Introduction
Eosinophilic granuloma is the mildest variant of Langerhans cell histiocytosis (LCH), a group of disorders characterized by the abnormal proliferation of antigen-presenting Langerhans cells. First described by Lichtenstein and Jaffe in 1940, the term "eosinophilic granuloma" was later grouped with Hand-Schüller-Christian disease and Letterer-Siwe disease under the umbrella term "histiocytosis X," which referred to histiocyte proliferation of unknown cause. This classification was renamed Langerhans cell histiocytosis in subsequent years. Eosinophilic granuloma is a benign tumor-like condition where Langerhans cells, originating from myeloid dendritic cells rather than skin, proliferate abnormally. Eosinophilic granuloma is the most common form of LCH and accounts for <1% of all bone tumors.[1]
The disease predominantly affects the axial skeleton, including the skull, jaw, spine, pelvis, ribs, and long bones, with long bone lesions typically located in the diaphysis. Adjacent soft tissues are often involved. The pattern of bone involvement varies with age: in children, the frontal bone of the skull is the most common site, while in adults, the jaw is more frequently affected. Similarly, the thoracic spine is commonly involved in children, whereas adults often exhibit cervical spine involvement.[2][3] Other less frequently affected organs include the skin, pituitary gland, lungs, brain, liver, spleen, and gastrointestinal tract.[4][5][6][7][8] Eosinophilic granuloma can present as a solitary lesion, which rarely requires treatment, or as a multisystem disease necessitating aggressive therapy.
Langerhans Cell Histiocytosis Classification
Langerhans cell histiocytosis is classified [9] into 3 main types:
-
Eosinophilic granuloma
- Monostotic: A single bone lesion, observed in approximately 90% of cases.
- Polyostotic: Multiple bone lesions, found in about 10% of cases.
-
Hand-Schüller-Christian disease: A systemic condition presenting with the classic triad of exophthalmos, diabetes insipidus, and osteolytic skull lesions, Hand-Schüller-Christian disease typically affects children older than 3 years and has a worse prognosis due to extraskeletal involvement.[10]
-
Letterer-Siwe disease: A severe systemic disease characterized by lymphadenopathy, skin rash, hepatosplenomegaly, and pancytopenia.[11] Letterer-Siwe disease usually presents in children younger than 3 years and is frequently fatal.
The age-related differences in presentation, site predilection, and systemic involvement of eosinophilic granuloma underscore the importance of tailored evaluation and management strategies.
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
The classification of eosinophilic granuloma as a reactive or neoplastic process remains a topic of debate. Eosinophilic granulomas are characterized by the abnormal proliferation of Langerhans cells, which are derived from mononuclear and dendritic precursor cells. These cells are primarily located in the bone marrow but have the ability to migrate into tissues, where they act as antigen-presenting cells to T lymphocytes. The proliferation of Langerhans cells may be triggered by viral infections (such as Epstein-Barr virus or Human Herpesvirus-6), bacterial infections, or immune dysfunction, leading to increased levels of cytokines like interleukin-1 and interleukin-10.[12] In particular, eosinophilic granulomas of the lung are strongly associated with cigarette smoking, further underscoring the role of external triggers in the disease process.[13]
The 2016 revised classification by the Histiocyte Society recognizes LCH as an inflammatory myeloid neoplasm.[14] This classification is supported by evidence linking antigenic stimulation to disease development. In the lungs, Langerhans cells accumulate in response to cigarette smoke exposure, with histologic and radiographic findings initially observed in peribronchial regions. Experts hypothesize that specific antigens in cigarette smoke stimulate this pathological response, emphasizing the connection between environmental exposure and disease progression.
Epidemiology
Eosinophilic granuloma is a rare disorder with an incidence of 4 to 5 cases per million per year in children younger than 15 years and 1 to 2 cases per million per year in adults.[15] The condition is more prevalent in individuals of white northern European descent and Hispanics compared to those of Black racial backgrounds. It typically affects children, adolescents, and young adults, with the most commonly affected age group being 5 to 10 years. Among patients with single bone lesions, the mean age of onset is approximately 5.5 years, while those with multiple bone lesions have a mean age of 4.5 years. Males are slightly more affected than females, with a male-to-female ratio of 1.2:1. Although rare, a few cases of eosinophilic granuloma occurring within the same family have been reported, but the inheritance pattern remains unclear.
Eosinophilic granuloma of the lungs is also a rare disorder, and its true prevalence is not well established. Less than 5% of patients undergoing lung biopsy for interstitial lung disease are diagnosed with lung eosinophilic granulomas.[16] A study in Japan estimated its prevalence at 0.27 cases per 100,000 males and 0.07 cases per 100,000 females based on hospital discharge diagnoses within a single year.[17]
Pathophysiology
The pathophysiology of eosinophilic granuloma remains poorly understood. Langerhans cells, differentiated members of the dendritic cell system closely related to the monocyte-macrophage lineage, play a central role. These antigen-presenting cells are normally found in the skin, reticuloendothelial system, heart, pleura, and lungs and can be identified through immunohistochemical staining or the presence of Birbeck granules on electron microscopy. The condition may arise from either a reactive or neoplastic process. Reports suggest somatic mutations, including a gain-of-function mutation in the BRAF V600E gene in approximately 50% of cases and MAP2K1 mutations in about 21%.[18][19] Langerhans cell histiocytosis has also been linked to mutations in the Ras-ERK pathway, with studies showing that 100% of cases involve ERK phosphorylation.[19][14]
The BRAF gene produces a protein involved in the RAS/MAPK signaling pathway, which regulates cellular proliferation, differentiation, migration, and apoptosis. Mutations in this gene result in an overactive protein that continuously signals cell growth and proliferation. This leads to the abnormal proliferation of Langerhans cells and the subsequent formation of eosinophilic granulomas, which may affect the skeletal system or other organs. The dendritic cell maturation stage at the time of mutation influences clinical presentation, with more mature cells resulting in less systemic involvement. Cases involving multiple organ systems are often associated with the BRAF mutation, which has shown responsiveness to chemotherapy.[14][18][20]
Histopathology
Histopathological examination is essential for diagnosing eosinophilic granulomatosis, as its clinical presentation and laboratory findings can resemble those of other conditions, including infections, malignancies, and benign bone tumors.
Histopathological Findings
The histopathological findings reveal Langerhans cells, which are mononuclear, histiocyte-like cells characterized by prominent nuclear grooves resembling a "coffee bean" and a pink appearance due to an eosinophilic cytoplasm. Other findings include scattered multinucleated Touton-like giant cells, inflammatory cells, and areas of necrosis. Immunohistochemical staining typically shows positivity for CD1 antigen, S-100 protein, CD207 (Langerin), cyclin D1, PNA (peanut agglutinin), and BRAF VE1 (in 50% of cases). A lack of nuclear atypia and atypical mitoses helps differentiate eosinophilic granulomatosis from malignant conditions.[20]
Electron Microscopy Findings
Electron microscopy further aids in the diagnosis by identifying Birbeck granules within Langerhans cells. These granules are "tennis racket-shaped" cytoplasmic inclusions with a zipper-like appearance, believed to play a role in antigen storage (see Image. Histiocytosis Microscopy).
History and Physical
Clinical Symptoms
Eosinophilic granuloma is a multisystem disease with diverse clinical presentations, typically involving 1 or more bones and occasionally other organs, including the skin, pituitary gland, liver, spleen, and lungs. The symptoms depend on the affected structures and surrounding tissues. The most common presentation includes pain and swelling in the region of the involved bone, often accompanied by pathological fractures. Vertebral lesions may cause back pain, stiffness, postural changes like kyphosis, and, in severe cases, neurological deficits.[21] Temporal bone involvement can present as ear canal masses, postauricular swelling, ear discharge, and hearing loss, especially in children with persistent otitis media unresponsive to antibiotics.
Periorbital lesions can mimic cellulitis, presenting with edema, redness, fever, and proptosis that does not improve with antibiotics.[22] Jaw lesions, though rare, may manifest as jaw masses, pain, and loose teeth.[23] Skull involvement can lead to headaches, increased thirst, and urination, suggesting diabetes insipidus. Pulmonary lesions are often asymptomatic but may cause nonproductive cough, weight loss, dyspnea, chest pain, fatigue, or spontaneous pneumothorax.
Physical Examination Findings
Physical findings vary based on the location and extent of the disease. Tenderness, restricted spinal mobility, or firm, immobile masses may be noted. Lesions in the pelvis or lower extremities can cause limping, while conductive or sensorineural hearing loss may result from temporal bone involvement. Neurological deficits may be observed in extensive spinal lesions, and abdominal examinations may reveal hepatosplenomegaly and enlarged lymph nodes in multisystem disease. Chest auscultation may detect abnormalities in cases with pulmonary involvement. This wide array of clinical features underscores the importance of thorough evaluation for accurate diagnosis and management.
Evaluation
A thorough, multicentric approach, including laboratory tests, radiographic studies, and biopsy, is required to diagnose an eosinophilic granuloma accurately.
Laboratory Studies
Laboratory investigations often begin with a complete blood count with differential, which is typically normal but may reveal mild leukocytosis. This test is crucial for excluding conditions that mimic eosinophilic granuloma, eg, osteomyelitis or malignancies. If unexplained cytopenias are detected, further evaluation with a bone marrow biopsy is necessary. The erythrocyte sedimentation rate (ESR) may be elevated in some cases, though this is not a consistent finding.[24]
Imaging Studies
Imaging studies play a pivotal role in diagnosing eosinophilic granuloma. X-rays of affected bones may reveal characteristic "punched-out" lytic lesions, with or without a periosteal reaction. In the skull, both the outer and inner tables may be involved, creating a beveled-edge appearance. Spinal x-rays can show lytic lesions, vertebral collapse (vertebra plana), and kyphosis. Differential diagnoses for these findings include multiple myeloma, plasmacytoma, osteomyelitis, tuberculosis, leukemia, and lymphoma.
A complete skeletal survey is often performed to identify additional lesions, alongside chest x-rays to evaluate for pulmonary involvement. Pulmonary involvement typically appears as bilateral, symmetric, ill-defined nodules and reticulonodular infiltrates. As the disease advances, cystic lesions may develop, with an upper lobe predominance and sparing of costophrenic angles.
Advanced imaging, including computed tomography (CT), magnetic resonance imaging (MRI), and positron emission tomography (PET), is recommended for cases involving complex anatomy, eg, the skull, mandible, and spine. These scans provide detailed visualization of punched-out lesions and any associated soft tissue involvement. Radionuclide bone scans can highlight areas of increased uptake at affected sites.
Biopsy Evaluation
Biopsy remains essential for definitive diagnosis. Fine-needle aspiration or CT-guided biopsy of the suspected lesion, with staining for CD1a and CD207, is performed to confirm eosinophilic granuloma.[20] Testing for the BRAF V600E mutation is indicated in cases involving central nervous system lesions, diagnostic uncertainty, or the need for targeted therapies. Additionally, electron microscopy may identify pathognomonic Birbeck granules.
Ancillary Tests
Ancillary tests are tailored to specific clinical presentations. Pulmonary function tests are performed for lung involvement, and bronchoalveolar lavage can confirm the diagnosis by identifying a 5% increase in Langerhans cells. Neurological and visual testing is necessary for skull and orbital lesions, while auditory testing is recommended for temporal bone involvement. These evaluations collectively guide accurate diagnosis and inform targeted management strategies.
Treatment / Management
The treatment of eosinophilic granuloma is categorized into nonoperative and operative approaches, depending on the severity, location, and systemic involvement of the disease. These approaches aim to address the symptoms, prevent disease progression, and improve patient outcomes based on the extent and nature of the eosinophilic granuloma.
Nonoperative Treatment
The following nonoperative therapies may be considered in patients with limited disease:
- Observation: Solitary lesions are often self-limiting and may regress spontaneously, especially in skeletally immature or asymptomatic patients. These patients are monitored for changes at regular intervals.
- Immobilization: Painful spinal lesions without or with minimal neurological deficits are managed with immobilization. Bracing can be employed to prevent the progression of kyphosis in affected patients.
- Low-dose irradiation: Radiation therapy, at doses ranging from 6 to 12 Gy delivered in 2 Gy fractions, is used for symptomatic lesions that persist or recur after conservative treatment. Low-dose irradiation is also effective for lesions affecting vital structures or causing mild neurological deficits. Studies suggest lower recurrence rates when surgery is combined with radiation compared to radiation alone.[25] Failures in radiation treatment tend to occur in soft tissue areas.
- Intralesional methylprednisolone: For symptomatic lesions of the spine and extremities, methylprednisolone injections (40 mg to 160 mg) are administered. Corticosteroids are also utilized in pulmonary eosinophilic granuloma when significant pulmonary or constitutional symptoms are present.
- Chemotherapy: Systemic chemotherapy with vinblastine and prednisone or cytarabine (for 12 months) is recommended for high-risk patients with polyostotic disease, central nervous system-risk bone involvement (eg, sphenoid, ethmoid, orbital, or temporal bones), or multisystem involvement.[14] Chemotherapy may also be indicated for recurrent disease, often suggesting multisystem involvement rather than isolated lesions.[23]
- Smoking cessation: Smoking cessation is critical in stabilizing pulmonary eosinophilic granuloma, halting disease progression, and preventing bronchogenic carcinoma. (B3)
Operative Treatment
The following operative therapies may be considered in patients with severe disease:
- Curettage and bone grafting: This method is employed for solitary skull lesions following biopsy or for lesions at risk of impending fracture. Recent literature suggests that isolated calvarial eosinophilic granuloma may be monitored clinically and radiologically without surgery.[26]
- Surgical fixation: Surgical intervention is indicated for severe pain, restricted range of motion, progressive neurological deficits, or spinal instability.[27][25]
- Chest intubation and pleurodesis: Pneumothorax associated with pulmonary eosinophilic granuloma is managed with chest intubation followed by pleurodesis. Recurrent cases may require pleurectomy.
- Lung transplantation: For advanced pulmonary eosinophilic granuloma, lung transplantation is a viable option. (A1)
Differential Diagnosis
The differential diagnoses that should also be considered when evaluating eosinophilic granulomas involving the spine include:
- Neurofibroma
- Schwannoma
- Leukemia
- Lymphomas
- Multiple myeloma
- Osteomyelitis
- Tuberculosis
- Plasmacytoma
- Metastasis
They have similar radiological findings; however, minimal laboratory findings and lack of constitutional symptoms favor eosinophilic granuloma. The biopsy of the lesion confirms the diagnosis.
For lesions involving the skull bone, the differential diagnoses include:
- Otitis media
- Mastoiditis
- Seborrheic dermatitis
- Periorbital cellulitis
For lesions involving the long bones of the extremities, the differential diagnoses include:
- Ewing's sarcoma
- Osteochondroma
- Osteoblastoma
- Osteosarcoma
- Paget's disease
In patients with suspected pulmonary eosinophilic granulomas, differential diagnoses include:
- Tuberous sclerosis
- Lymphangioleiomyomatosis
- Pulmonary histiocytic sarcoma
- Cystic fibrosis
- Emphysema
- Hypersensitivity pneumonitis
- Sarcoidosis
Biopsy and histopathological analysis, staining for CD1a, and electron microscopic findings help differentiate an eosinophilic granuloma from these conditions.[28]
Prognosis
Patients with solitary bone lesions generally have a favorable prognosis, as these lesions may regress spontaneously. However, high-risk groups, including those with multisystem involvement, skeletally mature patients, individuals with multiple bone lesions, or those with skull-base bone involvement (such as the sphenoid, ethmoid, orbital, and temporal bones), face a higher risk of recurrence and complications, leading to a poorer prognosis. Operative treatment often provides more immediate pain relief compared to nonoperative approaches.[29] Despite treatment advances, over 10% of patients succumb to the disease, and some experience reactivations and long-term morbidity.[15]
The prognosis for pulmonary eosinophilic granuloma largely depends on smoking cessation. Patients who quit smoking often see disease stabilization or regression, while those who continue to smoke typically experience disease progression. Poor prognostic factors include extreme age, the presence of large cysts or honeycombing on radiological imaging, multiorgan involvement, and prolonged corticosteroid therapy. These factors contribute to a more severe disease course and worse outcomes.
Complications
The complications associated with eosinophilic granuloma include:
- Musculoskeletal disability or restricted activity
- Growth disturbance
- Pathological fractures
- Hearing and vision impairment
- Neuropsychiatric problems like depression and anxiety
- Postradiation sarcomas and myelitis
- Methylprednisolone injection associated osteomyelitis
- Spontaneous pneumothorax
- Increased risk of Hodgkin and non-Hodgkin lymphoma, myeloproliferative disorders, and bronchogenic carcinoma
- Pulmonary arterial hypertension and cor pulmonale
Consultations
The management of eosinophilic granulomas may require multiple consultations, depending on the organs involved and clinical presentation, including:
- Orthopedist
- Spine neurosurgeon or orthopedist
- Radiologist
- Ophthalmologist
- Pulmonologist
- Otolaryngologist
- Neurologist
- Hematologist-oncologist
Deterrence and Patient Education
Eosinophilic granuloma is a benign bone disorder with a variable clinical course. Asymptomatic solitary lesions may resolve spontaneously without requiring treatment; however, there is a risk of developing additional bone lesions within 6 months to 2 years. Regular follow-up with a complete skeletal survey is essential to monitor for such developments.
Children with multiple bone lesions should undergo a thorough evaluation to rule out systemic involvement, as eosinophilic granuloma can sometimes extend beyond the skeletal system.[30] Additionally, the family members of patients with eosinophilic granuloma have an increased incidence of thyroid disease, suggesting a potential familial association that warrants further investigation.
Enhancing Healthcare Team Outcomes
The management of eosinophilic granuloma requires the collaborative efforts of an interprofessional healthcare team to ensure optimal patient outcomes. The orthopedic surgeon typically leads the care, especially when surgical intervention is necessary, but other specialists are crucial depending on the extent of the disease. Ophthalmologists, pulmonologists, hematologists-oncologists, otolaryngologists, neurosurgeons, and neurologists must be consulted for cases involving multiple organ systems or complex symptoms. Nurses are integral in providing patient education and support, ensuring families are well-informed about the condition and treatment options, and assisting with symptom management. Radiologists are essential in interpreting imaging studies to identify the lesions and help guide the diagnosis, relying on a comprehensive clinical history to determine the appropriate imaging modalities.
In the postoperative phase, pharmacists play a vital role by ensuring the correct analgesics are prescribed for pain management and that antibiotics are appropriately administered to prevent infection. Effective communication and coordination among these healthcare professionals are critical to enhancing patient safety and outcomes. By maintaining open lines of communication, healthcare teams can address any complications early, adjust treatment plans as necessary, and provide a seamless experience for the patient. This coordinated interprofessional approach ensures patient-centered care, maximizes the potential for positive treatment outcomes, and improves overall team performance.
Media
(Click Image to Enlarge)
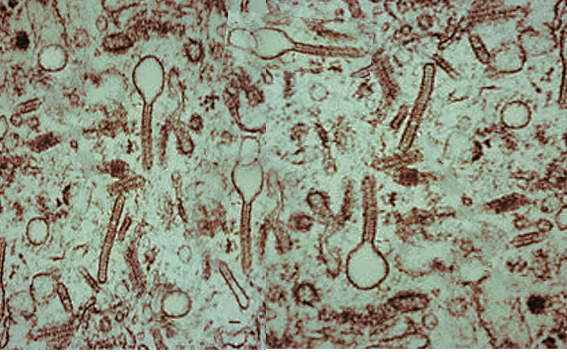
Histiocytosis Microscopy. Electron microscopy further aids in the diagnosis by identifying Birbeck granules within Langerhans cells. These granules are "tennis-racket-shaped" cytoplasmic inclusions with a zipper-like appearance, believed to play a role in antigen storage.
Contributed by S Bhimji, MD
References
Bang WS, Kim KT, Cho DC, Sung JK. Primary eosinophilic granuloma of adult cervical spine presenting as a radiculomyelopathy. Journal of Korean Neurosurgical Society. 2013 Jul:54(1):54-7. doi: 10.3340/jkns.2013.54.1.54. Epub 2013 Jul 31 [PubMed PMID: 24044083]
Level 3 (low-level) evidenceIslinger RB, Kuklo TR, Owens BD, Horan PJ, Choma TJ, Murphey MD, Temple HT. Langerhans' cell histiocytosis in patients older than 21 years. Clinical orthopaedics and related research. 2000 Oct:(379):231-5 [PubMed PMID: 11039811]
Level 2 (mid-level) evidenceCochrane LA, Prince M, Clarke K. Langerhans' cell histiocytosis in the paediatric population: presentation and treatment of head and neck manifestations. The Journal of otolaryngology. 2003 Feb:32(1):33-7 [PubMed PMID: 12779259]
Level 2 (mid-level) evidenceSherwani RK, Akhtar K, Qadri S, Ray PS. Eosinophilic granuloma of the mandible: a diagnostic dilemma. BMJ case reports. 2014 Apr 3:2014():. doi: 10.1136/bcr-2013-200274. Epub 2014 Apr 3 [PubMed PMID: 24700031]
Level 3 (low-level) evidenceBajracharya B , Poudel P , Bajracharya D , Bhattacharyya S , Shakya P . Eosinophilic Granuloma of Mandible: A Diagnostic Challenge. Kathmandu University medical journal (KUMJ). 2018 Apr-Jun:16(62):201-203 [PubMed PMID: 30636766]
Prasad GL, Divya S. Eosinophilic Granuloma of the Cervical Spine in Adults: A Review. World neurosurgery. 2019 May:125():301-311. doi: 10.1016/j.wneu.2019.01.230. Epub 2019 Feb 13 [PubMed PMID: 30771538]
Öğrenci A, Batçık OE, Ekşi MŞ, Koban O. Pandora's box: eosinophilic granuloma at the cerebellopontine angle-should we open it? Child's nervous system : ChNS : official journal of the International Society for Pediatric Neurosurgery. 2016 Aug:32(8):1513-6. doi: 10.1007/s00381-015-2982-1. Epub 2015 Dec 12 [PubMed PMID: 26661575]
Haupt R, Minkov M, Astigarraga I, Schäfer E, Nanduri V, Jubran R, Egeler RM, Janka G, Micic D, Rodriguez-Galindo C, Van Gool S, Visser J, Weitzman S, Donadieu J, Euro Histio Network. Langerhans cell histiocytosis (LCH): guidelines for diagnosis, clinical work-up, and treatment for patients till the age of 18 years. Pediatric blood & cancer. 2013 Feb:60(2):175-84. doi: 10.1002/pbc.24367. Epub 2012 Oct 25 [PubMed PMID: 23109216]
Huang WD, Yang XH, Wu ZP, Huang Q, Xiao JR, Yang MS, Zhou ZH, Yan WJ, Song DW, Liu TL, Jia NY. Langerhans cell histiocytosis of spine: a comparative study of clinical, imaging features, and diagnosis in children, adolescents, and adults. The spine journal : official journal of the North American Spine Society. 2013 Sep:13(9):1108-17. doi: 10.1016/j.spinee.2013.03.013. Epub 2013 Apr 18 [PubMed PMID: 23602327]
Level 2 (mid-level) evidenceLalitha Ch, Manjula M, Srikant K, Goyal S, Tanveer S. Hand schuller christian disease: a rare case report with oral manifestation. Journal of clinical and diagnostic research : JCDR. 2015 Jan:9(1):ZD28-30. doi: 10.7860/JCDR/2015/10985.5481. Epub 2015 Jan 1 [PubMed PMID: 25738095]
Level 3 (low-level) evidenceAl-Mohannadi M, Yakoub R, Soofi ME, Elsabah HM, Thandassery RB. Gastrointestinal: Letterer Siwe disease: An uncommon gastrointestinal presentation. Journal of gastroenterology and hepatology. 2016 Jun:31(6):1070. doi: 10.1111/jgh.13293. Epub [PubMed PMID: 26757250]
Level 3 (low-level) evidenceChadha M, Agarwal A, Agarwal N, Singh MK. Solitary eosinophilic granuloma of the radius. An unusual differential diagnosis. Acta orthopaedica Belgica. 2007 Jun:73(3):413-7 [PubMed PMID: 17715738]
Level 3 (low-level) evidenceMa J, Laird JH, Chau KW, Chelius MR, Lok BH, Yahalom J. Langerhans cell histiocytosis in adults is associated with a high prevalence of hematologic and solid malignancies. Cancer medicine. 2019 Jan:8(1):58-66. doi: 10.1002/cam4.1844. Epub 2018 Dec 30 [PubMed PMID: 30597769]
Kobayashi M, Tojo A. Langerhans cell histiocytosis in adults: Advances in pathophysiology and treatment. Cancer science. 2018 Dec:109(12):3707-3713. doi: 10.1111/cas.13817. Epub 2018 Oct 30 [PubMed PMID: 30281871]
Level 3 (low-level) evidenceAllen CE, Ladisch S, McClain KL. How I treat Langerhans cell histiocytosis. Blood. 2015 Jul 2:126(1):26-35. doi: 10.1182/blood-2014-12-569301. Epub 2015 Mar 31 [PubMed PMID: 25827831]
Gaensler EA, Carrington CB. Open biopsy for chronic diffuse infiltrative lung disease: clinical, roentgenographic, and physiological correlations in 502 patients. The Annals of thoracic surgery. 1980 Nov:30(5):411-26 [PubMed PMID: 7436611]
Watanabe R, Tatsumi K, Hashimoto S, Tamakoshi A, Kuriyama T, Respiratory Failure Research Group of Japan. Clinico-epidemiological features of pulmonary histiocytosis X. Internal medicine (Tokyo, Japan). 2001 Oct:40(10):998-1003 [PubMed PMID: 11688843]
Level 2 (mid-level) evidenceBadalian-Very G, Vergilio JA, Degar BA, MacConaill LE, Brandner B, Calicchio ML, Kuo FC, Ligon AH, Stevenson KE, Kehoe SM, Garraway LA, Hahn WC, Meyerson M, Fleming MD, Rollins BJ. Recurrent BRAF mutations in Langerhans cell histiocytosis. Blood. 2010 Sep 16:116(11):1919-23. doi: 10.1182/blood-2010-04-279083. Epub 2010 Jun 2 [PubMed PMID: 20519626]
Tran G, Huynh TN, Paller AS. Langerhans cell histiocytosis: A neoplastic disorder driven by Ras-ERK pathway mutations. Journal of the American Academy of Dermatology. 2018 Mar:78(3):579-590.e4. doi: 10.1016/j.jaad.2017.09.022. Epub 2017 Oct 26 [PubMed PMID: 29107340]
Kumar N, Sayed S, Vinayak S. Diagnosis of Langerhans cell histiocytosis on fine needle aspiration cytology: a case report and review of the cytology literature. Pathology research international. 2011 Jan 20:2011():439518. doi: 10.4061/2011/439518. Epub 2011 Jan 20 [PubMed PMID: 21331166]
Level 3 (low-level) evidenceRai S, Sridevi HB, Pai RR, Sanyal P. A Case of Multifocal Eosinophilic Granuloma Involving Spine and Pelvis in a Young Adult: A Radiopathological Correlation. Indian journal of medical and paediatric oncology : official journal of Indian Society of Medical & Paediatric Oncology. 2017 Oct-Dec:38(4):555-558. doi: 10.4103/ijmpo.ijmpo_130_16. Epub [PubMed PMID: 29333031]
Level 3 (low-level) evidenceAli-Ridha A, Brownstein S, O'Connor M, Gilberg S, Tang T. Langerhans cell histiocytosis in an 18-month-old child presenting as periorbital cellulitis. Saudi journal of ophthalmology : official journal of the Saudi Ophthalmological Society. 2018 Jan-Mar:32(1):52-55. doi: 10.1016/j.sjopt.2018.02.006. Epub 2018 Feb 16 [PubMed PMID: 29755272]
Xie X, Wang J, Ding Y. Recurrent eosinophilic granuloma involving maxilla and mandible in an adult male: an unusual case report. Australian dental journal. 2021 Mar:66 Suppl 1():S88-S92. doi: 10.1111/adj.12861. Epub 2021 Jul 11 [PubMed PMID: 34043826]
Level 3 (low-level) evidenceAngelini A, Mavrogenis AF, Rimondi E, Rossi G, Ruggieri P. Current concepts for the diagnosis and management of eosinophilic granuloma of bone. Journal of orthopaedics and traumatology : official journal of the Italian Society of Orthopaedics and Traumatology. 2017 Jun:18(2):83-90. doi: 10.1007/s10195-016-0434-7. Epub 2016 Oct 21 [PubMed PMID: 27770337]
Prasad GL, Krishna G, Kale A, Rajeshwari M, Kale SS. Eosinophilic granuloma of the cervical spine in a young adult: A rare case report. Surgical neurology international. 2024:15():198. doi: 10.25259/SNI_262_2024. Epub 2024 Jun 14 [PubMed PMID: 38974539]
Level 3 (low-level) evidenceBenato A, Riva G, Raneri F. Eosinophilic granuloma of the calvarium: is conservative management a valid option? Illustrative case and systematic review of the literature. Child's nervous system : ChNS : official journal of the International Society for Pediatric Neurosurgery. 2023 Aug:39(8):2221-2227. doi: 10.1007/s00381-023-05926-z. Epub 2023 Mar 20 [PubMed PMID: 36939904]
Level 1 (high-level) evidenceNezafati S, Yazdani J, Shahi S, Mehryari M, Hajmohammadi E. Outcome of Surgery as Sole Treatment of Eosinophilic Granuloma of Jaws. Journal of dentistry (Shiraz, Iran). 2019 Sep:20(3):210-214. doi: 10.30476/DENTJODS.2019.44903. Epub [PubMed PMID: 31579697]
Kamal AF, Luthfi APWY. Diagnosis and treatment of Langerhans Cell Histiocytosis with bone lesion in pediatric patient: A case report. Annals of medicine and surgery (2012). 2019 Sep:45():102-109. doi: 10.1016/j.amsu.2019.07.030. Epub 2019 Aug 2 [PubMed PMID: 31452877]
Level 3 (low-level) evidenceZhou Z, Zhang H, Guo C, Yu H, Wang L, Guo Q. Management of eosinophilic granuloma in pediatric patients: surgical intervention and surgery combined with postoperative radiotherapy and/or chemotherapy. Child's nervous system : ChNS : official journal of the International Society for Pediatric Neurosurgery. 2017 Apr:33(4):583-593. doi: 10.1007/s00381-017-3363-8. Epub 2017 Feb 28 [PubMed PMID: 28247113]
Kaul R, Gupta N, Gupta S, Gupta M. Eosinophilic granuloma of skull bone. Journal of cytology. 2009 Oct:26(4):156-7. doi: 10.4103/0970-9371.62188. Epub [PubMed PMID: 21938183]
Level 3 (low-level) evidence