov 8 [PubMed PMID: 18992166]
Level 3 (low-level) evidenceChen QK, Jiang GN, Ding JA. Surgical treatment for pulmonary aspergilloma: a 35-year experience in the Chinese population. Interactive cardiovascular and thoracic surgery. 2012 Jul:15(1):77-80. doi: 10.1093/icvts/ivs130. Epub 2012 Apr 11 [PubMed PMID: 22499801]
Level 2 (mid-level) evidenceLang M, Lang AL, Chauhan N, Gill A. Non-surgical treatment options for pulmonary aspergilloma. Respiratory medicine. 2020 Apr:164():105903. doi: 10.1016/j.rmed.2020.105903. Epub 2020 Feb 19 [PubMed PMID: 32217289]
Kravitz JN, Berry MW, Schabel SI, Judson MA. A modern series of percutaneous intracavitary instillation of amphotericin B for the treatment of severe hemoptysis from pulmonary aspergilloma. Chest. 2013 May:143(5):1414-1421. doi: 10.1378/chest.12-1784. Epub [PubMed PMID: 23117277]
Level 2 (mid-level) evidenceMohan A, Tiwari P, Madan K, Hadda V, Poulose R, Bhalla AS, Khandelwal R, Khilnani GC, Guleria R. Intrabronchial Voriconazole is a Safe and Effective Measure for Hemoptysis Control in Pulmonary Aspergilloma. Journal of bronchology & interventional pulmonology. 2017 Jan:24(1):29-34 [PubMed PMID: 27623423]
Stather DR, Tremblay A, Dumoulin E, MacEachern P, Chee A, Hergott C, Gelberg J, Gelfand GA, Burrowes P, MacGregor JH, Mody CH. A Series of Transbronchial Removal of Intracavitary Pulmonary Aspergilloma. The Annals of thoracic surgery. 2017 Mar:103(3):945-950. doi: 10.1016/j.athoracsur.2016.08.069. Epub 2016 Oct 17 [PubMed PMID: 27765172]
Stather DR, Tremblay A, MacEachern P, Chee A, Dumoulin E, Tourin O, Gelfand GA, Mody CH. Bronchoscopic removal of a large intracavitary pulmonary aspergilloma. Chest. 2013 Jan:143(1):238-241. doi: 10.1378/chest.12-0400. Epub [PubMed PMID: 23276848]
Level 3 (low-level) evidenceAlabdrabalnabi F, Alshahrani M, Ismail N. Nebulized tranexamic acid for recurring hemoptysis in critically ill patients: case series. International journal of emergency medicine. 2020 Aug 20:13(1):45. doi: 10.1186/s12245-020-00304-x. Epub 2020 Aug 20 [PubMed PMID: 32819268]
Level 2 (mid-level) evidencePanda A, Bhalla AS, Goyal A. Bronchial artery embolization in hemoptysis: a systematic review. Diagnostic and interventional radiology (Ankara, Turkey). 2017 Jul-Aug:23(4):307-317. doi: 10.5152/dir.2017.16454. Epub [PubMed PMID: 28703105]
Level 1 (high-level) evidenceIshikawa H, Ohbe H, Omachi N, Morita K, Yasunaga H. Spinal Cord Infarction after Bronchial Artery Embolization for Hemoptysis: A Nationwide Observational Study in Japan. Radiology. 2021 Mar:298(3):673-679. doi: 10.1148/radiol.2021202500. Epub 2021 Jan 19 [PubMed PMID: 33464182]
Level 2 (mid-level) evidenceAkbari JG, Varma PK, Neema PK, Menon MU, Neelakandhan KS. Clinical profile and surgical outcome for pulmonary aspergilloma: a single center experience. The Annals of thoracic surgery. 2005 Sep:80(3):1067-72 [PubMed PMID: 16122488]
Level 2 (mid-level) evidencePark CK, Jheon S. Results of surgical treatment for pulmonary aspergilloma. European journal of cardio-thoracic surgery : official journal of the European Association for Cardio-thoracic Surgery. 2002 May:21(5):918-23 [PubMed PMID: 12062287]
Level 2 (mid-level) evidenceGodet C, Laurent F, Bergeron A, Ingrand P, Beigelman-Aubry C, Camara B, Cottin V, Germaud P, Philippe B, Pison C, Toper C, Carette MF, Frat JP, Béraud G, Roblot F, Cadranel J, ACHROSCAN Study Group. CT Imaging Assessment of Response to Treatment in Chronic Pulmonary Aspergillosis. Chest. 2016 Jul:150(1):139-47. doi: 10.1016/j.chest.2016.02.640. Epub 2016 Feb 19 [PubMed PMID: 26905365]
Hirshberg B, Biran I, Glazer M, Kramer MR. Hemoptysis: etiology, evaluation, and outcome in a tertiary referral hospital. Chest. 1997 Aug:112(2):440-4 [PubMed PMID: 9266882]
Level 2 (mid-level) evidence