Introduction
Unlike a simple blood transfusion, where a patient receives blood or its components without any blood removal, exchange transfusion involves removing the patient's blood or its constituents and replacing them with donor or blood components. The therapeutic apheresis process removes abnormal cells or substances from the blood associated with specific disease states through either plasma exchange or cytapheresis. Plasmapheresis separates plasma via centrifugation or filtration, whereas cytapheresis involves separating red blood cells, white blood cells, and platelets, typically for subsequent use in another individual. During an exchange transfusion, patients undergo therapeutic cytapheresis to remove abnormal or excess cellular components, followed by replacement through the transfusion of allogenic blood products. Exchange transfusion is commonly used to lower hemoglobin S levels in patients with sickle cell disease and treat unconjugated hyperbilirubinemia or polycythemia in neonates.
Whether performed manually or using an automated machine, exchange transfusion poses risks of errors and complications, necessitating careful implementation by healthcare professionals. Careful monitoring is essential to avoid and manage common adverse events such as hypothermia, fluid overload, hypoglycemia, and hypocalcemia. With advances such as intravenous immune globulin, phototherapy, and Rho(D) immune globulin, exchange transfusion is not a standard clinical procedure in modern medicine. However, healthcare professionals must be well-versed in the indications, techniques, and possible complications for patients who do not respond to standard therapy.
Indications
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Indications
Sickle Cell Disease
Sickle cell disease, an autosomal recessive disorder, affects the production of hemoglobin's β-globin subunits. Red blood cells (RBCs) adopt a rigid sickle shape due to hypothermia, dehydration, or hypoxia, impeding their passage through blood vessels. Common complications include anemia, infections, vaso occlusive crises, and stroke. Exchange transfusion effectively lowers hemoglobin S levels by 30%, enhancing oxygen delivery and reducing morbidity, unlike simple transfusions, which elevate hemoglobin without effectively decreasing hemoglobin S. Indications for exchange transfusion in patients with sickle cell disease encompass acute emergencies such as multi-organ failure, suspected stroke, respiratory compromise, and acute chest syndrome, in addition to regularly scheduled transfusions to prevent stroke, acute chest syndrome, and recurrent painful episodes. Children younger than 10 undergo simple transfusions with iron chelation therapy, transitioning to exchange transfusions at 10 or 30 kg.
Neonatal Polycythemia
Neonatal polycythemia, defined by a hematocrit exceeding 65% in term infants, stems from various factors:
- Chromosomal anomalies such as trisomy 21, 18, and 13
- Delayed clamping of the umbilical cord
- Twin-to-twin transfusion syndrome
- Intrauterine fetal growth restriction
- Precipitous delivery
- Maternal preeclampsia or other hypertensive or vascular disorders
- Chronic maternal hypoxia due to tobacco use; cardiac or pulmonary disorders; certain medications, such as propranolol; or living at high altitudes
- Infants born to mothers with diabetes mellitus
- Infants who are large for gestational age
- Post-term infants
- Congenital adrenal hyperplasia
Symptoms of neonatal polycythemia vary; some infants are asymptomatic, whereas others may present with hypoglycemia, cyanosis, lethargy, poor feeding, nausea, vomiting, or apnea. Severe complications include pulmonary hypertension, seizures, and necrotizing enterocolitis. Initial management involves intravenous fluids for hypoglycemia, with partial exchange transfusion as a therapeutic option to reduce hematocrit. Studies suggest partial exchange transfusion improves cerebral blood flow, cardiac index, and oxygen transport in the short term but does not appear to improve long-term outcomes.[1][2][3][2] Gastrointestinal injury may be increased with partial exchange transfusion.[4][5] Unlike standard exchange transfusion, partial exchange transfusion removes a smaller blood volume and replaces it with normal saline instead of blood products.
Neonatal Hyperbilirubinemia
Nearly all newborns develop hyperbilirubinemia, a total serum or plasma bilirubin greater than 1 mg/dL or 17 µmol/L. Newborns 35 weeks gestation or older who develop severe hyperbilirubinemia or a total serum bilirubin level above 25 mg/dL or 428 µmol/L are at risk of developing bilirubin-induced neurologic disorder. Initial treatment includes phototherapy, intravenous hydration, and intravenous immune globulin for infants with alloimmune hemolytic anemia. Indications for exchange transfusion in the setting of neonatal hyperbilirubinemia include the following:
- Evidence of acute bilirubin encephalopathy: Lethargy, hyper- or hypotonia, poor suck, high-pitched cry, recurrent apnea, opisthotonos, retrocollis, or seizures
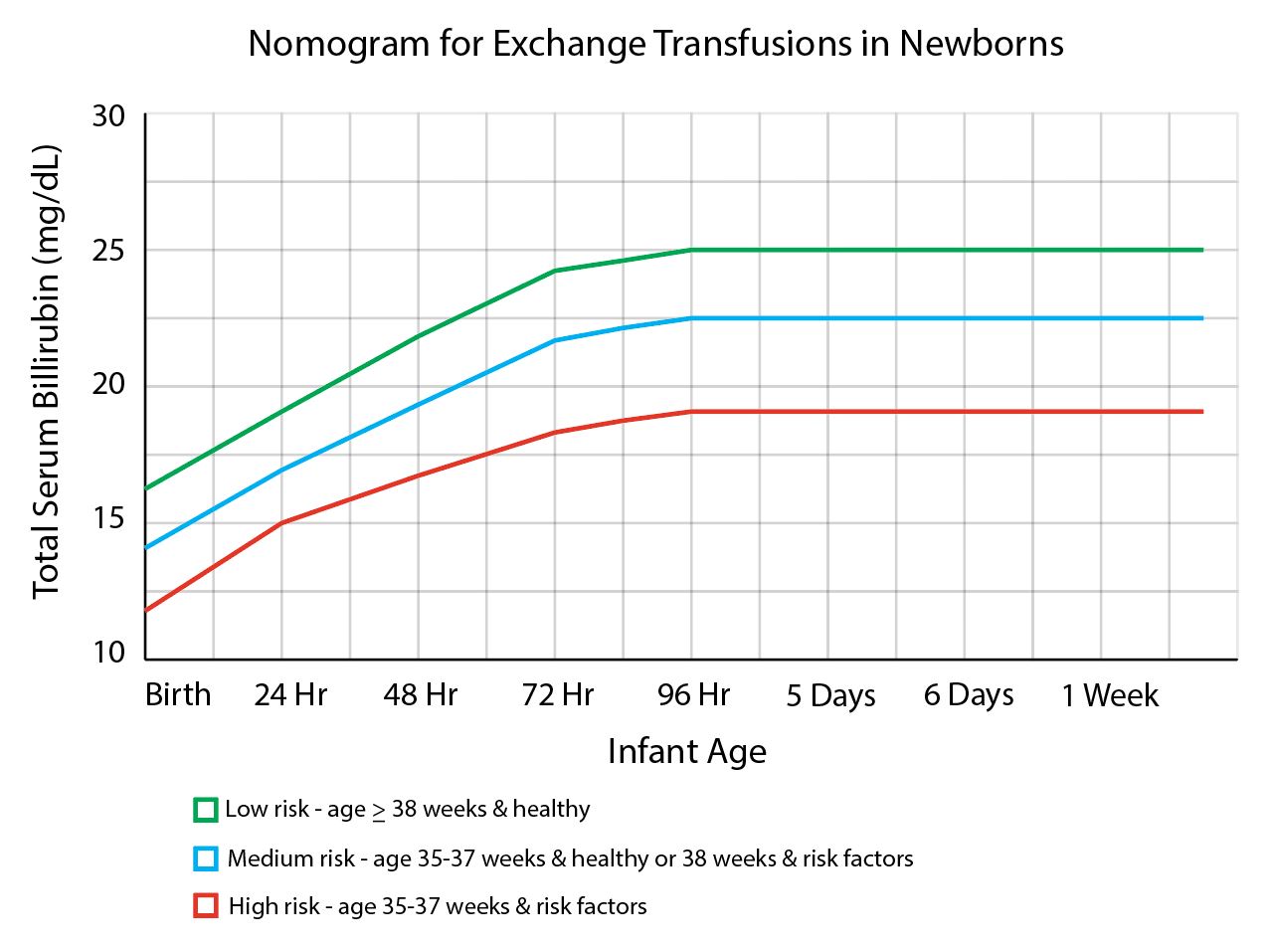
- Total serum bilirubin levels at or above the hour-specific exchange transfusion threshold: Levels vary based on gestational age and neurotoxicity risk factors (see Image. Nomogram for Exchange Transfusions in Newborns)
- Bilirubin/albumin (B/A) ratio at or above the exchange transfusion threshold: These thresholds vary based on gestational age and neurotoxicity risk factors.[6] The B/A ratio should not be used as the sole determining factor to proceed with an exchange transfusion. The B/A ratios serving as thresholds for exchange transfusion are as follows:
- B/A mass ratio ≥8.0 (molar ratio 0.9) for newborns with gestational age 38 weeks or older and no neurotoxicity risk factors
- B/A mass ratio ≥7.2 (molar ratio 0.81) for newborns with gestational age 38 weeks or older and at least 1 neurotoxicity risk factor
- B/A mass ratio ≥7.2 (molar ratio 0.81) for newborns with gestational age 35 through 37 weeks and no neurotoxicity risk factors
- B/A mass ratio ≥6.8 (molar ratio 0.77) for newborns with gestational age 35 through 37 weeks and at least 1 neurotoxicity risk factor
The B/A mass ratio is calculated using the following formula:
B/A mass ratio = Total serum or plasma bilirubin (in mg/dL) ÷ Serum albumin (in g/dL)
Risk factors for neurotoxicity include a gestational age of younger than 38 weeks, hemolytic disorders, hemodynamic instability, sepsis, and hypoalbuminemia.[7]
Gestational Alloimmune Liver Disease
Previously named neonatal hemochromatosis, gestational alloimmune liver disease is caused by the transplacental passage of reactive maternal immunoglobulin G that activates the fetal complement cascade, causing fetal liver injury.[8] Affected neonates present with hepatic failure and hepatic and extrahepatic iron accumulation. Clinical signs include coagulopathy, ascites, and hypoalbuminemia, along with both conjugated and unconjugated hyperbilirubinemia. Neonatal cirrhosis is also a common finding, confirming that the initial insult is during the antenatal period. Unlike other alloimmune conditions, gestational alloimmune liver disease may occur in initial or subsequent pregnancies, suggesting sensitization during gestation rather than solely during delivery. Treatment includes intravenous immune globulin and exchange transfusion.
Glucose-6-Phosphate Dehydrogenase Deficiency
Glucose-6-phosphate dehydrogenase generates nicotinamide-adenine dinucleotide phosphate and protects RBCs from oxidative injury. Patients with a severe deficiency may present with anemia and jaundice in the newborn period. Exchange transfusion may be necessary for severe cases.
Poisoning
Exchange transfusions may help treat massive hemolysis due to arsine or sodium chlorate poisoning, severe methemoglobinemia, severe sulfhemoglobinemia secondary to hydrogen sulfide exposure, or neonatal drug toxicity. Clinicians must generally repeat the process 2 to 3 times.
Babesiosis
Babesiosis, caused by the parasite Babesia microti and transmitted by deer ticks, leads to hemolytic anemia and flu-like symptoms by infecting and destroying RBCs. Standard treatment for uncomplicated cases includes atovaquone and azithromycin. In contrast, a significant parasite burden and end-organ damage may benefit from exchange transfusion to reduce parasite burden, correct anemia, and remove inflammatory mediators.[9] These recommendations are based on case reports and case series.[10][11] In Europe, prompt use of exchange transfusion has shown some improvement in mortality from Babesia divergens infection in immunocompromised individuals.[12]
Contraindications
Contraindications to Exchange Transfusion
- Hemodynamic instability preventing the patient from tolerating the procedure (hypotension is not a contraindication)
Contraindications to Placing an Umbilical Vein or Artery Catheter
- Omphalitis
- Malformation of the cord, as observed in gastroschisis or omphalocele
- Patent urachus
- Necrotizing enterocolitis
- Age greater than approximately 1 to 2 weeks
- Peritonitis
- Vascular compromise to the kidneys, buttocks, or lower limbs
Equipment
Clinicians can perform RBC exchange transfusions manually or by using an automated apheresis machine; manual transfusions are necessary for very small patients. Automated machines are highly effective as they have settings to achieve the desired hemoglobin level. Patients require 2 sets of access points for drawing and returning blood. Further, double-lumen central catheters can be placed in the femoral vein in emergencies.
Neonatal Exchange Transfusion
- Radiant warmer
- 2 Luer Lock umbilical catheters
- 24-gauge catheter if peripheral intravenous access is necessary
- 2 Luer Lock 20-mL syringes
- 2 Luer Lock 5-mL syringes
- Multiple syringe sizes for drawing blood aliquots for the push-pull method
- 2 extension tubing sets
- Blood tubing filter
- 1 empty bleeding bag
- 2 four-way stopcock for the push-pull method
- 2 three-way stopcock for the continuous method
- 1 umbilical cord tape
- 1 ruler
- Packed RBCs reconstituted with plasma or whole blood
- Fluid warmer to warm the blood
- Large volume infusion pump
- Secondary intravenous set
- Blood sampling tubes and lab labels
- Sterile gowns
- Sterile gloves
- Sterile towels
- Surgical masks
- Sterile gauze
- Sterile dressing
- Chlorohexidine swabs
- Cardiac monitor
- Pulse oximetry
- Blood pressure monitor
- Calcium gluconate 10%
- Glucometer
- Sharps container
- RBCs
Please see StatPearls' companion reference, "Umbilical Vein Catheterization," for more information.
Manual Exchange Transfusion for Sickle Cell Disease
Automated exchange transfusion is preferred over manual exchange for patients with sickle cell disease.
- Infusion pump
- Blood warmer
- 500 mL 0.9% sodium chloride (several packs)
- Packed red cells (amount dependent on patient's size and condition, typically 6-8 units)
- Calcium gluconate 10%
- Cardiac monitor
- Sterile gloves
- 2- to 16-gauge cannulae (24 gauge for pediatric patients)
- Chlorohexidine skin preparation
- 4- to 10-mL syringes for saline flushes
- 50-mL syringes for phlebotomy if needed
- 20 mL of normal saline for flush
- Heparinized saline for flush
- Dressings and tape to secure cannulae
- Sterile gauze
- Sharps container
- Scale to assist in the calculation of volume phlebotomized
- Hemoglobin point-of-care test
- RBCs
Automated Exchange Transfusion
- Apheresis machine
- 2- to 16-gauge cannulae (24 gauge for pediatric patients)
- Chlorhexidine skin preparation
- Dressings and tape to secure cannulae
- Sterile gauze
- Calcium gluconate 10%
- Cardiac monitor
- 4- to 10-mL syringes for saline flushes
- 20 mL of normal saline for flush
- Sharps container
- RBCs
- Sterile gloves
Personnel
The personnel required for an exchange transfusion depends on the patient's condition and the procedure type. For neonates and patients undergoing manual transfusions, the team typically includes the primary clinician overseeing the patient's care, the specialist performing the procedure, nurses, blood bank personnel, lab personnel, and pathologists. Patients undergoing an automated procedure also require an apheresis professional and possibly a phlebotomist.
Technique or Treatment
The 2 types of exchange transfusions typically performed in neonates are double-volume exchange transfusion, primarily used to treat hyperbilirubinemia, and partial exchange transfusion, most commonly used to treat polycythemia and other neonatal conditions. During a double-volume exchange transfusion, healthcare professionals replace the baby's total blood volume twice, leaving the intravascular amount the same. A partial exchange transfusion involves slowly removing some of the blood volume and replacing the withdrawn blood with normal saline to help dilute the RBC concentration. Before any procedure, informed consent must be obtained, including a complete discussion of the procedure, risks, benefits, and alternatives.
Neonatal Exchange Transfusion
Neonates typically undergo a double-volume isometric exchange transfusion for hyperbilirubinemia using the 1-catheter push-pull or the 2-catheter isovolumetric (continuous) approach. The required volume is 160 mL/kg, replacing approximately 85% of the neonate's circulating blood. Unstable infants may undergo single-volume exchange using an 80 mL/kg blood volume, replacing approximately 60% of the circulating blood. The neonate should receive irradiated, cytomegalovirus-safe, crossmatched reconstituted blood using washed RBCs and fresh frozen plasma with a hematocrit volume of approximately 40% to 45%. Infants are made nothing per mouth before the procedure, and feeds are withheld for 3 to 24 hours post-procedure, depending on local practice. Neonates who have eaten within 2 hours of the procedure generally have the stomach contents emptied through a nasogastric tube.
Laboratory testing
Before the procedure, the neonate should undergo the following laboratory tests:
- Prothrombin time
- Partial thromboplastin time
- International normalized ratio
- Fibrinogen
- Basic metabolic panel
- Complete blood count
- Bilirubin level (only for patients with hyperbilirubinemia)
- Arterial or venous blood gas
At the halfway point, immediately following the procedure, and 4 hours post-procedure, the following laboratory tests should be performed:
- Basic metabolic panel
- Complete blood count
- Arterial or venous blood gas
- Bilirubin level (only at the 4-hour post-procedure mark for patients with hyperbilirubinemia)
The neonate should undergo continuous cardiopulmonary monitoring throughout the procedure. Continuous exchange is the preferred method and requires 2 healthcare professionals. Donor blood hangs with tubing passing through the blood warmer. One healthcare professional infuses blood or saline through the venous line, whereas the other simultaneously withdraws blood through the arterial line at the same rate. Ideally, for this method, blood is withdrawn from an umbilical arterial catheter and infused into an umbilical venous catheter with a tip in the inferior vena cava or right atrium. Peripheral arterial or venous access may be necessary. A tip in the portal circulation increases the risk of necrotizing enterocolitis. Please see StatPearls' companion references, "Umbilical Vein Catheterization and Umbilical Artery Catheterization," for more information.
A 3-way stopcock connects the umbilical venous line, tubing from the donor blood, and an appropriately sized syringe. A second 3-way stopcock connects the umbilical arterial line, the syringe, and the discard bag. A predetermined aliquot is withdrawn from the arterial line, typically 8% to 10% of the total blood volume or 6 to 8 mL/kg per aliquot. In contrast, donor blood or normal saline infuses through the venous line at the same rate. The withdrawal catheter should be flushed with heparinized saline every 10 to 15 minutes to prevent clotting.
The push-pull method uses a single-lumen umbilical venous catheter. A four-way stopcock connects the tubing from the donor blood, the umbilical venous catheter, the discard bag, and an appropriately sized syringe. A predetermined aliquot, typically 5 mL/kg, is withdrawn from the umbilical venous line over approximately 1.5 minutes, cleared to the discard bag, and clinicians administer an equal aliquot of donor blood or normal saline during a partial exchange transfusion over approximately 1.5 minutes. The clinician performing the procedure should call out the amount removed and infused after each removal and addition, with another clinician documenting the volume.
Partial Exchange Transfusion
During a partial exchange transfusion, clinicians typically remove blood from an umbilical venous catheter and infuse normal saline through a peripheral intravenous catheter using an isovolumetric technique.[13][14] A systematic review and meta analysis revealed that a crystalloid is as effective as a colloid in managing the hematologic state during a partial exchange transfusion, making normal saline an appropriate choice.[14] In general, the exchange volume is 15 to 20 mL/kg. Alternatively, healthcare professionals can calculate the exchange volume using the following formula:
Exchange volume (in mL) = ([observed HCT – desired HCT] × blood volume in mL]) ÷ observed HCT
The desired HCT is 55%, and the estimated blood volume is 80 to 100 mL/kg body weight.
Manual Exchange Transfusion in Sickle Cell Anemia
Manual exchange transfusion is not ideal, as it provides a limited exchange transfusion and is only valuable for emergent situations in patients with sickle cell disease. Automated exchange transfusion is far superior.
Laboratory testing
On the day of the procedure, the patient should undergo the following tests:
- Complete blood count
- Reticulocyte count
- Hemoglobin S percent measurement
- Indirect antiglobulin test
- Calcium and electrolyte measurement
- Liver function tests
- Prothrombin time
- Partial thromboplastin time
- International normalized ratio
- Fibrinogen
Clinicians determine the required blood volume for transfusion based on the patient's weight and HCT. The following is used to calculate the necessary volume for partial exchange transfusion in manual exchange transfusion:
Manual partial exchange volume (mL) = ([dHCT – iHCT] × TBV) ÷ (rpHCT – ([iHCT + dHCT ÷ 2])
In this formula, dHCT is the desired HCT expressed as a percent; iHCT is the initial HCT expressed as a percent; TBV is the estimated total blood volume in mL; and rpHCT is the HCT of the replacement packed RBC with the typical range being 55% to 60%. The volume of each unit of packed RBCs varies depending on the anticoagulant used in the collection bag; most units are approximately 300 to 400 mL. The TBV is generally 60 mL/kg in adult females, 70 mL/kg in adult males, 80 mL/kg in children, and 100 mL/kg in infants.[17]
Decreasing the percentage of hemoglobin S below 30% is ideal, but with a manual exchange transfusion, this is difficult to predict and unlikely to occur. During a manual exchange transfusion, normal saline is infused in addition to RBCs to avoid significant changes in hemoglobin and viscosity. Manual exchange transfusions may require multiple sessions to achieve the desired outcome.
Two peripheral intravenous access sites are necessary. Patients may require a 500 mL normal saline bolus if dehydrated before the transfusion. Subsequently, they receive 500 mL of normal saline, followed by the removal of 500 mL of blood into an empty bleeding bag placed below the level of the patient's bed. If the blood flow is inadequate, the nurse may use a syringe to draw blood from the peripheral site. The patient then receives 1 unit of RBCs, followed by removing the same volume of blood. For each unit of blood transfused, healthcare professionals remove an equivalent unit of blood. Hemoglobin levels should be reassessed midway through the procedure. At the end of the transfusion, clinicians should check the hemoglobin S percentage, calcium, and hemoglobin levels.
Automated Exchange Transfusion
Clinicians program automated exchange machines to achieve the desired post-procedure HCT and hemoglobin S levels. During an automated exchange, patients have either 2 large peripheral intravenous catheters or 1 dual-lumen central venous catheter that allows the simultaneous withdrawal and return of blood. The catheter connects to sterile tubing in an apheresis machine, which separates the patient's blood into components. The machine removes a portion of the patient's RBCs and replaces them with donor RBCs.
Complications
General Complications of Transfusions
- Infection
- Febrile reactions
- Acute and delayed hemolytic reactions
- Anaphylactic reactions
- Transfusion-associated acute lung injury [13]
- Transfusion-associated circulatory overload
- Hypocalcemia or hypoglycemia due to citrate in the preservative
- Graft-versus-host disease
- Iron overload
Exchange Transfusion
- Arrhythmia and cardiac arrest if blood is not warmed
- Necrotizing enterocolitis
- Hyperkalemia
- Hypocalcemia
- Acidosis
- Hypertension
- Hypotension
- Hypothermia
- Hemolysis if the blood is too warm
- Umbilical vein perforation
- Bloody stools, abdominal distension, and emesis
- Thrombosis due to umbilical vein catheter placement [4][14]
Clinical Significance
Exchange transfusion holds paramount clinical significance, particularly in neonatology and hematology. In neonates at risk of developing bilirubin-induced neurologic disorder, exchange transfusion stands as the most efficient and immediate method for swiftly removing bilirubin and averting potential neurological complications. In addition, exchange transfusion plays a pivotal role in patients with sickle cell disease by substantially reducing hemoglobin S levels to less than 30% more effectively than a simple transfusion, mitigating vaso occlusion, and enhancing tissue oxygenation. Moreover, isovolumetric partial exchange transfusion emerges as a vital intervention for neonates with polycythemia, as it effectively lowers HCT levels without inducing hypovolemia. Evidence suggests that partial exchange transfusion yields beneficial short-term effects on perfusion metrics, underscoring its clinical significance in optimizing patient outcomes.
Enhancing Healthcare Team Outcomes
Exchange transfusion is a critical intervention in various medical contexts, particularly in neonatal care and hematology. This procedure serves as a life-saving measure by swiftly removing bilirubin in neonates at risk of developing bilirubin-induced neurologic disorder, a condition that can lead to severe neurological complications. In patients with sickle cell disease, exchange transfusion effectively lowers hemoglobin S levels, mitigating vaso-occlusion and enhancing tissue oxygenation in multi-organ failure, a stroke, or acute chest syndrome. Routinely scheduled exchange transfusions also serve to prevent stroke, acute chest syndrome, and recurrent painful episodes. In addition, exchange transfusion addresses neonatal polycythemia by reducing HCT levels without causing hypovolemia, offering short-term benefits in perfusion metrics. Although exchange transfusions are not as common in modern medicine due to medical advancements, they remain an essential skill for healthcare professionals when standard therapies are ineffective. Furthermore, exchange transfusions are helpful in diverse clinical scenarios, ranging from treating conditions such as glucose-6-phosphate dehydrogenase deficiency to poisoning, underscoring their broad clinical significance and potential life-saving benefits.
Exchange transfusion necessitates a multidisciplinary approach. Healthcare professionals must possess advanced skills in identifying suitable indications for exchange transfusion, choosing the appropriate technique, and effectively managing potential complications. Effective communication and collaborative strategies are paramount to ensure thorough patient preparation and vigilant monitoring during the procedure. Developing effective strategies, including establishing protocols, guidelines, and risk mitigation protocols, is crucial. Interprofessional communication is vital to keep all team members updated, foster collaboration, and address emerging concerns. Care coordination among healthcare professionals ensures smooth transitions and consistent care, enhancing patient-centered care, reducing morbidity, improving patient safety, and optimizing team performance throughout the exchange transfusion process.
Media
(Click Image to Enlarge)
References
Kallimath A, Kolkur K, Malshe N, Klimek J, Suryawanshi P. Hemodynamics in neonates with polycythemia before and after partial exchange transfusion: an observational study. Frontiers in pediatrics. 2023:11():1296184. doi: 10.3389/fped.2023.1296184. Epub 2024 Jan 5 [PubMed PMID: 38250589]
Level 2 (mid-level) evidenceErgenekon E, Hirfanoglu IM, Turan O, Beken S, Gucuyener K, Atalay Y. Partial exchange transfusion results in increased cerebral oxygenation and faster peripheral microcirculation in newborns with polycythemia. Acta paediatrica (Oslo, Norway : 1992). 2011 Nov:100(11):1432-6. doi: 10.1111/j.1651-2227.2011.02358.x. Epub 2011 Jun 11 [PubMed PMID: 21595746]
Rosenkrantz TS, Oh W. Cerebral blood flow velocity in infants with polycythemia and hyperviscosity: effects of partial exchange transfusion with Plasmanate. The Journal of pediatrics. 1982 Jul:101(1):94-8 [PubMed PMID: 7086628]
Dempsey EM, Barrington K. Short and long term outcomes following partial exchange transfusion in the polycythaemic newborn: a systematic review. Archives of disease in childhood. Fetal and neonatal edition. 2006 Jan:91(1):F2-6 [PubMed PMID: 16174666]
Level 1 (high-level) evidenceOzek E, Soll R, Schimmel MS. Partial exchange transfusion to prevent neurodevelopmental disability in infants with polycythemia. The Cochrane database of systematic reviews. 2010 Jan 20:(1):CD005089. doi: 10.1002/14651858.CD005089.pub2. Epub 2010 Jan 20 [PubMed PMID: 20091569]
Level 1 (high-level) evidenceKemper AR, Newman TB, Slaughter JL, Maisels MJ, Watchko JF, Downs SM, Grout RW, Bundy DG, Stark AR, Bogen DL, Holmes AV, Feldman-Winter LB, Bhutani VK, Brown SR, Maradiaga Panayotti GM, Okechukwu K, Rappo PD, Russell TL. Clinical Practice Guideline Revision: Management of Hyperbilirubinemia in the Newborn Infant 35 or More Weeks of Gestation. Pediatrics. 2022 Sep 1:150(3):. pii: e2022058859. doi: 10.1542/peds.2022-058859. Epub [PubMed PMID: 35927462]
Level 1 (high-level) evidenceIskander I, Gamaleldin R, El Houchi S, El Shenawy A, Seoud I, El Gharbawi N, Abou-Youssef H, Aravkin A, Wennberg RP. Serum bilirubin and bilirubin/albumin ratio as predictors of bilirubin encephalopathy. Pediatrics. 2014 Nov:134(5):e1330-9. doi: 10.1542/peds.2013-1764. Epub 2014 Oct 20 [PubMed PMID: 25332491]
Pan X, Kelly S, Melin-Aldana H, Malladi P, Whitington PF. Novel mechanism of fetal hepatocyte injury in congenital alloimmune hepatitis involves the terminal complement cascade. Hepatology (Baltimore, Md.). 2010 Jun:51(6):2061-8. doi: 10.1002/hep.23581. Epub [PubMed PMID: 20512994]
Saifee NH, Krause PJ, Wu Y. Apheresis for babesiosis: Therapeutic parasite reduction or removal of harmful toxins or both? Journal of clinical apheresis. 2016 Oct:31(5):454-8. doi: 10.1002/jca.21429. Epub 2015 Oct 20 [PubMed PMID: 26481763]
Krause PJ, Auwaerter PG, Bannuru RR, Branda JA, Falck-Ytter YT, Lantos PM, Lavergne V, Meissner HC, Osani MC, Rips JG, Sood SK, Vannier E, Vaysbrot EE, Wormser GP. Clinical Practice Guidelines by the Infectious Diseases Society of America (IDSA): 2020 Guideline on Diagnosis and Management of Babesiosis. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2021 Jan 27:72(2):185-189. doi: 10.1093/cid/ciab050. Epub [PubMed PMID: 33501959]
Level 1 (high-level) evidencePadmanabhan A, Connelly-Smith L, Aqui N, Balogun RA, Klingel R, Meyer E, Pham HP, Schneiderman J, Witt V, Wu Y, Zantek ND, Dunbar NM, Schwartz GEJ. Guidelines on the Use of Therapeutic Apheresis in Clinical Practice - Evidence-Based Approach from the Writing Committee of the American Society for Apheresis: The Eighth Special Issue. Journal of clinical apheresis. 2019 Jun:34(3):171-354. doi: 10.1002/jca.21705. Epub [PubMed PMID: 31180581]
Hildebrandt A, Gray JS, Hunfeld KP. Human babesiosis in Europe: what clinicians need to know. Infection. 2013 Dec:41(6):1057-72. doi: 10.1007/s15010-013-0526-8. Epub 2013 Oct 9 [PubMed PMID: 24104943]
Rzayev T, Kersin SG, Memisoglu A, Ozdemir H, Bilgen H, Ozek E. Acute lung injury after exchange transfusion in two newborns with Glucose-6-phosphate dehydrogenase deficiency. Transfusion and apheresis science : official journal of the World Apheresis Association : official journal of the European Society for Haemapheresis. 2021 Aug:60(4):103133. doi: 10.1016/j.transci.2021.103133. Epub 2021 Apr 6 [PubMed PMID: 33846094]
Black VD, Rumack CM, Lubchenco LO, Koops BL. Gastrointestinal injury in polycythemic term infants. Pediatrics. 1985 Aug:76(2):225-31 [PubMed PMID: 4022696]