Introduction
The conjunctiva is a transparent, thin mucous membrane covering the sclera. This structure extends from the limbus, or the perimeter of the cornea, and covers both the sclera, where it is termed the "bulbar conjunctiva," and the posterior eyelid surfaces, where it is called the "palpebral conjunctiva." Keratoconjunctivitis is an inflammatory condition that affects both the superficial cornea and the conjunctiva. The term is derived from the words "keratitis," which refers to superficial corneal inflammation, and "conjunctivitis," which denotes conjunctival involvement. This ocular condition can arise from an expansive list of etiologies, including viral, bacterial, autoimmune, toxic, or allergic causes.[1]
Keratoconjunctivitis often results in significant discomfort and potential vision impairment. Viruses, particularly adenoviruses, are among the most common culprits, followed by bacterial and fungal pathogens in certain cases. Allergic keratoconjunctivitis, often associated with environmental allergens, as well as dry eye-related forms linked to tear film dysfunction, also contributes significantly to the condition. Autoimmune diseases such as Sjögren syndrome can further complicate the clinical spectrum of this disease by disrupting the ocular surface.[2]
Epidemiologically, keratoconjunctivitis is widespread, with varying prevalence depending on the subtype and geographic location. Viral keratoconjunctivitis, for instance, tends to occur in outbreaks, especially in crowded environments like schools or workplaces. Conversely, dry eye-related keratoconjunctivitis predominantly affects older populations, particularly postmenopausal women, due to hormonal changes affecting tear production.
Patients with keratoconjunctivitis often present with a constellation of symptoms, including redness, photophobia, blurred vision, excessive tearing or dryness, and a gritty or foreign body sensation in the eye. In more severe cases, patients may report sharp pain, mucopurulent discharge, or difficulty keeping the eye open due to irritation. On examination, clinicians may note conjunctival hyperemia, corneal epithelial defects, lid swelling, or papillary reactions on the tarsal conjunctiva. These signs, combined with the patient's history, aid in differentiating the various subtypes of keratoconjunctivitis.[3]
A thorough investigation is essential to establish a definitive diagnosis. Evaluation typically includes a detailed clinical history, slit-lamp biomicroscopy, and, where indicated, laboratory tests such as viral cultures, polymerase chain reaction (PCR) testing for adenoviruses, or conjunctival scrapings for cytology and bacterial cultures. Tear film assessments, including the Schirmer test and tear break-up time (TBUT), are valuable in diagnosing dry eye-associated keratoconjunctivitis. Advanced imaging techniques, such as anterior segment optical coherence tomography (AS-OCT) and confocal microscopy, can provide further insights into the structural integrity of the cornea and conjunctiva.[4]
Management of keratoconjunctivitis highly depends on the underlying etiology. Viral keratoconjunctivitis typically requires supportive care, including artificial tears and cold compresses, as antiviral medications are rarely indicated. In contrast, bacterial keratoconjunctivitis demands prompt antibiotic therapy to prevent complications, such as corneal ulcers. Allergic keratoconjunctivitis often responds well to antihistamines, mast cell stabilizers, and allergen avoidance strategies. For dry eye-related cases, artificial tears, punctal plugs, and, in severe cases, immunomodulatory agents like cyclosporine can provide significant relief. Corticosteroids may be cautiously used in autoimmune-related keratoconjunctivitis, but their prolonged use must be carefully monitored due to the risk of cataracts and glaucoma.[5]
Looking to the future, advancements in the understanding of ocular surface inflammation and tear film dynamics are paving the way for more targeted therapies. Emerging treatments, such as biologic agents targeting specific inflammatory pathways, alongside gene therapies, hold promise in addressing refractory cases. Additionally, innovations in diagnostic tools, including point-of-care tests for tear biomarkers and artificial intelligence-driven imaging technologies, are expected to enhance early detection and personalized management of keratoconjunctivitis.[6]
Overall, keratoconjunctivitis is a complex and multifaceted condition requiring a nuanced approach to diagnosis and management. By integrating current evidence-based practices with emerging therapeutic and diagnostic advancements, healthcare providers can significantly improve outcomes for individuals affected by this debilitating condition. This activity will focus on more well-known entities causing combined keratoconjunctivitis. Specifically, this activity will discuss the entities known as epidemic keratoconjunctivitis (EKC), vernal keratoconjunctivitis (VKC), superior limbic keratoconjunctivitis (SLK), and keratoconjunctivitis sicca.[7]
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
The list of causes of keratoconjunctivitis is extensive. A general classification scheme is helpful, dividing cases into infectious and noninfectious categories. Within the infectious category, viral and bacterial etiologies are the most common, with viral infections accounting for the majority of suspected cases with an infectious cause. Noninfectious keratoconjunctivitis can be allergic, toxic, or immune-mediated. Additionally, keratoconjunctivitis can be classified as unilateral or bilateral. In terms of duration, the condition may be classified as hyperacute if the onset is less than a week, acute if the onset is within 3 to 4 weeks, or chronic if it lasts longer than 4 weeks.[8]
EKC is a viral form of keratoconjunctivitis caused by adenovirus, a ubiquitous, nonenveloped, double-stranded DNA virus. Human adenovirus primarily targets the eyes and the respiratory, gastrointestinal, and genitourinary tracts. Research has identified human adenovirus types 8, 19, 37, and 54 as the strains responsible for EKC outbreaks.[9]
VKC is a severe allergic conjunctivitis with an unclear etiology. A strong allergic component is believed to be involved, with the mast cell pathway, mediated by immunoglobulin E (IgE), playing a key role in symptomatology. The condition is thought to result from a combination of immune, genetic, and environmental factors, including air pollutants and allergens.[10]
SLK is a chronic inflammatory condition with an unknown etiology. One leading theory suggests that constant friction between the superior bulbar and tarsal conjunctival interfaces leads to chronic microtrauma, triggering the development of the condition.[11]
Keratoconjunctivitis sicca, commonly known as dry eye syndrome (DES) or disease (DED), has several identified underlying etiologies, often associated with autoimmune systemic conditions. The final common pathway involves abnormalities in the precorneal tear film and lymphocyte-mediated inflammatory changes in the ocular surface, including the conjunctiva and cornea.
Keratoconjunctivitis has many causes, reflecting its complex and multifactorial nature. The etiology of this disorder can be further expanded, as explained below.
Infectious Causes
Adenoviruses are responsible for epidemic, highly contagious keratoconjunctivitis. Herpes simplex virus (HSV) can cause recurrent keratitis with dendritic ulcerations. Varicella-zoster virus (VZV) may lead to herpes zoster ophthalmicus, which involves both the cornea and conjunctiva. Cytomegalovirus (CMV) can cause keratoconjunctivitis in immunosuppressed individuals, such as those with HIV/AIDS or a history of organ transplantation.[12]
Common bacterial pathogens that give rise to acute bacterial keratoconjunctivitis include Staphylococcus aureus and Streptococcus pneumoniae. More severe infections by Pseudomonas aeruginosa are prevalent in contact lens wearers and can rapidly lead to corneal perforation if untreated. Neonatal keratoconjunctivitis, often caused by Neisseria gonorrhoeae or Chlamydia trachomatis, presents within the first few days of life.
Fungal keratoconjunctivitis, though rare, occurs after trauma with vegetative matter or during agricultural work. Candida and Fusarium species are common culprits. These infections can be sight-threatening if not treated promptly.[13]
Parasitic infections of the cornea and conjunctiva can also occur. Acanthamoeba and Pythium keratitis are particularly dangerous, often associated with improper contact lens hygiene or exposure to contaminated water sources.[14][15]
Allergic Causes
Allergic conjunctivitis arises from IgE-mediated hypersensitivity to environmental allergens. Seasonal allergic conjunctivitis (SAC) occurs during specific months, while perennial allergic conjunctivitis (PAC) persists year-round. Severe forms, such as VKC, often affect children and young adults living in warm regions. These conditions are characterized by thick, ropy discharge and giant papillae. Atopic keratoconjunctivitis (AKC) is chronic and can lead to corneal scarring and vision loss if untreated.
Autoimmune and Systemic Diseases
Autoimmune diseases like Sjögren syndrome can lead to aqueous tear deficiency, which can cause severe dry eye and keratoconjunctivitis sicca. Rheumatoid arthritis can give rise to secondary keratoconjunctivitis through immune-mediated inflammation of the lacrimal glands. Severe hypersensitivity reactions, such as Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis, can produce extensive ocular surface damage and chronic keratoconjunctivitis.[16]
Dry Eye Disease
This condition is one of the leading causes commonly linked to meibomian gland dysfunction (MGD), which disrupts the lipid layer of the tear film and increases evaporation. Other contributors include reduced aqueous production from lacrimal gland dysfunction, which is often exacerbated by systemic conditions like diabetes and thyroid disease, as well as long-term computer use, and leads to a reduced blink rate.[17][18]
Environmental and Chemical Factors
Chronic exposure to smoke, air pollution, or excessive screen time can disrupt the tear film and induce keratoconjunctivitis. Chemical injuries from industrial accidents and exposure to strong acids, alkalis, or irritants, such as household cleaning agents, can result in acute or chronic inflammation of the cornea and conjunctiva.[19]
Mechanical and Trauma-Related Causes
Direct trauma to the ocular surface that may be caused by foreign bodies, corneal abrasions, or surgical instruments can disrupt the corneal epithelium and predispose the eye to secondary keratoconjunctivitis. Prolonged use of contact lenses, particularly if they are ill-fitting or improperly maintained, can lead to hypoxia and mechanical irritation, increasing the risk of infection and inflammation.[20] Trichiasis, a condition characterized by misdirected eyelashes rubbing the ocular surface, increases the risk of recurrent keratoconjunctivitis.
Iatrogenic Causes
Extended use of topical medications, especially those containing preservatives like benzalkonium chloride, can cause toxic keratoconjunctivitis. Postsurgical keratoconjunctivitis may occur after procedures like cataract extraction, laser-assisted in situ keratomileusis (LASIK), and corneal transplantation due to inflammation or exposure to surgical disinfectants.[21]
Nutritional Deficiencies
Vitamin A deficiency remains a significant cause of keratoconjunctivitis in developing countries. This disorder produces xerophthalmia, with conjunctival and corneal dryness progressing to keratinization, corneal ulceration, and, potentially, blindness. Deficiencies in essential fatty acids or antioxidants can also disrupt tear production and ocular surface integrity.[22]
Neurological Disorders
Neurologic conditions like facial nerve palsy can lead to exposure keratopathy, which results from incomplete eyelid closure and produces chronic keratoconjunctivitis. Reduced corneal sensitivity in trigeminal nerve dysfunction may impair the protective blink reflex, increasing susceptibility to ocular surface damage.[23]
Congenital or Genetic Factors
Rare genetic conditions, such as aniridia, keratoconus, and congenital ichthyosis, can predispose individuals to keratoconjunctivitis through structural and functional abnormalities of the cornea and conjunctiva.[24] Consequently, people with these conditions may require specialized eye health care.
Occupational Hazards
Workers in industries requiring prolonged exposure to heat, ultraviolet radiation, or chemicals are at a higher risk of developing occupational keratoconjunctivitis, which may become chronic without proper protective measures. These individuals often need regular eye examinations to monitor for early signs of ocular damage.
Psychogenic or Behavioral Factors
Patients who compulsively rub their eyes, often seen in keratoconus, have a higher risk of developing recurrent keratoconjunctivitis.[25] The constant physical trauma caused by such behaviors can compromise the eye’s natural defenses, promoting recurrent corneal and conjunctival inflammation.
Rare and Miscellaneous Causes
Rare infectious agents, such as Chlamydia in trachoma-endemic regions, can cause chronic keratoconjunctivitis, leading to blindness if untreated. Radiation keratopathy, seen in patients undergoing ocular radiation therapy, can cause significant damage to the cornea and conjunctiva.[26]
Understanding the diverse etiologies of keratoconjunctivitis is crucial for accurate diagnosis and tailored management. Different causes necessitate specific treatment approaches to prevent complications and preserve vision.
Epidemiology
The prevalence of keratoconjunctivitis varies widely according to the underlying cause, which is also influenced by age, environment, and genetics. Some estimates suggest as many as 6 million people annually in the United States present to a healthcare provider with signs and symptoms of conjunctivitis or keratoconjunctivitis.
Allergic causes account for most conjunctivitis and keratoconjunctivitis cases, affecting up to 40% of the population. VKC primarily occurs in school-aged children, with the peak incidence between 4 and 7 years old. However, the condition can present as early as 5 months or as late as the 3rd or 4th decade of life. Young male individuals living in dry, hot climates, particularly in West Africa, the Mediterranean basin, the Middle East, India, and South America, are at the highest risk. The condition is less common in North America and Western Europe.[27][28]
For infections, the pediatric population is most susceptible to bacterial keratoconjunctivitis and may account for approximately 50% of cases. However, viruses are, by far, the most common microbial pathogens, regardless of age, with estimates reaching up to 75%. Adenovirus is responsible for up to 90% of viral infections. Other viral agents include enterovirus, HSV, and herpes zoster.[29]
Keratoconjunctivitis sicca has become a growing public health concern, affecting up to 17% of women and 11% of men in the United States. Some studies suggest that these figures may be underestimated, as many patients self-treat without consulting a healthcare provider. The prevalence is higher in individuals with autoimmune conditions, such as Sjögren syndrome, rheumatoid arthritis, systemic lupus erythematosus (SLE), sarcoidosis, and scleroderma. The condition is also more common in dry climates.[30]
SLK is a relatively rare condition with a female-to-male predominance of approximately 3:1. The age of presentation ranges from 20 to 60. A racial predilection has not been documented. SLK is associated with thyroid dysfunction and keratoconjunctivitis sicca.
Pathophysiology
The complex pathophysiology of keratoconjunctivitis involves the interplay between various inflammatory, immune, and mechanical factors, disrupting the integrity of the corneal and conjunctival tissues. The mechanisms vary depending on the underlying etiology, but they generally involve a cascade of events that lead to ocular surface damage, discomfort, and potential vision impairment.[31]
Inflammatory Cascade and Immune Response
The immune system's activation primarily mediates infectious and noninfectious keratoconjunctivitis. In viral keratoconjunctivitis, pathogens such as adenoviruses and HSV infect epithelial cells, triggering the release of pro-inflammatory cytokines like interleukin-1 (IL-1) and tumor necrosis factor α (TNF-α). These cytokine releases recruit neutrophils and lymphocytes, leading to tissue damage and increased vascular permeability, manifesting as conjunctival hyperemia and corneal edema.[32]
In bacterial keratoconjunctivitis, bacterial components such as lipopolysaccharides (LPS) activate toll-like receptors (TLRs) on corneal and conjunctival epithelial cells. Downstream signaling pathways are then triggered, producing chemokines that recruit polymorphonuclear leukocytes (PMNs) to the infection site. Polymorphonuclear leukocytes contribute to tissue inflammation, ulceration, and discharge.[33]
Allergic keratoconjunctivitis involves a type I hypersensitivity reaction. Allergen exposure causes the release of histamines, leukotrienes, and prostaglandins from mast cells. This event leads to vasodilation, increased capillary permeability, and intense itching from activating sensory nerves. Chronic allergic forms, such as VKC and AKC, involve both types I and IV hypersensitivity, resulting in persistent inflammation and fibrosis.
Tear Film Dysfunction
The tear film plays a crucial role in maintaining ocular surface homeostasis. In DED, the tear film’s aqueous, lipid, and mucin layers are disrupted. Aqueous deficiency, often due to lacrimal gland dysfunction in autoimmune conditions like Sjögren syndrome, reduces tear volume and increases tear osmolarity. This hyperosmolar environment causes epithelial cell damage, goblet cell loss, and further inflammation by releasing matrix metalloproteinases (MMPs). MGD disrupts the tear film's lipid layer, increasing tear evaporation and leading to ocular surface desiccation. This desiccation stimulates the innate immune system, perpetuating chronic inflammation and epithelial damage.[34]
Corneal Epithelial Disruption
Regardless of the etiology, keratoconjunctivitis often damages the corneal epithelium. Viral infections, such as HSV, cause cytopathic effects, forming dendritic ulcers. Bacterial keratoconjunctivitis can result in epithelial breakdown due to the release of bacterial toxins and proteases. Mechanical factors, such as abrasion from foreign bodies or poorly fitting contact lenses, can also disrupt the epithelial barrier, making the eye more susceptible to secondary infections. Chronic inflammation from allergic or autoimmune conditions further weakens the epithelial tight junctions, exacerbating tissue damage and increasing the risk of corneal scarring.[35]
Conjunctival Involvement and Goblet Cell Loss
The conjunctiva, an integral part of the ocular surface, is significantly involved in keratoconjunctivitis. Inflammatory mediators induce vascular dilation and immune cell infiltration, resulting in the conjunctiva's hallmark redness and swelling. Prolonged inflammation, as seen in conditions like SJS, can lead to goblet cell loss, reducing mucin production and further destabilizing the tear film. In allergic keratoconjunctivitis, the conjunctiva develops giant papillae due to chronic mechanical and inflammatory stimulation, leading to further discomfort and visual disturbances.[36]
Role of Oxidative Stress
Oxidative stress plays a pivotal role in the pathophysiology of keratoconjunctivitis, particularly in DED and autoimmune conditions. Reactive oxygen species generated during chronic inflammation can damage epithelial cells, extracellular matrix proteins, and cellular organelles. This oxidative damage contributes to epithelial apoptosis, subepithelial fibrosis, and chronic ocular surface inflammation.[37]
Fibrosis and Scarring
Chronic or severe keratoconjunctivitis can activate fibroblasts and remodel the extracellular matrix. In autoimmune conditions like ocular cicatricial pemphigoid (OCP), persistent inflammation induces conjunctival fibrosis, resulting in symblepharon formation and fornical shortening. Similarly, prolonged inflammation in AKC can produce corneal scarring and vision loss.[38]
Neuropathic Component
Neuropathic pain may also contribute to the symptomatology of keratoconjunctivitis. Damage to the corneal nerve endings from inflammation, infection, or trauma results in hypersensitivity, photophobia, and persistent pain, even after the resolution of inflammation. In some cases, nerve regeneration is incomplete, leading to chronic symptoms and impaired healing.[39]
Chronicity and Recurrence
Certain forms of keratoconjunctivitis, such as viral and allergic types, tend to recur due to persistent triggers or incomplete resolution of the underlying cause. Recurrences are often associated with exacerbated inflammation, which causes progressive damage to the ocular surface and complicates treatment.
The pathophysiology of keratoconjunctivitis involves a dynamic interplay of immune activation, tear film instability, epithelial damage, and oxidative stress, which collectively disrupt the ocular surface's homeostasis. These mechanisms, compounded by chronic inflammation, nerve involvement, and potential scarring, highlight the complexity of the condition and the importance of tailored therapeutic approaches to prevent long-term visual impairment and improve patient outcomes.
Treatment strategies should address the underlying causes, such as restoring tear film stability, reducing inflammation, and promoting epithelial healing. Emerging therapies, including biologics, autologous serum, and regenerative treatments like amniotic membrane transplantation (AMT) and stem cell therapy (SCT), hold promise in managing refractory cases. A comprehensive approach combining conventional and advanced treatments, guided by a thorough understanding of the disease’s pathophysiology, is crucial to preserving ocular surface integrity and enhancing the quality of life for affected individuals.
Histopathology
Histopathological analysis provides critical insights into the structural and cellular changes occurring within the ocular surface in keratoconjunctivitis cases. This condition involves a variety of inflammatory, infectious, and immune-mediated mechanisms that disrupt the normal anatomy and function of the conjunctiva, cornea, and tear film. Microscopic examination is essential for diagnosing certain keratoconjunctivitis subtypes, such as DED, allergic conjunctivitis, infectious keratoconjunctivitis, and autoimmune-related forms like VKC and AKC.[40]
Key histopathological features include the following:
- Epithelial changes: Loss of corneal and conjunctival epithelial integrity is a hallmark of keratoconjunctivitis. Inflammatory subtypes may exhibit hyperplasia, dyskeratosis, or erosion. Goblet cell density is often reduced in dry eye-related keratoconjunctivitis, leading to compromised mucin production and tear film instability. Epithelial metaplasia with keratinization may occur in severe or chronic cases.[41]
- Stromal involvement: Subepithelial stromal infiltration by inflammatory cells, ie, lymphocytes, plasma cells, and macrophages, is commonly observed. Edema and neovascularization may develop in chronic or severe inflammatory keratoconjunctivitis. Collagen degeneration and fibrosis in the corneal stroma can result in scarring, which impairs transparency and visual acuity.[42]
- Inflammatory infiltrate: Acute keratoconjunctivitis often presents with neutrophilic infiltration. Chronic cases tend to involve lymphocytic and plasmacytic infiltration, indicating prolonged immune activation. Mast cell degranulation is a key feature in allergic subtypes, especially in VKC and AKC.
- Basement membrane abnormalities: Thickening or fragmentation of the epithelial basement membrane is often seen in chronic keratoconjunctivitis. In autoimmune conditions like SJS, basement membrane disruption may lead to conjunctivalization of the cornea.[43]
- Tear film and mucin layer changes: The tear film may show a reduced lipid or mucin layer, affecting tear stability. Deficient mucin production, secondary to goblet cell loss, is evident in conditions like Sjögren syndrome-associated keratoconjunctivitis.[44]
- Fibrosis and scarring: Long-standing inflammation can lead to conjunctival fibrosis, which may manifest as symblepharon or adhesions between the palpebral and bulbar conjunctivae. Corneal scarring due to stromal remodeling can result in permanent visual impairment.[45]
- Nerve involvement: Inflammatory damage to corneal nerves may reduce nerve density, as seen in neurotrophic keratopathy. Loss of corneal sensation, also known as hypoesthesia, is associated with decreased epithelial healing and increased risk of recurrent epithelial defects.[46]
Specific histopathological patterns are associated with various subtypes of keratoconjunctivitis. In DED, histopathological changes include goblet cell loss, epithelial thinning, and subclinical inflammation characterized by lymphocyte infiltration. VKC is marked by eosinophil infiltration, giant papillae, and mast cell degranulation within the conjunctival tissue.
Bacterial keratoconjunctivitis typically presents with dense neutrophilic infiltration and areas of stromal necrosis. Herpetic keratoconjunctivitis is identified by the presence of multinucleated giant cells and intranuclear inclusion bodies. In chlamydial keratoconjunctivitis, also known as trachoma, follicular conjunctivitis is observed along with lymphoid follicle hyperplasia and subepithelial fibrosis. SJS displays extensive epithelial necrosis, full-thickness conjunctival sloughing, and disruption of the basement membrane.[47]
Histopathological evaluation remains critical to understanding the diverse presentations of keratoconjunctivitis. This modality aids in identifying specific patterns of epithelial, stromal, and inflammatory changes, providing diagnostic clarity and guiding therapeutic interventions.
Advanced techniques, such as immunohistochemistry, allow for the identification of specific immune markers, cytokines, and cellular components involved in the disease process. These insights can help tailor treatments based on underlying pathophysiological mechanisms, such as anti-inflammatory agents, immunomodulators, and surgical interventions. By combining histopathological findings with clinical evaluation, a comprehensive approach to managing keratoconjunctivitis can be achieved, ultimately improving patient outcomes and preserving vision.[48]
Toxicokinetics
Chemical and toxic exposures are frequent causes of keratoconjunctivitis. Examples of substances that can cause corneal and conjunctival inflammation include the following:
- Preservatives in eye drops: Benzalkonium chloride has been shown to induce oxidative stress, inflammation, and apoptosis in corneal epithelial cells, aggravating keratoconjunctivitis.
- Chemicals and pollutants: Prolonged exposure to airborne pollutants, such as sulfur dioxide and particulate matter, may cause chronic irritation, leading to inflammatory changes in the conjunctiva and cornea.
- Medications: Topical anesthetics, antibiotics, and antiglaucoma drugs may exhibit dose-dependent toxic effects, including delayed epithelial healing and stromal edema.
- Contact lens solutions: Improper use of lens-cleaning agents containing hydrogen peroxide or other disinfectants can lead to chemical-induced keratoconjunctivitis.[49]
Key toxicokinetic considerations include the following:
- Absorption: The ocular surface can absorb chemical agents, especially through the conjunctiva and corneal epithelium. Lipophilic compounds penetrate the cornea more efficiently, whereas hydrophilic agents primarily accumulate in the conjunctiva. Prolonged or repeated exposure to irritants, such as eyedrop preservatives like benzalkonium chloride, can disrupt the epithelial barrier and increase the absorption of harmful substances.[50]
- Distribution: Once absorbed, toxic substances may diffuse into the stroma, aqueous humor, or even deeper structures like the ciliary body and retina. Topical agents may concentrate in tear film or conjunctival tissue, leading to localized toxic effects, such as epithelial damage or inflammation. Systemic toxins, including chemotherapeutic agents and heavy metals, may enter the bloodstream and reach the ocular surface, especially when the blood-tear or blood-aqueous barrier is compromised.[51]
- Metabolism: Ocular tissues possess limited enzymatic activity for detoxifying xenobiotics. For example, cytochrome P450 enzymes are present in the cornea and ciliary body but at lower levels than in the liver. Reactive metabolites generated during detoxification may exacerbate oxidative stress, contributing to epithelial damage and inflammation seen in keratoconjunctivitis.[52]
- Excretion: The primary route of excretion for toxic substances from the ocular surface is via tear drainage into the nasolacrimal duct. Some compounds may bind to proteins within the stroma, leading to prolonged retention and delayed clearance, potentially increasing the risk of chronic toxicity.[53]
Clinical Implications and Management
Careful evaluation of patient history, including the use of topical agents and environmental exposure, is crucial in identifying potential toxic causes of keratoconjunctivitis. Preservative-free formulations and proper medication dosing should be considered to reduce the risk of ocular toxicity. Lubricants, antioxidants, and anti-inflammatory agents can help mitigate toxic damage and promote epithelial healing.
Understanding the toxicokinetics of substances involved in keratoconjunctivitis helps clinicians identify and manage potential toxic contributors to the disease. Tailoring therapeutic regimens to minimize toxic exposures while addressing the underlying pathophysiological processes is essential to optimize treatment outcomes and prevent long-term ocular surface damage.[54]
History and Physical
Typical Presentations
Frequent symptoms of keratoconjunctivitis, regardless of etiology, include eye discomfort or irritation, pruritus, light sensitivity, minor but often intermittent blurring of vision, and epiphora. Common signs include conjunctival injection, conjunctival chemosis, and eye discharge. Examiners should query all patients about contact lens wear, any possible exposures, such as from sick contacts, chemical splashes, use of current eye drops or face creams, and recent activities that may have resulted in foreign body retention or corneal abrasion. Additionally, a time course regarding their symptoms and any relieving or exacerbating factors is necessary. Systemic conditions, including autoimmune disorders, atopy, and thyroid disease, should be discerned during the historical investigation.
Viral keratoconjunctivitis typically begins with unilateral involvement that later spreads to the opposite eye, resulting in asymmetric findings. Patients commonly report ocular discomfort or itching, accompanied by pronounced light sensitivity. Conjunctival injection and chemosis are frequently observed, along with a primarily watery discharge. The palpebral conjunctiva often exhibits a follicular reaction, and petechial hemorrhages or pseudomembrane formation may occur. Lymphadenopathy and a history of an upper respiratory illness are present in up to 50% of cases. Contact with affected individuals is common but not always necessary for transmission. Symptoms can persist for 7 to 21 days, and patients may remain infectious for 10 to 14 days.
VKC presents with bilateral symptoms, including pruritus, a foreign body sensation, photophobia, mucous discharge, epiphora, and conjunctival injection. Symptom severity varies widely, with severe cases sometimes impairing daily activities. A characteristic feature is early morning mucoid discharge, often causing the eyelids to stick together. The upper palpebral conjunctiva typically exhibits a papillary reaction caused by epithelial hyperplasia with a fibrovascular core. The conjunctivae may demonstrate a cobblestone appearance when they develop numerous large papillae.
Papillae can also arise in the limbal region, and those topped with chalky-white excrescences are referred to as "Horner-Trantas spots." Corneal signs include punctate epithelial erosions and keratitis. Micro-erosions can progress to macro-erosions, known as shield ulcers. Although termed “vernal,” approximately 1/4 of cases persist year-round, and up to 60% recur during the winter months.[55]
Keratoconjunctivitis sicca typically presents with chronic but often intermittent symptoms, which are usually bilateral but may be asymmetric. Common complaints include burning, stinging, foreign body sensation, photophobia, and pruritus. Patients may also report eye fatigue, a sense of heavy eyelids, epiphora, watery discharge, and blurred vision. Activities that reduce the natural blink reflex—such as reading, computer use, watching television, or driving—frequently exacerbate intermittent visual blurring. On examination, findings may include conjunctival injection, inspissated meibomian glands, eyelid margin telangiectasias, erythema, a reduced tear lake, rapid TBUT, punctate epithelial erosions, and diminished tear production.[56]
SLK displays bilateral burning, irritation, and foreign body sensation. Symptoms are typically chronic with gradual clearing but may have periods of remission. Asymmetric involvement is not uncommon. Characteristic examination findings include watery discharge and epiphora, superior palpebral and bulbar conjunctival injection, and chemosis. A conjunctival papillary reaction and redundant conjunctiva or conjunctivochalasis may be present. Punctate epithelial erosions involving the upper 3rd of the cornea are often present. In chronic or severe disease, conjunctival overgrowth or pannus may be present on the cornea.
A thorough history and physical examination are essential for diagnosing and managing keratoconjunctivitis. This multifactorial condition encompasses many etiologies, including infectious, allergic, autoimmune, and environmental causes, which present with overlapping yet distinct clinical features.
History-Taking
Presenting symptoms that should be elicited during history-taking include the following:
- Ocular discomfort: Patients may report a foreign body sensation or a feeling of burning and grittiness, particularly in DED cases.
- Redness: This manifestation is common in most forms of keratoconjunctivitis, indicating conjunctival hyperemia.
- Tearing: Reflex tearing may occur in response to irritation or dryness.
- Itching: Predominant in allergic keratoconjunctivitis, ie, VKC and AKC.
- Photophobia: Suggests significant corneal involvement, such as in cases of epithelial defects or keratitis.
- Blurring of vision: This symptom results from tear film instability, epithelial irregularities, or stromal scarring.
- Discharge: Watery discharge is common in viral cases. Bacterial keratoconjunctivitis is often associated with mucopurulent discharge.[57]
Acute, severe presentations may indicate infectious or toxic etiologies. Chronic, progressive symptoms usually suggest DED, autoimmune conditions, or AKC.[58]
The following may be reported upon investigation of the medical history:
- Associated allergic conditions: Asthma, rhinitis, or eczema in patients with allergic keratoconjunctivitis
- Underlying autoimmune disorders: Sjögren syndrome, rheumatoid arthritis, or SLE in immune-mediated keratoconjunctivitis
- Ocular history: Prior ocular surgeries, contact lens use, or history of recurrent infections
- Medication history: Use of topical medications, systemic drugs, or preservatives that may contribute to ocular toxicity [59]
Environmental and lifestyle factors may include exposure to irritants, such as smoke, dust, and pollutants. Prolonged screen time or reduced blink rate also contributes to evaporative dry eye.[60]
Physical Examination Findings
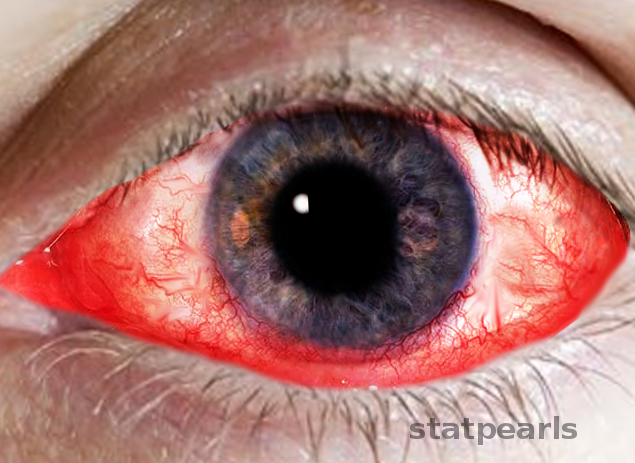
External inspection may reveal eyelid abnormalities, such as blepharitis, entropion, or lagophthalmos, which can disrupt tear distribution. Swelling, redness, or crusting along the eyelid margins may also be evident (see Image Keratoconjunctivitis).[61]
Conjunctival findings may include the following:
- Injection: Diffuse redness may be observed in viral or bacterial infections. Redness is often sectoral in episcleritis.
- Papillae: Small, raised lesions on the palpebral conjunctivae are common in allergic keratoconjunctivitis.
- Follicles: Dome-shaped lymphoid aggregates are often seen in viral or chlamydial infections.
- Conjunctival scarring: This complication may be evident in chronic inflammatory or autoimmune diseases like SJS or OCP.
Examination of the cornea may reveal the following:
- Punctate epithelial erosions (PEE): Fine epithelial defects that indicate dry eye or toxic keratopathy.
- Filaments: Mucus strands attached to the cornea are characteristic of severe dry eye.
- Ulceration: Corneal damage seen in infectious keratoconjunctivitis caused by bacterial, fungal, or herpetic infections.
- Neovascularization: The presence of new blood vessels in the cornea, typically resulting from chronic hypoxia or severe inflammation.
- Scarring or opacities: Permanent corneal changes that impair visual acuity, often observed in advanced or recurrent disease.[62]
Special tests may reveal additional features. On tear film assessment, TBUT is reduced in cases of tear film instability and tear meniscus height is diminished in aqueous-deficient dry eye. The Schirmer test, which measures tear production, should demonstrate reduced values, indicating aqueous deficiency.[63] Limbal and episcleral findings on slit-lamp biomicroscopy include inflammatory cell infiltration or pigmentation, which may occur in chronic conditions.[64] Staining with fluorescein or rose bengal helps detect epithelial defects, evaluate tear film break-up, and assess ocular surface integrity.[65]
A detailed history and comprehensive physical examination provide the foundation for diagnosing keratoconjunctivitis and differentiating between its various subtypes. Recognizing the characteristic constellations of symptoms and signs facilitates prompt identification and accurate management, helping to prevent complications and preserve ocular health.
Evaluation
Irrespective of the underlying etiology, evaluation should include an assessment of best-corrected visual acuity, pupillary reactions, extraocular motility, and confrontational visual fields. Intraocular pressure (IOP) should also be measured. External examination should note any preauricular lymph node swelling or periorbital erythema or edema. An eyelid examination is essential to check for hyperpigmentation, erythema, edema, rashes (eg, vesicles), or discharge.
Discharge should be categorized as purulent, mucopurulent, mucoid, or watery. Purulent discharge can reform as soon as it is cleared from the eye. Mucoid discharge adheres to the eyelashes and appears sticky. Watery discharge is clear and copious. If purulent discharge is found, a culture should be sent for analysis. Eversion of the eyelids is necessary to examine the palpebral conjunctiva, especially to evaluate for the presence of follicles, papillae, or pseudomembranes.
The bulbar and forniceal conjunctiva should be examined for injection and chemosis, as well as the geographic pattern of injection. Fluorescein staining and subsequent examination with either a Wood lamp slit-lamp microscope or direct ophthalmoscope, utilizing a cobalt blue filter, can help evaluate the presence and pattern of corneal abrasions, microtrauma, or epithelial defects. If a herpetic infection is suspected, particular attention should be given to identifying dendritic or pseudodendritic lesions. More specific testing can be considered, depending on the suspected underlying etiology.[66]
Schirmer testing can evaluate tear production in suspected cases of keratoconjunctivitis sicca. Corneal sensation testing is an option in cases of suspected herpetic viral infection, which can cause decreased corneal sensitivity. Tear osmolarity has been increasing in popularity and shows elevated tear film osmolarity in patients with keratoconjunctivitis sicca. Rapid sequence adenoviral testing is available in cases of suspected EKC, though this condition is most commonly a clinical diagnosis. If keratoconjunctivitis sicca is suspected, the provider may consider initial testing for Sjogren syndrome by obtaining levels of the antibodies SS-A (anti-Ro) and SS-B (anti-La).
The evaluation of keratoconjunctivitis involves a combination of clinical examination, laboratory investigations, and specialized diagnostic tests to identify the underlying etiology and assess the severity of the condition. Accurate evaluation is crucial for effective management, particularly in distinguishing between infectious, allergic, autoimmune, and toxic subtypes. Both national and international guidelines, such as those from the Dry Eye Workshop (DEWS) and the American Academy of Ophthalmology (AAO), emphasize a comprehensive approach to evaluation.
Clinical and Diagnostic Tests
Clinical and diagnostic tests include tear film assessment, ocular surface staining, and the measurement of inflammatory markers. Tear film assessment involves evaluating the stability and production of tears. The TBUT test measures tear film stability. A TBUT of less than 10 seconds indicates tear film instability, commonly seen in DES. The Schirmer test assesses tear production. A result of less than 5 mm in 5 minutes suggests severe aqueous deficiency. Tear osmolarity may be measured using devices such as TearLab. Values greater than 308 mOsm/L or an intereye difference exceeding 8 mOsm/L indicate DES.[67]
Ocular surface staining is performed using various dyes. Fluorescein staining highlights corneal epithelial defects and punctate erosions, while rose bengal and lissamine green staining detect areas of epithelial damage and mucin deficiency, particularly in conjunctival tissue.
Inflammatory markers such as matrix metalloproteinase-9 (MMP-9) may also be measured. MMP-9 levels greater than 40 ng/mL are associated with ocular surface inflammation. This test may be conducted using point-of-care devices like InflammaDry.[68]
Laboratory Tests for Specific Etiologies
Specific laboratory tests, including microbiological studies, allergy testing, autoimmune workup, and cytology and biopsy, can confirm some cases of keratoconjunctivitis. Microbiological studies include conjunctival or corneal swabs for bacterial, fungal, or viral cultures in suspected infectious keratoconjunctivitis. PCR may also be used to detect microbial DNA, such as from HSV, adenovirus, and Chlamydia.[69]
Allergy testing involves measuring serum IgE levels, which are elevated in allergic keratoconjunctivitis, and conducting a skin prick test to identify specific allergens causing hypersensitivity.[70] Autoimmune workup includes testing for antinuclear antibodies (ANA) and rheumatoid factor (RF), which are useful in diagnosing conditions like Sjögren syndrome, rheumatoid arthritis, and SLE. Anti-SSA/Ro and Anti-SSB/La antibodies are specific for Sjögren syndrome.[71]
Conjunctival impression cytology evaluates goblet cell density and epithelial metaplasia. Conjunctival biopsy is performed in suspected cases of OCP or SJS.[72]
Imaging Studies
Imaging studies may be used in certain cases to gain more detailed insight into the ocular surface and its structures. AS-OCT provides cross-sectional imaging of the cornea and tear meniscus. This modality is particularly useful in assessing corneal thickness and epithelial integrity.[73] Meibography evaluates the structure and function of meibomian glands, which may be altered in evaporative dry eye.[74] In vivo confocal microscopy (IVCM) allows for detailed evaluation of corneal nerves and inflammatory cell infiltration. This technique is particularly helpful in assessing conditions like neurotrophic keratopathy or infectious keratitis.[75]
Guidelines-Based Recommendations
The International Dry Eye Workshop (DEWS II) recommends using a combination of symptom questionnaires, such as the Ocular Surface Disease Index (OSDI) or Dry Eye Questionnaire (DEQ-5), alongside objective tests like TBUT, osmolarity measurements, and ocular surface staining to diagnose and classify DED. The American Academy of Ophthalmology suggests tailored diagnostic approaches depending on clinical presentation, with a focus on microbiological studies for infectious cases and autoimmune workups when systemic involvement is suspected.
A comprehensive evaluation of keratoconjunctivitis integrates clinical findings, specialized diagnostic tests, and laboratory investigations. By adhering to established guidelines and tailoring the diagnostic approach to the suspected etiology, clinicians can optimize treatment strategies and improve patient outcomes.[76]
Treatment / Management
Addressing keratoconjunctivitis involves a thorough evaluation to identify its root cause. Management largely depends on the underlying etiology.
Epidemic Keratoconjunctivitis
Viral infection in EKC cases is extremely contagious, necessitating a strong focus on preventing transmission to others. Spread occurs through direct contact with contaminated fingers, household objects, or medical instruments. Diligent handwashing and thorough cleaning of household and medical office equipment are essential to minimize the risk of transmission.
The incubation period is estimated at 5 to 12 days, with individuals remaining contagious for 10 to 14 days. Patients and caregivers should be made aware of this extended recovery period to address concerns about persistent symptoms and reduce unnecessary follow-up visits to healthcare providers.[77] Although no definitive treatment exists, symptomatic relief may be achieved with artificial tears, cool compresses, and, in some cases, topical antihistamines. In vitro studies have demonstrated the potential of topical ganciclovir and cidofovir as treatment options.[78]
Vernal Keratoconjunctivitis
VKC often requires an interprofessional approach and should include a referral to ophthalmology. Treatment should aim to balance symptom relief with minimizing medication or treatment side effects. First-line options include lubricating treatments for the ocular surface, such as preservative-free artificial tears, gels, and ointments. While these therapies have virtually no side effects, they are often insufficient on their own. Additional measures, such as cool compresses and lid scrubs, may help remove potential allergens.
Topical antihistamines may provide adequate symptom relief in mild cases. For moderate cases, topical mast cell stabilizers, such as cromolyn sodium, nedocromil sodium, and lodoxamide, are often added to antihistamines and can effectively control symptoms. Mast cell stabilizers require a loading period to achieve therapeutic effects. When a seasonal variation in symptoms is noted, initiating mast cell stabilizers before the onset of symptoms can help prevent recurrence. Combination antihistamine and mast cell stabilizer formulations, including olopatadine, azelastine, and ketotifen, are also available.
Systemic antihistamines may be considered. However, evidence supporting their use in keratoconjunctivitis is limited, and their side effect profiles should be carefully evaluated before initiation. Topical nonsteroidal agents can be trialed, but they are often less effective. Topical corticosteroids are typically the most effective treatment, particularly when started at a high initial dose followed by rapid tapering. Although topical corticosteroids have a low systemic side effect profile, careful monitoring is imperative to detect ocular side effects, such as elevated IOP. For severe or persistent cases, long-term immunomodulation with steroid-sparing agents like cyclosporine or tacrolimus can be beneficial.[79][80][81](A1)
Keratoconjunctivitis Sicca
Initial management is typically conservative and includes preservative-free artificial tears, gels, and ointments. Warm compresses and lid scrubs help increase oil production and release from the meibomian glands, which stabilizes the tear film. Oral flaxseed and fish oil supplements are effective in alleviating symptoms and reducing dependence on topical treatments.
Procedural treatments, such as inserting punctal plugs to prevent lacrimal drainage of the tear film, are available. In some cases, more permanent punctal treatment through cautery may be performed. Prescription options like topical cyclosporine A, tacrolimus, and lifitegrast act as steroid-sparing anti-inflammatory agents. While topical steroids may also be effective, they should be used in short courses, and IOP monitoring is mandatory.
Oral doxycycline at low doses has been shown to provide benefits through its unique anti-inflammatory properties. More recently, topical autologous serum drops have gained popularity. These serum drops contain anti-inflammatory factors that inhibit mediators of the dry eye inflammatory cascade, although they can be prohibitively expensive.
A combination of these treatment options is often necessary to achieve adequate symptomatic relief. Patients may require a tailored approach, with adjustments made based on the severity of symptoms and response to initial treatments.
Superior Limbic Keratoconjunctivitis
First-line treatment is conservative, involving preservative-free artificial tears, gels, and ointments. Other topical treatments, such as mast-cell stabilizers, antihistamines, vitamin A, and nonsteroidal anti-inflammatory drugs, are used with varying degrees of success. Punctal occlusion techniques can also help increase ocular surface lubrication.
Topical autologous serum eye drops have shown effectiveness, although long-term treatment is often required, and the high cost of the medication can be a barrier. Chemocautery of the affected conjunctiva using silver nitrate often provides symptomatic relief for 1 to 2 months but typically requires retreatment. Surgical conjunctival resection of the affected or redundant conjunctiva is highly effective. Alternatively, liquid nitrogen cryotherapy may be used to treat the affected conjunctiva and generally provides long-term symptomatic relief, though repeat treatments may be necessary.[82][83][84](B2)
Differential Diagnosis
Patients with keratoconjunctivitis typically present with a red, irritated eye. Therefore, the differential diagnosis should include the following conditions:
- Conjunctivitis (bacterial, fungal, viral, chemical, exposure, toxic) [85]
- Episcleritis or scleritis
- Uveitis or iritis
- Endophthalmitis [86]
- Corneal abrasion or ulcer
- Corneal foreign body
- Acute angle closure glaucoma
- Subconjunctival hemorrhage
Urgent conditions to consider from the above list include acute angle-closure glaucoma, endophthalmitis, corneal ulcer, corneal foreign body, and uveitis or iritis. Concern for any of these conditions should prompt referral to ophthalmology. Other signs and symptoms that might trigger an ophthalmology referral include the following:
- History of foreign body retention or trauma to the eye
- Elevated IOP
- An asymmetric or nonreactive pupil
- Marked visual acuity reduction
- Extensive or debilitating pain, including an inability to open the eye or keep the eye open
- Corneal opacity
- Copious or hyperacute and progressive purulent discharge
Keratoconjunctivitis presents with symptoms such as redness, tearing, discomfort, and blurred vision, which can overlap with a variety of other ocular surface disorders. Accurate diagnosis requires careful differentiation from other conditions to ensure appropriate treatment. Below is a discussion of diseases and conditions that may be mistaken for keratoconjunctivitis and must be ruled out.
Allergic Conjunctivitis
Allergic conjunctivitis shares similarities with allergic keratoconjunctivitis, such as redness, itching, and tearing. However, this condition is distinguished by the absence of significant corneal involvement, alongside the greater prominence of conjunctival papillae. A seasonal association may also be present.
Blepharitis
Blepharitis presents with symptoms of ocular irritation, redness, and dryness, overlapping with some features of keratoconjunctivitis. The distinguishing characteristics of this condition include inflammation of the eyelid margins, MGD, and crusting at the base of the eyelashes. Significant conjunctival or corneal staining is typically not observed in blepharitis.
Infectious Conjunctivitis
Bacterial conjunctivitis shares features with keratoconjunctivitis, such as redness, mucopurulent discharge, and conjunctival injection. However, this condition is distinguished by its acute onset, the absence of punctate corneal erosions, and a significantly purulent discharge. Viral conjunctivitis also presents with redness, watery discharge, and irritation. This disease is differentiated by a follicular reaction, preauricular lymphadenopathy, and its typically self-limiting course.[87]
Superior Limbic Keratoconjunctivitis
SLK overlaps with keratoconjunctivitis in terms of redness, irritation, and punctate corneal staining. SLK is distinguished by its localization to the superior conjunctiva and cornea and its frequent association with thyroid dysfunction.[88]
Neurotrophic Keratopathy
Neurotrophic keratopathy shares features with keratoconjunctivitis, such as corneal epithelial defects and reduced vision. However, this pathology is distinguished by decreased corneal sensation, delayed epithelial healing, and a lack of significant inflammation.
Dry Eye Disease
DED presents with ocular discomfort, dryness, and punctate epithelial erosions. DED is distinguished by tear film instability, reduced Schirmer test results, and abnormal tear osmolarity. This condition may coexist with keratoconjunctivitis.
Herpetic Keratitis
This disease shares corneal epithelial lesions and photophobia with keratoconjunctivitis. However, herpetic keratitis is distinguished by dendritic or geographic ulcers, reduced corneal sensation, and a history of recurrence.
Contact Lens-Associated Keratitis
Contact lens-associated keratitis presents with redness, irritation, and corneal staining. A history of contact lens use, localized corneal infiltrates, and potential microbial involvement differentiates this condition from keratoconjunctivitis.[89]
Ocular Cicatricial Pemphigoid
This pathology shares chronic conjunctival inflammation and the potential for scarring with keratoconjunctivitis. However, the distinguishing features of OCP include progressive conjunctival fibrosis, symblepharon formation, and systemic autoimmune involvement.[90]
Stevens-Johnson Syndrome
SJS presents with severe ocular surface damage and conjunctival inflammation. Differentiating features include a history of systemic drug reactions, widespread mucocutaneous involvement, and conjunctival sloughing.[91]
Episcleritis and Scleritis
Episcleritis and scleritis both present with ocular redness and discomfort. Episcleritis is typically mild, sectoral, and self-limiting, while scleritis is associated with severe pain, deeper redness, and possible systemic autoimmune disease.[92]
Pterygium
Pterygium shares redness and irritation with keratoconjunctivitis, especially in chronic cases. However, this condition is distinguished by a triangular fibrovascular growth extending onto the cornea, often associated with UV exposure.[93]
Uveitis
Uveitis presents with redness, photophobia, and blurred vision. Distinguishing features include anterior chamber inflammation, the presence of keratic precipitates, and posterior synechiae.[94]
Differentiating keratoconjunctivitis from other ocular surface disorders requires a detailed history, thorough physical examination, and appropriate diagnostic tests. Specific clinical features must be recognized, and adjunctive tests such as tear film analysis, corneal staining, and microbiological studies must be used for accurate diagnosis and effective management.
Pertinent Studies and Ongoing Trials
Robust clinical studies and ongoing trials exploring therapeutic strategies, mechanisms, and outcomes have yielded evidence-based recommendations for managing keratoconjunctivitis. These investigations aim to optimize the treatment of the ocular surface and address underlying inflammatory, infectious, or autoimmune components.
Key Pertinent Studies
- DEWS Reports: The DEWS I (2007) and DEWS II (2017) reports provided a comprehensive framework for diagnosing and managing DED, a significant subtype of keratoconjunctivitis. These studies highlighted the role of inflammation and tear film instability in the pathogenesis of dry eye, which led to the development of targeted therapies such as cyclosporine and lifitegrast. Additionally, these reports standardized diagnostic criteria and treatment protocols for dry eye-related keratoconjunctivitis.[95]
- Lifitegrast Clinical Trials (OPUS-1, OPUS-2, OPUS-3): These clinical trials investigated the efficacy and safety of lifitegrast, an antagonist of lymphocyte function-associated antigen 1 (LFA-1), in reducing DED signs and symptoms. Significant improvements in ocular surface staining and patient-reported symptoms were observed, supporting its approval for the treatment of moderate-to-severe dry eye.[96]
- Cyclosporine trials (RESTASIS and Ikervis): These trials evaluated the efficacy of cyclosporine A and demonstrated its ability to reduce inflammation and improve tear production in patients with aqueous-deficient dry eye. Long-term benefits included reduced corneal staining and increased tear production, establishing its role in managing chronic dry eye conditions.[97]
- Studies on AMT: This research explored the use of AMT in severe ocular surface diseases, including persistent epithelial defects and chemical burns. Findings demonstrated that AMT promotes epithelial healing, reduces inflammation, and prevents scarring in refractory cases of keratoconjunctivitis, making it a valuable option for severe conditions.[98]
- ω3 fatty acid supplementation (DES Trial): Studies assessing ω3 supplementation investigated its role in reducing inflammation and improving dry eye symptoms. While early studies suggested potential benefits, the DREAM study (2018) provided mixed results, highlighting the need for further investigation into the effectiveness of ω3 in managing DES.[99]
Ongoing Clinical Trials
- Biologics for severe keratoconjunctivitis: Ongoing trials evaluate the efficacy of biologics such as dupilumab, an IL-4/IL-13 inhibitor, for treating allergic keratoconjunctivitis. These studies aim to determine whether biologics can reduce inflammatory markers and improve ocular surface integrity.[100]
- Novel ρ kinase inhibitors: Trials investigating ρ kinase inhibitors are focused on their ability to promote corneal endothelial healing and reduce inflammation. If successful, these inhibitors could provide an innovative approach to managing refractory keratoconjunctivitis.[101]
- Regenerative therapies: Research into stem cell-derived therapies and exosome-based treatments explores their potential for ocular surface reconstruction. Expected outcomes include enhanced epithelial repair and reduced scarring, particularly in severe cases of keratoconjunctivitis.[102]
- Advanced tear film analysis techniques: Efforts to develop imaging-based diagnostic tools aim to assess real-time tear film dynamics and osmolarity. These advanced techniques could improve the accuracy of diagnosing tear film abnormalities in keratoconjunctivitis and guide more precise therapeutic interventions.[103]
Keratoconjunctivitis management has witnessed significant advancements due to extensive research and ongoing trials. These studies have led to the development of new diagnostic approaches, ensuring better management and improved outcomes. Continued research is essential to further refine treatment strategies and address unmet clinical needs.
Treatment Planning
Treatment planning for keratoconjunctivitis involves a personalized approach to addressing the underlying cause, relieving symptoms, and preventing complications such as corneal scarring and vision loss. Dryness, infection, allergies, autoimmune disorders, or exposure to environmental irritants may cause this condition. A comprehensive treatment plan focuses on restoring the ocular surface's homeostasis and reducing inflammation. The key steps in treatment planning are explained below.
- Comprehensive assessment: Clinical evaluation of keratoconjunctivitis involves assessing ocular symptoms, including redness, discomfort, photophobia, and foreign body sensation. Diagnostic tests play a crucial role in identifying underlying causes. The TBUT and the Schirmer test evaluate tear film stability and production. Staining with fluorescein, rose bengal, or lissamine green helps detect corneal or conjunctival epithelial damage. Microbiological cultures or PCR are recommended for suspected infections, while allergy testing is indicated for cases of suspected allergic keratoconjunctivitis.[104]
- Identification of the underlying cause: Determining the cause of keratoconjunctivitis involves recognizing specific contributing factors. DED often results from tear film instability or reduced tear production. Allergic keratoconjunctivitis arises from a hypersensitivity reaction, frequently with a seasonal pattern. Bacterial, viral, or fungal pathogens can cause infectious keratoconjunctivitis. Autoimmune disorders, such as Sjögren syndrome and SJS, may also underlie keratoconjunctivitis in certain cases.[105]
- Setting treatment goals: Treatment should aim to address several key objectives. Relieving symptoms such as irritation and discomfort is a primary focus. Efforts should also promote the healing of the corneal and conjunctival epithelium to restore ocular health. Preventing long-term complications, including scarring and vision loss, is essential. Additionally, addressing the underlying cause remains critical to preventing recurrence and ensuring sustained improvement.
Treatment strategies depend on the etiology. Evidence-based therapies for various types of keratoconjunctivitis are outlined below.
- Dry eye-related keratoconjunctivitis: Management often involves multiple approaches. Artificial tears, particularly preservative-free formulations, help maintain tear film integrity and alleviate symptoms. Anti-inflammatory agents, such as topical cyclosporine A (eg, Restasis) and lifitegrast, are effective in reducing inflammation. Punctal occlusion, achieved through the insertion of plugs, can enhance tear retention in severe cases. Lifestyle modifications also play a significant role, including maintaining adequate hydration, using humidifiers, and practicing frequent blinking during prolonged screen time.[106]
- Allergic keratoconjunctivitis: Therapy focuses on symptom control and inflammation reduction. Topical antihistamines and mast cell stabilizers, such as olopatadine, effectively manage allergic symptoms. For severe inflammation, short-term use of low-dose corticosteroids, such as loteprednol, can provide rapid relief. Cold compresses are also recommended to alleviate redness and itching.[107]
- Infectious keratoconjunctivitis: Treatment depends on the causative pathogen. For bacterial infections, broad-spectrum topical antibiotics such as moxifloxacin are effective. Viral infections often require supportive care, including lubricants and cold compresses, while topical antivirals like ganciclovir are used specifically for herpetic keratitis. Fungal infections are treated with topical antifungals such as natamycin, with systemic therapy added in severe or refractory cases.[108]
- Autoimmune or severe cases: Systemic immunosuppressants are essential for addressing autoimmune-related keratoconjunctivitis. AMT may be utilized to promote healing in cases of severe ocular surface damage. Scleral lenses protect the ocular surface and enhance vision in advanced or refractory cases.[109]
Adjunctive therapies include autologous serum eye drops and ω3 fatty acids. Autologous serum eye drops are beneficial for persistent epithelial defects and severe dry eye. Nutritional support with ω3 fatty acids helps reduce inflammation and enhance tear quality.[110]
Regular follow-up ensures timely assessment of therapy response and allows modification of the treatment plan as needed. Monitoring for potential side effects of medications, such as IOP elevation with corticosteroids, is crucial. Periodic evaluation of tear film and ocular surface stability helps prevent recurrence.
Effective treatment planning for keratoconjunctivitis requires a thorough understanding of the underlying cause and a tailored approach to therapy. By addressing symptoms, managing inflammation, and preventing complications, patients can achieve significant relief and improved quality of life.
Toxicity and Adverse Effect Management
Managing the Adverse Effects of Keratoconjunctivitis Therapy
Clinicians should anticipate adverse effects from keratoconjunctivitis treatments and be prepared to manage them. Addressing these effects helps preserve vision, enhance patient comfort, and minimize long-term complications.
The risks of using topical corticosteroids long-term include increased IOP, cataract formation, and secondary infections. To manage these risks, clinicians should regularly monitor IOP, use the lowest effective dose for the shortest duration, and consider switching to safer alternatives like nonsteroidal anti-inflammatory drugs when appropriate.[111] Similarly, preservatives like benzalkonium chloride, commonly found in eye drops, can cause chronic epithelial toxicity and worsen dryness with prolonged use. To mitigate these effects, switching to preservative-free formulations is recommended, and lubricating eye drops should be used to support epithelial healing.
Cyclosporine A (Restasis) may cause a burning sensation or irritation upon application. To manage these effects, clinicians can advise patients to store the medication in a cool place to reduce discomfort upon instilling. Additionally, adjunctive lubricants may be used to alleviate symptoms.[112] Lifitegrast may cause dysgeusia (altered taste) or a burning sensation. Clinicians should encourage patients to continue using this medication, as these symptoms often diminish with time. Lubricating eye drops may be recommended alongside the treatment to reduce irritation.[113]
Contact or scleral lenses may cause ocular injury, eg, corneal abrasions, infections, or hypoxia, if they do not fit properly or are used for an extended period. Poor hygiene by the user may also contribute to ocular damage. To mitigate these risks, the specialist should ensure proper lens fitting and stress the importance of hygiene, regular lens replacement, and proper care. Use should be discontinued if active infection or inflammation occurs.[114]
Allergy or hypersensitivity to medications may cause redness, swelling, and increased discomfort. Management involves discontinuing the offending medication and switching to an alternative treatment. Antihistamines may also be used to address allergic symptoms and alleviate discomfort.[115]
Managing the Long-Term Complications of Chronic Inflammation
Chronic inflammation in keratoconjunctivitis can lead to fibrosis and scarring of the conjunctiva or cornea, resulting in visual impairment. In severe cases, antiscarring agents such as mitomycin C may be used to manage scarring. For advanced cases, surgical interventions like conjunctival transplantation may be necessary.[116]
Additionally, neurotrophic keratopathy, which results from a loss of corneal sensation, can lead to delayed epithelial healing and recurrent defects. Preservative-free lubricants and autologous serum eye drops are recommended to manage this condition. In severe instances, protective measures like tarsorrhaphy may be considered.[117]
Chronic inflammation also increases the risk of recurrent infections, whether bacterial or viral. Prophylactic use of antimicrobial agents during flare-ups, along with strict hygiene practices, can help reduce the occurrence of infections.
Supportive Care and Patient Education
Patients should be educated on the proper use of medications and the importance of adherence to prescribed treatment. Regular follow-up visits should be encouraged to monitor for early signs of toxicity or adverse effects. Lifestyle recommendations, such as avoiding allergens or irritants and using protective eyewear, should be provided.[118]
Effective toxicity and adverse effect management are essential when treating keratoconjunctivitis to optimize outcomes and preserve vision. By identifying potential complications early and implementing tailored interventions, healthcare providers can minimize the risk of long-term damage and enhance the patient's quality of life.
Staging
Keratoconjunctivitis encompasses various ocular surface diseases with different severity levels. Specific subtypes, such as DED, VKC, and SJS-associated and infectious keratoconjunctivitis, have distinct clinical staging systems. However, no universal staging protocol that applies to all cases has been established. The staging systems explained in this section may be used to identify the specific type, track disease progression, and customize treatment approaches.
Dry Eye Disease Staging
DEWS II classifies DED into stages based on symptom severity, tear film stability, and ocular surface damage. In Stage 1 (mild), patients experience mild symptoms of dryness, irritation, or intermittent visual fluctuations. The TBUT is greater than 10 seconds, and minimal or no ocular surface staining occurs. Stage 2 (moderate) involves more frequent symptoms, including burning and sensitivity to light. The TBUT ranges between 5 and 10 seconds, and moderate conjunctival and corneal staining is observed.
In Stage 3 (severe), patients experience constant symptoms of discomfort and visual disturbances. The TBUT is less than 5 seconds, and extensive ocular surface staining is observed, with filamentary keratitis present in some cases. Stage 4 (very severe) is characterized by severe symptoms that significantly impact daily activities. Complete tear film instability or a marked reduction in Schirmer test values is present. Severe corneal epithelial damage, scarring, or ulceration may also be present.[119]
Vernal Keratoconjunctivitis Staging
VKC is staged based on clinical findings and disease severity. In the mild stage, symptoms are intermittent, including itching and redness. Small papillae, less than 1 mm in size, are observed on the upper tarsal conjunctiva. The moderate stage involves persistent symptoms, with increased photophobia and mucus discharge. Giant papillae greater than 1 mm in size are present, along with mild corneal involvement, which may manifest as superficial punctate keratitis. In the severe stage, intense symptoms significantly impact vision. Shield ulcers, extensive punctate keratitis, or corneal plaques are observed.
Stevens-Johnson Syndrome-Associated Keratoconjunctivitis Staging
Staging in SJS is often based on acuity, the degree of ocular surface damage, and the presence of complications. In the acute stage, conjunctival hyperemia, epithelial sloughing, and pseudomembrane formation are observed. Early corneal epithelial defects may also occur. In the chronic stage, symblepharon formation, severe conjunctival scarring, and lid margin keratinization are prominent. In more advanced stages, progressive corneal neovascularization, opacification, or perforation can occur.
General Classification for Infectious Keratoconjunctivitis
The staging of infectious keratoconjunctivitis focuses on corneal involvement and the severity of infection. In the mild (early) stage, superficial corneal epithelial defects and minimal infiltration are present, along with limited conjunctival hyperemia and discharge. In the moderate stage, stromal involvement occurs, with moderate corneal haze or edema. Increased conjunctival redness and significant discharge are also seen. In the severe stage, dense stromal infiltration, ulceration, and impending perforation are observed. Vision is significantly impaired, and the risk of corneal scarring is high.[120]
The staging of keratoconjunctivitis varies by subtype, but all systems help assess severity, monitor progression, and guide appropriate management. Early identification of the disease stage is crucial to implementing effective treatment and preventing complications, including scarring and vision loss.
Prognosis
EKC is a self-limiting disease that typically resolves within 2 to 3 weeks. Recurrence may occur with subsequent viral infections, but long-term complications are rare. Similarly, VKC is usually benign and self-limiting, often resolving spontaneously with age, particularly during the peripubescent period. If symptoms persist beyond childhood and adolescence, which can occur in up to 12% of patients, the condition may evolve into AKC. This form shares similar symptoms, diagnostic features, and treatment protocols but requires chronic management.
DES presents with a broad spectrum of disease severity. Most patients can achieve symptomatic control by following a regular treatment regimen. Although a cure is not possible, symptoms can be well-managed. Environmental factors, particularly changes in the weather, geography, or air pollution levels, may also help alleviate symptoms. Patients with well-controlled disease may occasionally experience breakthrough episodes, which may require a temporary increase in treatment intensity.
The treatment of SLK is typically very successful. In cases that do not respond to or are symptomatically unmanageable with conservative treatments, more aggressive approaches, such as chemocautery and surgical conjunctival resection, have proven effective. The prognosis of keratoconjunctivitis depends on the underlying cause, severity, prompt diagnosis, and treatment efficacy. Manifestations of this condition can range from mild and self-limiting to severe and vision-threatening, particularly when associated with chronic inflammation, infections, or autoimmune disorders. Early intervention and adherence to treatment help prevent complications and ensure favorable outcomes.[121]
Factors Influencing Prognosis
Several factors influence the prognosis of keratoconjunctivitis. These factors include the underlying cause, severity and duration of the condition, timeliness of diagnosis and treatment, and patient compliance with prescribed therapies.
For DED, management with lubricants and anti-inflammatory treatments typically results in a good prognosis. However, severe cases with chronic inflammation may lead to corneal epithelial damage or scarring, which can affect vision. Allergic keratoconjunctivitis generally has an excellent prognosis when managed with antihistamines and allergen avoidance, although severe forms like VKC and AKC can produce complications, such as shield ulcers or corneal scarring.
In cases of infectious keratoconjunctivitis, bacterial forms usually respond well to antibiotics and have a favorable prognosis if treated early. Viral keratoconjunctivitis, including adenoviral and herpetic forms, may cause recurrent episodes and increase the risk of stromal damage or scarring, which can affect long-term vision. Infectious keratoconjunctivitis caused by fungi and parasites, such as Acanthamoeba, tends to have a worse prognosis due to delayed diagnosis and resistance to treatment.
The severity and duration of the condition also impact prognosis. Mild cases of keratoconjunctivitis typically resolve without complications, but severe or chronic cases may lead to persistent epithelial defects, corneal scarring, neovascularization, or even vision loss if not adequately managed.[122]
Timely diagnosis and intervention are crucial for improving outcomes. Early treatment can control inflammation and prevent complications, while delayed treatment increases the risk of permanent damage to the ocular surface and visual impairment.[123]
Patient compliance with prescribed therapies and regular follow-up are essential to determining outcomes. Adherence to treatment plans ensures better results, while poor compliance, particularly in chronic conditions, can lead to disease progression or recurrence.[124]
Prognosis Based on Keratoconjunctivitis Subtypes
For DED, mild to moderate cases generally have a favorable prognosis with appropriate management. Severe disease, particularly when corneal complications are present, may require advanced therapies, such as autologous serum and scleral lenses, to restore vision and comfort.[125]
VKC and AKC typically have a good prognosis if treated early. However, severe forms can lead to vision-threatening complications, including shield ulcers or corneal opacities.
Autoimmune conditions, such as SJS and OCP, have a more guarded prognosis. These diseases are associated with the potential for severe conjunctival scarring, symblepharon, and corneal opacification. Early and aggressive treatment can help preserve vision.[126]
Infectious keratoconjunctivitis generally has a good prognosis with timely treatment, particularly for bacterial cases. However, viral and fungal infections may relapse or require prolonged treatment, which can affect the long-term prognosis.[127]
Complications Impacting Prognosis
Complications that may impact prognosis include the following:
- Persistent epithelial defects
- Corneal scarring or opacification
- Secondary infections
- Neovascularization leading to vision loss
- Chronic pain or discomfort affecting quality of life [128]
The prognosis of keratoconjunctivitis varies widely, depending on the etiology, severity, and timeliness of the intervention. With early diagnosis and appropriate treatment, most cases achieve favorable outcomes. However, severe or chronic cases require ongoing management to prevent complications and preserve vision. Regular follow-up and patient education are vital to ensuring the best possible prognosis.
Complications
Symptoms of keratoconjunctivitis result from prolonged inflammation, persistent rubbing, and certain treatment modalities. Although rare, chronic inflammation of the conjunctiva and cornea can lead to complications, such as symblepharon, forniceal shortening, conjunctival scarring, limbal stem cell deficiency, and corneal scarring. Persistent and aggressive eye rubbing can cause acquired eyelid ptosis and early cataract formation, both of which correlate with the development of keratoconus.
Healthcare providers must also be aware of the potential side effects of medications. Topical corticosteroids, for example, can increase IOP, leading to steroid-induced glaucoma, which is irreversible and vision-threatening. Both topical and systemic steroids can also accelerate cataract formation. Additionally, topical corticosteroids may predispose patients to ocular HSV reactivation or worsen ocular herpetic or bacterial infections. Therefore, providers must rule out herpetic and bacterial infections before prescribing topical corticosteroids.
If not diagnosed and treated promptly, keratoconjunctivitis can produce various complications involving the ocular surface and vision. These complications arise from ongoing inflammation, structural damage, and secondary infections. Recognizing the potential complications is crucial for developing effective treatment plans to prevent long-term sequelae.[129]
Common complications of keratoconjunctivitis include the following:
- Corneal epithelial defects: Chronic inflammation can impair epithelial healing, resulting in recurrent or persistent epithelial defects, which increase the risk of secondary bacterial infections and corneal ulceration.
- Corneal scarring and opacification: Severe or untreated cases may lead to stromal involvement and scarring, reducing corneal transparency and causing visual impairment. Scarring is particularly common in VKC, herpetic keratitis, and chemical injuries.
- Neovascularization: Chronic hypoxia or inflammation can induce new blood vessel formation in the cornea. Neovascularization compromises corneal clarity and may lead to lipid deposition and further vision loss.[130]
- Shield ulcers: These common VKC sequelae present as large, oval corneal ulcers that form due to mechanical irritation from giant papillae on the tarsal conjunctiva. Shield ulcers may become infected or result in scarring if not managed promptly.[131]
- Secondary infections: Persistent epithelial defects or an impaired tear film predispose the eye to bacterial, viral, or fungal infections, which can worsen the condition and complicate treatment.[132]
- Symblepharon formation: In severe inflammatory or autoimmune conditions, such as SJS and OCP, adhesions may form between the palpebral and bulbar conjunctiva, leading to restricted ocular movements and discomfort.[133]
- Limbal stem cell deficiency: Chronic inflammation or chemical injuries can damage limbal stem cells, resulting in conjunctivalization of the corneal surface, recurrent erosions, and poor visual outcomes.[134]
- Keratopathy and keratinization: Prolonged inflammation or scarring can lead to corneal keratinization, which is characterized by the formation of an opaque, rough surface that furthers the impression.
- Reduced corneal sensitivity: Nerve damage from inflammation, infections (eg, HSV), or neurotrophic keratopathy can lead to corneal insensitivity, delaying healing and leading to recurrent epithelial breakdown.[135]
- Increased IOP: Long-term use of topical corticosteroids to manage inflammation may result in steroid-induced glaucoma, necessitating careful IOP monitoring.
- Chronic pain and photophobia: Persistent inflammation or corneal complications can cause chronic ocular pain and light sensitivity, significantly affecting the patient's quality of life.[136]
- Vision loss: Severe complications, such as corneal perforation, extensive scarring, and secondary glaucoma, may result in permanent vision loss if left untreated.[137]
Early diagnosis and aggressive management of inflammation and infections can prevent most complications. Regular follow-up and monitoring are essential, especially in individuals with chronic or recurrent keratoconjunctivitis. Advanced therapies, such as AMT, scleral lens use, and SCT, can help manage severe cases and restore ocular surface integrity.
Keratoconjunctivitis can cause complications ranging from mild epithelial damage to severe vision-threatening conditions. Prompt recognition and comprehensive management are crucial to minimizing these risks and preserving long-term ocular health and vision.
Postoperative and Rehabilitation Care
Postoperative and rehabilitation care are crucial for ensuring optimal recovery and long-term outcomes following surgical or advanced therapeutic intervention for severe or refractory keratoconjunctivitis. This care promotes healing, prevents complications, and restores visual function.[138] The key elements of postoperative care include the following:
- Monitoring and follow-up: Regular follow-up appointments assess the healing process and identify early signs of complications, such as infection, graft rejection, and scarring. Slit-lamp examination and imaging techniques, such as AS-OCT, may be performed for detailed ocular surface and corneal integrity evaluation.[139]
- Medications and eye drops: Topical antibiotics are prescribed to prevent postoperative infections. Corticosteroids or cyclosporine help control inflammation and reduce the risk of scarring. Preservative-free artificial tears maintain ocular surface hydration and promote epithelial healing.[140]
- Pain management: Oral analgesics or topical anesthetic drops are used to manage postoperative discomfort. Cold compresses may be used to alleviate inflammation and reduce swelling.[141]
- Lifestyle modifications and eye protection: Patients must be advised to avoid strenuous activities, eye rubbing, and exposure to irritants, such as smoke and dust. Protective eyewear should be recommended to shield the eyes from potential trauma or UV exposure.[142]
Rehabilitation care consists of the following:
- Ocular surface restoration: AMT effectively promotes healing in cases involving persistent epithelial defects or severe inflammation. Scleral lenses provide corneal protection, improve tear film stability, and enhance visual acuity in patients with significant scarring or irregular astigmatism.
- Physical and visual rehabilitation: Vision therapy helps manage visual disturbances or binocular vision problems from chronic keratoconjunctivitis. Eyelid hygiene and warm compresses are recommended for managing associated blepharitis and improving tear film quality.[143]
- Psychological support: Providing counseling or encouragement to join support groups can help patients cope with chronic symptoms, reduced vision, or the emotional impact of the disease.[144]
- Advanced therapies for severe cases: Autologous serum eye drops promote epithelial repair in cases of severe ocular surface damage. SCT is recommended for patients with limbal stem cell deficiency or significant conjunctival injury.[145]
Identifying and managing underlying systemic conditions, such as autoimmune disorders and allergies, can prevent the recurrence of keratoconjunctivitis. Adherence to prescribed treatment regimens and regular follow-ups are also encouraged for long-term disease control.
Postoperative and rehabilitation care are vital in optimizing outcomes for patients with keratoconjunctivitis. A comprehensive and individualized approach ensures proper healing, minimizes complications, and restores ocular surface integrity and visual function. By integrating medical, surgical, and supportive care strategies, healthcare providers can significantly improve the quality of life for patients, enabling them to return to daily activities with minimal discomfort and reduced risk of long-term sequelae. Continuous monitoring and patient education are essential to ensure adherence to treatment plans, early detection of complications, and sustained disease control, ultimately preserving vision and ocular health.
Consultations
Consultations are critical in managing keratoconjunctivitis, especially in complex or refractory cases. Collaboration with specialists ensures accurate diagnosis, effective treatment, and appropriate management of underlying systemic conditions or complications. Key consultations include the following:
- Ophthalmologist: The cornea specialist, essential for diagnosing and managing severe or refractory keratoconjunctivitis cases. The ophthalmologist provides expertise in advanced therapeutic options, such as AMT, scleral lenses, and limbal SCT.[146]
- Allergist or immunologist: Recommended for patients with allergic keratoconjunctivitis, ie, VKC and AKC. These specialists assist in identifying allergens through testing and prescribe systemic or topical antihistamines, mast cell stabilizers, or immunotherapy to manage symptoms effectively.
- Rheumatologist: Necessary for individuals with autoimmune-related keratoconjunctivitis, such as in cases associated with Sjögren syndrome, rheumatoid arthritis, and SLE. These experts guide systemic immunosuppressive therapies to control the underlying autoimmune conditions.[147]
- Infectious disease specialist: Consulted for cases of infectious keratoconjunctivitis caused by bacteria, viruses, fungi, or parasites, especially when resistance or atypical pathogens complicate management. This specialist provides advice on systemic or topical antimicrobial therapies tailored to the identified pathogen.[148]
- Dermatologist: Required for keratoconjunctivitis related to skin conditions, such as SJS and atopic dermatitis. These professionals help treat skin and mucosal lesions and minimize ocular complications.[149]
- Endocrinologist: Involved when keratoconjunctivitis occurs alongside hormonal dysfunctions, such as diabetes or thyroid disease. The endocrinologist focuses on optimizing systemic control of endocrine disorders, which can improve ocular outcomes.[150]
- Psychologist or psychiatrist: Provides critical support in cases where chronic keratoconjunctivitis significantly impacts mental health or quality of life. These professionals offer counseling and treatment for psychological challenges such as anxiety or depression.[151]
Interprofessional consultations are crucial for effectively managing keratoconjunctivitis, especially in severe or systemic cases. Coordinated care between ophthalmologists and other specialists ensures a holistic approach, addressing both ocular and systemic aspects of the disease, ultimately improving patient outcomes.
Deterrence and Patient Education
Patients should receive counseling about the prolonged time required for symptomatic resolution. Immediate relief is uncommon, even with strict adherence to the treatment regimen. Clear and purposeful discussions with patients, or with parents in pediatric cases, can help set realistic expectations and prevent unnecessary, time-consuming, and costly return visits to primary care, urgent care, or emergency departments. Additionally, patients should be specifically advised to watch for warning signs, including decreased vision, worsening pain, increased photophobia, worsening discharge, and a lack of improvement after a week of treatment.
Preventing the recurrence or exacerbation of keratoconjunctivitis requires a proactive approach that emphasizes deterrence strategies and comprehensive patient education. Providing patients with a clear understanding of the disease, identifying potential triggers, and highlighting the importance of proper eye care can greatly reduce the disease burden and enhance treatment outcomes. Strategies for deterrence and patient education include the following:
- Promoting ocular hygiene: Educating patients on the importance of maintaining clean eyelids and avoiding eye rubbing helps prevent mechanical irritation and reduce the risk of infection. Regular use of sterile wipes or warm compresses for eyelid hygiene should be encouraged, especially for individuals with associated blepharitis.[152]
- Avoidance of triggers: Patients should be educated on identifying and avoiding environmental factors that exacerbate symptoms, including dust, pollen, smoke, and air pollution. Screen time should be minimized, and proper lighting must be ensured to reduce digital eye strain and dryness.[153]
- Encouraging treatment adherence: The importance of adherence to prescribed medications, such as artificial tears, anti-inflammatory agents, and antihistamines, should be emphasized. Nonadherence must be explained as a risk for worsening symptoms or complications, including scarring and vision loss.
- Protective measures: Protective eyewear, such as goggles and sunglasses, should be advised for use outdoors or in dusty environments. Proper lens hygiene, including cleaning, disinfecting, and replacing lenses as instructed, must also be stressed for contact lens users.[154]
- Lifestyle and nutritional recommendations: A balanced diet rich in ω3 fatty acids, antioxidants, and vitamins should be encouraged to support ocular surface health. Adequate hydration is also recommended to promote tear production and prevent dryness.[155]
- Recognizing early warning signs: Patients should be taught to recognize symptoms that require prompt medical attention, such as increased redness, swelling, severe pain, and vision changes. Regular follow-ups must be emphasized, particularly for those with chronic or recurrent keratoconjunctivitis.
- Allergen management: For allergic keratoconjunctivitis, allergen-proof bedding, regular air filter cleaning, and limiting outdoor exposure during high pollen seasons should be suggested. The potential benefits of allergen immunotherapy may also be discussed in collaboration with an allergist.[156]
Patient education techniques that may be employed include the following:
- Interactive education: Models or diagrams may be used to explain the ocular surface anatomy and the impact of keratoconjunctivitis. Proper techniques for applying eye drops, cleaning eyelids, or wearing contact lenses should be demonstrated.
- Written and digital resources: Educational brochures or links to reputable online resources may be provided for further reading. Mobile apps may be utilized to send medication reminders or track symptom improvement.[157]
- Individualized counseling: Education should be tailored to the patient’s specific condition, lifestyle, and treatment plan, with individual concerns and barriers to adherence being addressed.[158]
Deterrence and patient education are essential components of keratoconjunctivitis management. Healthcare providers can enhance treatment adherence, prevent disease recurrence, and promote long-term ocular health by equipping patients with the necessary knowledge and skills to manage their condition effectively. A proactive approach, including regular follow-ups, lifestyle modifications, and clear communication, empowers patients to take an active role in their care, ultimately leading to improved clinical outcomes and a better quality of life.
Pearls and Other Issues
Pearls
Keratoconjunctivitis often involves a complex interplay of tear film instability, inflammation, and epithelial damage. Recognizing this multifactorial nature aids in implementing a comprehensive management approach rather than focusing on a single cause. Early diagnosis plays a critical role in preventing more severe complications, such as corneal scarring or perforation, by promptly identifying underlying etiologies, including dry eye, infection, allergy, and autoimmune disorders. Management should be individualized, balancing symptomatic relief with addressing the root cause, such as using artificial tears for dryness, antihistamines for allergies, and antibiotics for infections.[159] In refractory cases, newer treatments like lifitegrast, cyclosporine, and autologous serum eye drops offer significant benefits and should be considered early in severe cases.[160]
Mild-to-moderate keratoconjunctivitis may be managed in an outpatient setting with topical medications and lifestyle modifications. However, severe cases involving significant corneal injury, vision impairment, or systemic autoimmune conditions may require interprofessional care and closer follow-up, sometimes necessitating hospitalization for intensive therapy.
Chronic or severe keratoconjunctivitis can affect mental health due to discomfort and visual impairment, highlighting the need for holistic patient care. Psychosocial interventions may complement medical treatment to improve the quality of life for affected individuals.
Research into regenerative therapies, such as SCT and exosome-based treatments, holds promise for addressing severe ocular surface damage. These advancements could offer new treatment options for patients with refractory or severe ocular conditions.
Pitfalls
Misdiagnosis of viral keratoconjunctivitis as bacterial or allergic types may lead to inappropriate treatment, prolonged recovery, or worsening symptoms.[161] The overuse of corticosteroids, particularly when prolonged or inappropriate, can result in steroid-induced glaucoma, cataracts, or secondary infections.[162]
Nonadherence to therapy is another concern, as patients may discontinue treatment prematurely once symptoms improve, which increases the risk of recurrence or incomplete resolution.[163] Additionally, underestimating systemic associations linked to keratoconjunctivitis, such as Sjögren syndrome or HSV infection, can delay effective treatment.[164]
Prevention
Preventing keratoconjunctivitis involves several key measures. Environmental modifications, such as reducing exposure to allergens, using humidifiers, and avoiding irritants like smoke or wind, can help minimize risks. Proper hygiene, including educating patients on handwashing and avoiding eye rubbing, is crucial to reducing the risk of infection. For those who wear contact lenses, ensuring proper cleaning, storage, and timely replacement is essential to prevent microbial keratitis. Regular eye exams, particularly for high-risk populations such as individuals with autoimmune disorders or chronic dry eye, can help detect early signs of keratoconjunctivitis.[165]
Additional Information
A thorough understanding of the clinical nuances, early recognition, and tailored management strategies are key to improving outcomes in keratoconjunctivitis. Avoiding common pitfalls and focusing on prevention can significantly reduce the risk of complications, enhance patient satisfaction, and improve quality of life. By staying updated on advances in diagnostic tools and therapeutic options, healthcare providers can offer comprehensive care that addresses both the ocular and systemic aspects of this multifaceted condition.
Enhancing Healthcare Team Outcomes
Keratoconjunctivitis and conjunctivitis are common reasons for visits to primary care providers and nurse practitioners. Referring every case of red eye to an ophthalmologist is unnecessary and can be burdensome for the patient, both in terms of time and cost. Referral to ophthalmology should occur when any of the following develop: persistent or worsening disease, vision loss, moderate to severe pain, severe purulent discharge, significant corneal involvement, conjunctival scarring, suspicion of herpetic disease, or lack of response to therapy. Special attention should be given to patients who wear contact lenses, may require steroid treatment, or present with severe photophobia.[166][167][168]
Keratoconjunctivitis requires an interprofessional team approach, with physicians, specialists, specialty-trained nurses, and pharmacists all collaborating across disciplines to achieve optimal patient outcomes. Effective management, particularly in complex or chronic cases, relies on clear communication, coordinated care, and adherence to ethical and professional responsibilities. This approach ensures the best possible patient-centered outcomes, safety, and overall team performance.
Skills and Competencies
Physicians, particularly ophthalmologists, must maintain clinical expertise by staying updated on the latest diagnostic tools and treatment strategies, including advanced therapies like biologics and regenerative medicine. Critical thinking and decision-making are essential skills for advanced practitioners, such as nurse practitioners or physician assistants, who contribute by assessing patient progress, identifying complications, and adjusting treatment plans as needed. Patient education is a crucial role for nurses, who are responsible for educating patients on medication use, eye hygiene, and adherence to prescribed treatments.[169]
Interprofessional Communication
Regular case discussions should be conducted through team meetings to review patient cases, share insights, and ensure alignment of treatment goals. Electronic health records (EHR) should be used to document patient progress, allowing all team members to access up-to-date information. Clear referral processes are essential for streamlined communication between primary care providers, specialists, and allied health professionals to ensure timely consultations and interventions.[170]
Care Coordination
A cohesive plan should be developed with input from ophthalmologists, allergists, rheumatologists, and other relevant specialists. Follow-up care should be coordinated, ensuring seamless transitions between outpatient, inpatient, and rehabilitation settings to prevent gaps in care. Pharmacists are responsible for ensuring accurate medication dosing, identifying potential drug interactions, and providing guidance on preservative-free formulations or compounded medications.[171]
Patient-Centered Care
Individualized treatment should be provided by tailoring therapies to address specific patient needs, such as severity, underlying conditions, and lifestyle factors. Patients must be empowered by being involved in decision-making, receiving clear instructions, and setting realistic expectations for outcomes. Holistic support is essential, addressing psychological and emotional well-being while recognizing the impact of chronic symptoms on mental health and quality of life.[172]
Ethical and Professional Responsibilities
Respect for patient autonomy involves ensuring informed consent for all procedures and upholding patient preferences. Maintaining confidentiality is crucial, requiring privacy to be upheld in all communications and record-keeping. Cultural competence should be practiced by understanding and valuing cultural differences that may influence treatment acceptance and adherence.[173]
Strategies for Improving Team Performance
Ongoing training and education should be prioritized, with the team regularly updated on advances in keratoconjunctivitis management and emerging treatments. Simulation-based training, such as mock scenarios, can be conducted to practice managing complications such as infections or medication side effects. Evaluation mechanisms should be implemented, allowing team members to provide feedback and suggestions for improving care processes.[174][175]
Ensuring Patient Safety
Monitoring and reporting adverse events should involve establishing protocols to track and address side effects or complications from treatments such as corticosteroids or immunosuppressants. Errors may be minimized by double-checking prescriptions, verifying patient instructions, and maintaining accurate documentation.[176]
Enhancing healthcare team outcomes in keratoconjunctivitis management requires a collaborative, patient-centered approach involving physicians, advanced practitioners, nurses, pharmacists, and other allied health professionals. Healthcare teams can improve patient outcomes, ensure safety, and optimize overall care delivery through effective communication, coordinated care, and adherence to ethical responsibilities. Continuous training, clear roles, and mutual respect among team members are essential for achieving these goals.
Media
(Click Image to Enlarge)
References
Alfonso SA, Fawley JD, Alexa Lu X. Conjunctivitis. Primary care. 2015 Sep:42(3):325-45. doi: 10.1016/j.pop.2015.05.001. Epub 2015 Jul 29 [PubMed PMID: 26319341]
Kaur K, Gurnani B. Vernal Keratoconjunctivitis. StatPearls. 2025 Jan:(): [PubMed PMID: 35015458]
Stapleton F, Abad JC, Barabino S, Burnett A, Iyer G, Lekhanont K, Li T, Liu Y, Navas A, Obinwanne CJ, Qureshi R, Roshandel D, Sahin A, Shih K, Tichenor A, Jones L. TFOS lifestyle: Impact of societal challenges on the ocular surface. The ocular surface. 2023 Apr:28():165-199. doi: 10.1016/j.jtos.2023.04.006. Epub 2023 Apr 14 [PubMed PMID: 37062429]
Kaur K, Gurnani B. Slit-Lamp Biomicroscope. StatPearls. 2025 Jan:(): [PubMed PMID: 36512656]
Labib BA, Minhas BK, Chigbu DI. Management of Adenoviral Keratoconjunctivitis: Challenges and Solutions. Clinical ophthalmology (Auckland, N.Z.). 2020:14():837-852. doi: 10.2147/OPTH.S207976. Epub 2020 Mar 17 [PubMed PMID: 32256043]
Hashmi MF, Gurnani B, Benson S. Conjunctivitis. StatPearls. 2025 Jan:(): [PubMed PMID: 31082078]
Baab S, Le PH, Gurnani B, Kinzer EE. Allergic Conjunctivitis. StatPearls. 2025 Jan:(): [PubMed PMID: 28846256]
Azari AA, Barney NP. Conjunctivitis: a systematic review of diagnosis and treatment. JAMA. 2013 Oct 23:310(16):1721-9. doi: 10.1001/jama.2013.280318. Epub [PubMed PMID: 24150468]
Level 1 (high-level) evidenceChigbu DI, Labib BA. Pathogenesis and management of adenoviral keratoconjunctivitis. Infection and drug resistance. 2018:11():981-993. doi: 10.2147/IDR.S162669. Epub 2018 Jul 17 [PubMed PMID: 30046247]
Doğan Ü, Ağca S. Investigation of possible risk factors in the development of seasonal allergic conjunctivitis. International journal of ophthalmology. 2018:11(9):1508-1513. doi: 10.18240/ijo.2018.09.13. Epub 2018 Sep 18 [PubMed PMID: 30225226]
Cher I. Superior limbic keratoconjunctivitis: multifactorial mechanical pathogenesis. Clinical & experimental ophthalmology. 2000 Jun:28(3):181-4 [PubMed PMID: 10981793]
Ahmad B, Gurnani B, Patel BC. Herpes Simplex Keratitis. StatPearls. 2025 Jan:(): [PubMed PMID: 31424862]
Gurnani B, Kaur K. Bacterial Keratitis. StatPearls. 2025 Jan:(): [PubMed PMID: 34662023]
Somani SN, Ronquillo Y, Moshirfar M. Acanthamoeba Keratitis. StatPearls. 2025 Jan:(): [PubMed PMID: 31751053]
Gurnani B, Kaur K. Anti-infective therapies for Pythium insidiosum keratitis. Expert review of anti-infective therapy. 2024 Oct:22(10):805-817. doi: 10.1080/14787210.2024.2403146. Epub 2024 Sep 13 [PubMed PMID: 39268901]
Kılıççıoğlu A, Oncel D, Celebi ARC. Autoimmune Disease-Related Dry Eye Diseases and Their Placement Under the Revised Classification Systems: An Update. Cureus. 2023 Dec:15(12):e50276. doi: 10.7759/cureus.50276. Epub 2023 Dec 10 [PubMed PMID: 38196419]
Kaur RP, Gurnani B, Kaur K. Intricate insights into immune response in dry eye disease. Indian journal of ophthalmology. 2023 Apr:71(4):1248-1255. doi: 10.4103/IJO.IJO_481_23. Epub [PubMed PMID: 37026255]
Gurnani B, Christy J, Narayana S, Kaur K, Moutappa F. Corneal Perforation Secondary to Rosacea Keratitis Managed with Excellent Visual Outcome. Nepalese journal of ophthalmology : a biannual peer-reviewed academic journal of the Nepal Ophthalmic Society : NEPJOPH. 2022 Jan:14(27):162-167. doi: 10.3126/nepjoph.v14i1.36454. Epub [PubMed PMID: 35996914]
Rauchman SH, Locke B, Albert J, De Leon J, Peltier MR, Reiss AB. Toxic External Exposure Leading to Ocular Surface Injury. Vision (Basel, Switzerland). 2023 Apr 3:7(2):. doi: 10.3390/vision7020032. Epub 2023 Apr 3 [PubMed PMID: 37092465]
Domingo E, Moshirfar M, Zeppieri M, Zabbo CP. Corneal Abrasion. StatPearls. 2025 Jan:(): [PubMed PMID: 30422555]
Sarkar J, Chaudhary S, Namavari A, Ozturk O, Chang JH, Yco L, Sonawane S, Khanolkar V, Hallak J, Jain S. Corneal neurotoxicity due to topical benzalkonium chloride. Investigative ophthalmology & visual science. 2012 Apr 6:53(4):1792-802. doi: 10.1167/iovs.11-8775. Epub 2012 Apr 6 [PubMed PMID: 22410563]
Sommer A. Effects of vitamin A deficiency on the ocular surface. Ophthalmology. 1983 Jun:90(6):592-600 [PubMed PMID: 6604247]
Walker NR, Mistry RK, Mazzoni T. Facial Nerve Palsy. StatPearls. 2025 Jan:(): [PubMed PMID: 31747222]
Asimellis G, Kaufman EJ. Keratoconus. StatPearls. 2025 Jan:(): [PubMed PMID: 29262160]
Jaskiewicz K, Maleszka-Kurpiel M, Michalski A, Ploski R, Rydzanicz M, Gajecka M. Non-allergic eye rubbing is a major behavioral risk factor for keratoconus. PloS one. 2023:18(4):e0284454. doi: 10.1371/journal.pone.0284454. Epub 2023 Apr 13 [PubMed PMID: 37053215]
Ahmad B, Zeppieri M, Patel BC. Trachoma. StatPearls. 2025 Jan:(): [PubMed PMID: 32644461]
Singhal D, Sahay P, Maharana PK, Raj N, Sharma N, Titiyal JS. Vernal Keratoconjunctivitis. Survey of ophthalmology. 2019 May-Jun:64(3):289-311. doi: 10.1016/j.survophthal.2018.12.001. Epub 2018 Dec 12 [PubMed PMID: 30550738]
Level 3 (low-level) evidenceKumar S. Vernal keratoconjunctivitis: a major review. Acta ophthalmologica. 2009 Mar:87(2):133-47. doi: 10.1111/j.1755-3768.2008.01347.x. Epub 2008 Sep 11 [PubMed PMID: 18786127]
Jhanji V, Chan TC, Li EY, Agarwal K, Vajpayee RB. Adenoviral keratoconjunctivitis. Survey of ophthalmology. 2015 Sep-Oct:60(5):435-43. doi: 10.1016/j.survophthal.2015.04.001. Epub 2015 May 5 [PubMed PMID: 26077630]
Level 3 (low-level) evidenceHessen M, Akpek EK. Dry eye: an inflammatory ocular disease. Journal of ophthalmic & vision research. 2014 Apr:9(2):240-50 [PubMed PMID: 25279127]
Narayanan S, Redfern RL, Miller WL, Nichols KK, McDermott AM. Dry eye disease and microbial keratitis: is there a connection? The ocular surface. 2013 Apr:11(2):75-92. doi: 10.1016/j.jtos.2012.12.002. Epub 2013 Jan 29 [PubMed PMID: 23583043]
Pennington MR, Saha A, Painter DF, Gavazzi C, Ismail AM, Zhou X, Chodosh J, Rajaiya J. Disparate Entry of Adenoviruses Dictates Differential Innate Immune Responses on the Ocular Surface. Microorganisms. 2019 Sep 13:7(9):. doi: 10.3390/microorganisms7090351. Epub 2019 Sep 13 [PubMed PMID: 31540200]
Yu FS, Hazlett LD. Toll-like receptors and the eye. Investigative ophthalmology & visual science. 2006 Apr:47(4):1255-63 [PubMed PMID: 16565355]
Rolando M, Zierhut M. The ocular surface and tear film and their dysfunction in dry eye disease. Survey of ophthalmology. 2001 Mar:45 Suppl 2():S203-10 [PubMed PMID: 11587144]
Level 3 (low-level) evidenceLobo AM, Agelidis AM, Shukla D. Pathogenesis of herpes simplex keratitis: The host cell response and ocular surface sequelae to infection and inflammation. The ocular surface. 2019 Jan:17(1):40-49. doi: 10.1016/j.jtos.2018.10.002. Epub 2018 Oct 11 [PubMed PMID: 30317007]
Dartt DA, Masli S. Conjunctival epithelial and goblet cell function in chronic inflammation and ocular allergic inflammation. Current opinion in allergy and clinical immunology. 2014 Oct:14(5):464-70. doi: 10.1097/ACI.0000000000000098. Epub [PubMed PMID: 25061855]
Level 3 (low-level) evidenceBöhm EW, Buonfiglio F, Voigt AM, Bachmann P, Safi T, Pfeiffer N, Gericke A. Oxidative stress in the eye and its role in the pathophysiology of ocular diseases. Redox biology. 2023 Dec:68():102967. doi: 10.1016/j.redox.2023.102967. Epub 2023 Nov 18 [PubMed PMID: 38006824]
Moretti L, Stalfort J, Barker TH, Abebayehu D. The interplay of fibroblasts, the extracellular matrix, and inflammation in scar formation. The Journal of biological chemistry. 2022 Feb:298(2):101530. doi: 10.1016/j.jbc.2021.101530. Epub 2021 Dec 23 [PubMed PMID: 34953859]
Watson SL, Le DT. Corneal neuropathic pain: a review to inform clinical practice. Eye (London, England). 2024 Aug:38(12):2350-2358. doi: 10.1038/s41433-024-03060-x. Epub 2024 Apr 16 [PubMed PMID: 38627548]
Nebbioso M, Del Regno P, Gharbiya M, Sacchetti M, Plateroti R, Lambiase A. Analysis of the Pathogenic Factors and Management of Dry Eye in Ocular Surface Disorders. International journal of molecular sciences. 2017 Aug 13:18(8):. doi: 10.3390/ijms18081764. Epub 2017 Aug 13 [PubMed PMID: 28805710]
Burrow MK, Gurnani B, Patel BC. Keratoconjunctivitis. StatPearls. 2025 Jan:(): [PubMed PMID: 31194419]
Kamil S, Mohan RR. Corneal stromal wound healing: Major regulators and therapeutic targets. The ocular surface. 2021 Jan:19():290-306. doi: 10.1016/j.jtos.2020.10.006. Epub 2020 Oct 28 [PubMed PMID: 33127599]
Foster MH. Basement membranes and autoimmune diseases. Matrix biology : journal of the International Society for Matrix Biology. 2017 Jan:57-58():149-168. doi: 10.1016/j.matbio.2016.07.008. Epub 2016 Aug 2 [PubMed PMID: 27496347]
Hodges RR, Dartt DA. Tear film mucins: front line defenders of the ocular surface; comparison with airway and gastrointestinal tract mucins. Experimental eye research. 2013 Dec:117():62-78. doi: 10.1016/j.exer.2013.07.027. Epub 2013 Aug 14 [PubMed PMID: 23954166]
Georgoudis P, Sabatino F, Szentmary N, Palioura S, Fodor E, Hamada S, Scholl HPN, Gatzioufas Z. Ocular Mucous Membrane Pemphigoid: Current State of Pathophysiology, Diagnostics and Treatment. Ophthalmology and therapy. 2019 Mar:8(1):5-17. doi: 10.1007/s40123-019-0164-z. Epub 2019 Jan 29 [PubMed PMID: 30694513]
Fu L, Zeppieri M. Corneal Neurotization. StatPearls. 2025 Jan:(): [PubMed PMID: 39383277]
Kaur K, Gurnani B, Nayak S, Deori N, Kaur S, Jethani J, Singh D, Agarkar S, Hussaindeen JR, Sukhija J, Mishra D. Digital Eye Strain- A Comprehensive Review. Ophthalmology and therapy. 2022 Oct:11(5):1655-1680. doi: 10.1007/s40123-022-00540-9. Epub 2022 Jul 9 [PubMed PMID: 35809192]
Jaffet J, Singh V, Chaurasia S, Jakati S, Hazari A, Sangwan V. Clinical, histological and immunohistochemistry characteristics of cornea in the sequelae stage of chronic vernal keratoconjunctivitis. Indian journal of ophthalmology. 2022 Jan:70(1):59-64. doi: 10.4103/ijo.IJO_1179_21. Epub [PubMed PMID: 34937208]
Goldstein MH, Silva FQ, Blender N, Tran T, Vantipalli S. Ocular benzalkonium chloride exposure: problems and solutions. Eye (London, England). 2022 Feb:36(2):361-368. doi: 10.1038/s41433-021-01668-x. Epub 2021 Jul 14 [PubMed PMID: 34262161]
Santana CP, Matter BA, Patil MA, Silva-Cunha A, Kompella UB. Corneal Permeability and Uptake of Twenty-Five Drugs: Species Comparison and Quantitative Structure-Permeability Relationships. Pharmaceutics. 2023 Jun 2:15(6):. doi: 10.3390/pharmaceutics15061646. Epub 2023 Jun 2 [PubMed PMID: 37376094]
Agrahari V, Mandal A, Agrahari V, Trinh HM, Joseph M, Ray A, Hadji H, Mitra R, Pal D, Mitra AK. A comprehensive insight on ocular pharmacokinetics. Drug delivery and translational research. 2016 Dec:6(6):735-754 [PubMed PMID: 27798766]
Nakano M, Lockhart CM, Kelly EJ, Rettie AE. Ocular cytochrome P450s and transporters: roles in disease and endobiotic and xenobiotic disposition. Drug metabolism reviews. 2014 Aug:46(3):247-60. doi: 10.3109/03602532.2014.921190. Epub 2014 May 26 [PubMed PMID: 24856391]
Wang L, Zhou MB, Zhang H. The Emerging Role of Topical Ocular Drugs to Target the Posterior Eye. Ophthalmology and therapy. 2021 Sep:10(3):465-494. doi: 10.1007/s40123-021-00365-y. Epub 2021 Jul 4 [PubMed PMID: 34218424]
Paley GL, Lubniewski AJ, Reidy JJ, Farooq AV. Toxic Keratoconjunctivitis. Eye & contact lens. 2018 Sep:44 Suppl 1():S8-S15. doi: 10.1097/ICL.0000000000000392. Epub [PubMed PMID: 28445208]
La Rosa M, Lionetti E, Reibaldi M, Russo A, Longo A, Leonardi S, Tomarchio S, Avitabile T, Reibaldi A. Allergic conjunctivitis: a comprehensive review of the literature. Italian journal of pediatrics. 2013 Mar 14:39():18. doi: 10.1186/1824-7288-39-18. Epub 2013 Mar 14 [PubMed PMID: 23497516]
. The epidemiology of dry eye disease: report of the Epidemiology Subcommittee of the International Dry Eye WorkShop (2007). The ocular surface. 2007 Apr:5(2):93-107 [PubMed PMID: 17508117]
Milner MS, Beckman KA, Luchs JI, Allen QB, Awdeh RM, Berdahl J, Boland TS, Buznego C, Gira JP, Goldberg DF, Goldman D, Goyal RK, Jackson MA, Katz J, Kim T, Majmudar PA, Malhotra RP, McDonald MB, Rajpal RK, Raviv T, Rowen S, Shamie N, Solomon JD, Stonecipher K, Tauber S, Trattler W, Walter KA, Waring GO 4th, Weinstock RJ, Wiley WF, Yeu E. Dysfunctional tear syndrome: dry eye disease and associated tear film disorders - new strategies for diagnosis and treatment. Current opinion in ophthalmology. 2017 Jan:27 Suppl 1(Suppl 1):3-47. doi: 10.1097/01.icu.0000512373.81749.b7. Epub [PubMed PMID: 28099212]
Level 3 (low-level) evidenceGolden MI, Meyer JJ, Patel BC. Dry Eye Syndrome. StatPearls. 2024 Jan:(): [PubMed PMID: 29262012]
Krishna MT, Subramanian A, Adderley NJ, Zemedikun DT, Gkoutos GV, Nirantharakumar K. Allergic diseases and long-term risk of autoimmune disorders: longitudinal cohort study and cluster analysis. The European respiratory journal. 2019 Nov:54(5):. pii: 1900476. doi: 10.1183/13993003.00476-2019. Epub 2019 Nov 14 [PubMed PMID: 31413164]
Patel S, Mittal R, Kumar N, Galor A. The environment and dry eye-manifestations, mechanisms, and more. Frontiers in toxicology. 2023:5():1173683. doi: 10.3389/ftox.2023.1173683. Epub 2023 Aug 23 [PubMed PMID: 37681211]
Fu L, Patel BC. Lagophthalmos. StatPearls. 2025 Jan:(): [PubMed PMID: 32809496]
Byrd LB, Gurnani B, Martin N. Corneal Ulcer. StatPearls. 2025 Jan:(): [PubMed PMID: 30969511]
Messmer EM. The pathophysiology, diagnosis, and treatment of dry eye disease. Deutsches Arzteblatt international. 2015 Jan 30:112(5):71-81; quiz 82. doi: 10.3238/arztebl.2015.0071. Epub [PubMed PMID: 25686388]
Anguria P, Carmichael T, Ntuli S, Kitinya J. Chronic inflammatory cells and damaged limbal cells in pterygium. African health sciences. 2013 Sep:13(3):725-30. doi: 10.4314/ahs.v13i3.29. Epub [PubMed PMID: 24250313]
Srinivas SP, Rao SK. Ocular surface staining: Current concepts and techniques. Indian journal of ophthalmology. 2023 Apr:71(4):1080-1089. doi: 10.4103/ijo.IJO_2137_22. Epub [PubMed PMID: 37026238]
Leibowitz HM. The red eye. The New England journal of medicine. 2000 Aug 3:343(5):345-51 [PubMed PMID: 10922425]
Hwang HB, Ku YH, Kim EC, Kim HS, Kim MS, Hwang HS. Easy and effective test to evaluate tear-film stability for self-diagnosis of dry eye syndrome: blinking tolerance time (BTT). BMC ophthalmology. 2020 Nov 4:20(1):438. doi: 10.1186/s12886-020-01686-5. Epub 2020 Nov 4 [PubMed PMID: 33148200]
Lanza NL, Valenzuela F, Perez VL, Galor A. The Matrix Metalloproteinase 9 Point-of-Care Test in Dry Eye. The ocular surface. 2016 Apr:14(2):189-95. doi: 10.1016/j.jtos.2015.10.004. Epub 2016 Feb 2 [PubMed PMID: 26850527]
Elnifro EM, Cooper RJ, Klapper PE, Yeo AC, Tullo AB. Multiplex polymerase chain reaction for diagnosis of viral and chlamydial keratoconjunctivitis. Investigative ophthalmology & visual science. 2000 Jun:41(7):1818-22 [PubMed PMID: 10845603]
Ansotegui IJ, Melioli G, Canonica GW, Caraballo L, Villa E, Ebisawa M, Passalacqua G, Savi E, Ebo D, Gómez RM, Luengo Sánchez O, Oppenheimer JJ, Jensen-Jarolim E, Fischer DA, Haahtela T, Antila M, Bousquet JJ, Cardona V, Chiang WC, Demoly PM, DuBuske LM, Ferrer Puga M, Gerth van Wijk R, González Díaz SN, Gonzalez-Estrada A, Jares E, Kalpaklioğlu AF, Kase Tanno L, Kowalski ML, Ledford DK, Monge Ortega OP, Morais Almeida M, Pfaar O, Poulsen LK, Pawankar R, Renz HE, Romano AG, Rosário Filho NA, Rosenwasser L, Sánchez Borges MA, Scala E, Senna GE, Sisul JC, Tang MLK, Thong BY, Valenta R, Wood RA, Zuberbier T. IgE allergy diagnostics and other relevant tests in allergy, a World Allergy Organization position paper. The World Allergy Organization journal. 2020 Feb:13(2):100080. doi: 10.1016/j.waojou.2019.100080. Epub 2020 Feb 25 [PubMed PMID: 32128023]
Nosal RS, Superville SS, Amraei R, Varacallo MA. Biochemistry, Antinuclear Antibodies (ANA). StatPearls. 2025 Jan:(): [PubMed PMID: 30725756]
Singh R, Joseph A, Umapathy T, Tint NL, Dua HS. Impression cytology of the ocular surface. The British journal of ophthalmology. 2005 Dec:89(12):1655-9 [PubMed PMID: 16299150]
Mahmoud MSE, Hamid MA, Abdelkader MF. Anterior Segment Optical Coherence Tomography of Tear Film and Cornea in Systemic Lupus Erythematosus Patients. Clinical ophthalmology (Auckland, N.Z.). 2021:15():3391-3399. doi: 10.2147/OPTH.S323673. Epub 2021 Aug 12 [PubMed PMID: 34408395]
Kaur K, Stokkermans TJ. Meibomian Gland Disease. StatPearls. 2025 Jan:(): [PubMed PMID: 35593799]
Cruzat A, Qazi Y, Hamrah P. In Vivo Confocal Microscopy of Corneal Nerves in Health and Disease. The ocular surface. 2017 Jan:15(1):15-47. doi: 10.1016/j.jtos.2016.09.004. Epub 2016 Oct 19 [PubMed PMID: 27771327]
Wolffsohn JS, Arita R, Chalmers R, Djalilian A, Dogru M, Dumbleton K, Gupta PK, Karpecki P, Lazreg S, Pult H, Sullivan BD, Tomlinson A, Tong L, Villani E, Yoon KC, Jones L, Craig JP. TFOS DEWS II Diagnostic Methodology report. The ocular surface. 2017 Jul:15(3):539-574. doi: 10.1016/j.jtos.2017.05.001. Epub 2017 Jul 20 [PubMed PMID: 28736342]
Høvding G. Acute bacterial conjunctivitis. Acta ophthalmologica. 2008 Feb:86(1):5-17 [PubMed PMID: 17970823]
Martínez-Aguado P, Serna-Gallego A, Marrugal-Lorenzo JA, Gómez-Marín I, Sánchez-Céspedes J. Antiadenovirus drug discovery: potential targets and evaluation methodologies. Drug discovery today. 2015 Oct:20(10):1235-42. doi: 10.1016/j.drudis.2015.07.007. Epub 2015 Jul 23 [PubMed PMID: 26210958]
Bielory BP, O'Brien TP, Bielory L. Management of seasonal allergic conjunctivitis: guide to therapy. Acta ophthalmologica. 2012 Aug:90(5):399-407. doi: 10.1111/j.1755-3768.2011.02272.x. Epub 2011 Nov 8 [PubMed PMID: 22067457]
Chen JJ, Applebaum DS, Sun GS, Pflugfelder SC. Atopic keratoconjunctivitis: A review. Journal of the American Academy of Dermatology. 2014 Mar:70(3):569-75. doi: 10.1016/j.jaad.2013.10.036. Epub 2013 Dec 15 [PubMed PMID: 24342754]
Gokhale NS. Systematic approach to managing vernal keratoconjunctivitis in clinical practice: Severity grading system and a treatment algorithm. Indian journal of ophthalmology. 2016 Feb:64(2):145-8. doi: 10.4103/0301-4738.179727. Epub [PubMed PMID: 27050351]
Level 1 (high-level) evidenceGris O, Plazas A, Lerma E, Güell JL, Pelegrín L, Elíes D. Conjunctival resection with and without amniotic membrane graft for the treatment of superior limbic keratoconjunctivitis. Cornea. 2010 Sep:29(9):1025-30. doi: 10.1097/ICO.0b013e3181d1d1cc. Epub [PubMed PMID: 20539210]
Level 2 (mid-level) evidenceFraunfelder FW. Liquid nitrogen cryotherapy of superior limbic keratoconjunctivitis. American journal of ophthalmology. 2009 Feb:147(2):234-238.e1. doi: 10.1016/j.ajo.2008.07.047. Epub 2008 Oct 4 [PubMed PMID: 18835475]
Goto E, Shimmura S, Shimazaki J, Tsubota K. Treatment of superior limbic keratoconjunctivitis by application of autologous serum. Cornea. 2001 Nov:20(8):807-10 [PubMed PMID: 11685056]
Level 3 (low-level) evidenceGilani CJ, Yang A, Yonkers M, Boysen-Osborn M. Differentiating Urgent and Emergent Causes of Acute Red Eye for the Emergency Physician. The western journal of emergency medicine. 2017 Apr:18(3):509-517. doi: 10.5811/westjem.2016.12.31798. Epub 2017 Mar 3 [PubMed PMID: 28435504]
Mahabadi N, Gurnani B, Czyz CN. Bacterial Endophthalmitis. StatPearls. 2025 Jan:(): [PubMed PMID: 31424768]
Gurnani B, Kaur K, Chaudhary S, Balakrishnan H. Ophthalmic manifestations of monkeypox infection. Indian journal of ophthalmology. 2023 May:71(5):1687-1697. doi: 10.4103/ijo.IJO_2032_22. Epub [PubMed PMID: 37203020]
Nelson JD. Superior limbic keratoconjunctivitis (SLK). Eye (London, England). 1989:3 ( Pt 2)():180-9 [PubMed PMID: 2695351]
Gurnani B, Kaur K. Contact Lens–Related Complications. StatPearls. 2025 Jan:(): [PubMed PMID: 36512659]
Chang JH, McCluskey PJ. Ocular cicatricial pemphigoid: manifestations and management. Current allergy and asthma reports. 2005 Jul:5(4):333-8 [PubMed PMID: 15967079]
Tóth G, Lukács A, Schirra F, Sándor GL, Killik P, Maneschg OA, Nagy ZZ, Szentmáry N. Ophthalmic Aspects of Stevens-Johnson Syndrome and Toxic Epidermal Necrolysis: A Narrative Review. Ophthalmology and therapy. 2023 Aug:12(4):1795-1811. doi: 10.1007/s40123-023-00725-w. Epub 2023 May 4 [PubMed PMID: 37140876]
Level 3 (low-level) evidenceSchonberg S, Stokkermans TJ. Episcleritis. StatPearls. 2025 Jan:(): [PubMed PMID: 30521217]
Shahraki T, Arabi A, Feizi S. Pterygium: an update on pathophysiology, clinical features, and management. Therapeutic advances in ophthalmology. 2021 Jan-Dec:13():25158414211020152. doi: 10.1177/25158414211020152. Epub 2021 May 31 [PubMed PMID: 34104871]
Level 3 (low-level) evidenceMurugan SRB, Sanjay S, Somanath A, Mahendradas P, Patil A, Kaur K, Gurnani B. Artificial Intelligence in Uveitis: Innovations in Diagnosis and Therapeutic Strategies. Clinical ophthalmology (Auckland, N.Z.). 2024:18():3753-3766. doi: 10.2147/OPTH.S495307. Epub 2024 Dec 14 [PubMed PMID: 39703602]
Craig JP,Nichols KK,Akpek EK,Caffery B,Dua HS,Joo CK,Liu Z,Nelson JD,Nichols JJ,Tsubota K,Stapleton F, TFOS DEWS II Definition and Classification Report. The ocular surface. 2017 Jul [PubMed PMID: 28736335]
Holland EJ, Luchs J, Karpecki PM, Nichols KK, Jackson MA, Sall K, Tauber J, Roy M, Raychaudhuri A, Shojaei A. Lifitegrast for the Treatment of Dry Eye Disease: Results of a Phase III, Randomized, Double-Masked, Placebo-Controlled Trial (OPUS-3). Ophthalmology. 2017 Jan:124(1):53-60. doi: 10.1016/j.ophtha.2016.09.025. Epub 2016 Oct 27 [PubMed PMID: 28079022]
Level 1 (high-level) evidencePeriman LM, Mah FS, Karpecki PM. A Review of the Mechanism of Action of Cyclosporine A: The Role of Cyclosporine A in Dry Eye Disease and Recent Formulation Developments. Clinical ophthalmology (Auckland, N.Z.). 2020:14():4187-4200. doi: 10.2147/OPTH.S279051. Epub 2020 Dec 2 [PubMed PMID: 33299295]
Azuara-Blanco A, Pillai CT, Dua HS. Amniotic membrane transplantation for ocular surface reconstruction. The British journal of ophthalmology. 1999 Apr:83(4):399-402 [PubMed PMID: 10434859]
Hussain M, Shtein RM, Pistilli M, Maguire MG, Oydanich M, Asbell PA, DREAM Study Research Group. The Dry Eye Assessment and Management (DREAM) extension study - A randomized clinical trial of withdrawal of supplementation with omega-3 fatty acid in patients with dry eye disease. The ocular surface. 2020 Jan:18(1):47-55. doi: 10.1016/j.jtos.2019.08.002. Epub 2019 Aug 16 [PubMed PMID: 31425752]
Level 1 (high-level) evidenceFukuda K, Kishimoto T, Sumi T, Yamashiro K, Ebihara N. Biologics for allergy: therapeutic potential for ocular allergic diseases and adverse effects on the eye. Allergology international : official journal of the Japanese Society of Allergology. 2023 Apr:72(2):234-244. doi: 10.1016/j.alit.2022.09.005. Epub 2022 Nov 1 [PubMed PMID: 36333219]
Okumura N, Kinoshita S, Koizumi N. The Role of Rho Kinase Inhibitors in Corneal Endothelial Dysfunction. Current pharmaceutical design. 2017:23(4):660-666. doi: 10.2174/1381612822666161205110027. Epub [PubMed PMID: 27917718]
Niu Y, Ji J, Yao K, Fu Q. Regenerative treatment of ophthalmic diseases with stem cells: Principles, progress, and challenges. Advances in ophthalmology practice and research. 2024 May-Jun:4(2):52-64. doi: 10.1016/j.aopr.2024.02.001. Epub 2024 Feb 28 [PubMed PMID: 38586868]
Level 3 (low-level) evidenceDohlman TH, Ciralsky JB, Lai EC. Tear film assessments for the diagnosis of dry eye. Current opinion in allergy and clinical immunology. 2016 Oct:16(5):487-91. doi: 10.1097/ACI.0000000000000307. Epub [PubMed PMID: 27585060]
Level 3 (low-level) evidenceKoh S, Rao SK, Srinivas SP, Tong L, Young AL. Evaluation of ocular surface and tear function - A review of current approaches for dry eye. Indian journal of ophthalmology. 2022 Jun:70(6):1883-1891. doi: 10.4103/ijo.IJO_1804_21. Epub [PubMed PMID: 35647953]
Chester T, Garg SS, Johnston J, Ayers B, Gupta P. How Can We Best Diagnose Severity Levels of Dry Eye Disease: Current Perspectives. Clinical ophthalmology (Auckland, N.Z.). 2023:17():1587-1604. doi: 10.2147/OPTH.S388289. Epub 2023 Jun 5 [PubMed PMID: 37304329]
Level 3 (low-level) evidenceAgarwal P, Craig JP, Rupenthal ID. Formulation Considerations for the Management of Dry Eye Disease. Pharmaceutics. 2021 Feb 3:13(2):. doi: 10.3390/pharmaceutics13020207. Epub 2021 Feb 3 [PubMed PMID: 33546193]
Ben-Eli H, Solomon A. Topical antihistamines, mast cell stabilizers, and dual-action agents in ocular allergy: current trends. Current opinion in allergy and clinical immunology. 2018 Oct:18(5):411-416. doi: 10.1097/ACI.0000000000000473. Epub [PubMed PMID: 30020258]
Level 3 (low-level) evidenceCabrera-Aguas M, Khoo P, Watson SL. Infectious keratitis: A review. Clinical & experimental ophthalmology. 2022 Jul:50(5):543-562. doi: 10.1111/ceo.14113. Epub 2022 Jun 3 [PubMed PMID: 35610943]
Chen Z, Lao HY, Liang L. Update on the application of amniotic membrane in immune-related ocular surface diseases. Taiwan journal of ophthalmology. 2021 Apr-Jun:11(2):132-140. doi: 10.4103/tjo.tjo_16_21. Epub 2021 Jun 2 [PubMed PMID: 34295618]
Pan Q, Angelina A, Marrone M, Stark WJ, Akpek EK. Autologous serum eye drops for dry eye. The Cochrane database of systematic reviews. 2017 Feb 28:2(2):CD009327. doi: 10.1002/14651858.CD009327.pub3. Epub 2017 Feb 28 [PubMed PMID: 28245347]
Level 1 (high-level) evidenceFeroze KB, Zeppieri M, Khazaeni L. Steroid-Induced Glaucoma. StatPearls. 2024 Jan:(): [PubMed PMID: 28613653]
Schultz C. Safety and efficacy of cyclosporine in the treatment of chronic dry eye. Ophthalmology and eye diseases. 2014:6():37-42. doi: 10.4137/OED.S16067. Epub 2014 Jun 24 [PubMed PMID: 25002818]
Srikanth K, Sridhanya V. Comment on efficacy, safety, and tolerability of Lifitegrast 5% eye drops: A randomized, double-blind, active-controlled trial in Indian patients with dry eye disease. Indian journal of ophthalmology. 2025 Jan 1:73(1):154. doi: 10.4103/IJO.IJO_2424_24. Epub 2024 Dec 23 [PubMed PMID: 39723862]
Level 1 (high-level) evidenceGurnani B, Kaur K. Contact Lenses. StatPearls. 2025 Jan:(): [PubMed PMID: 35593861]
Walker HK, Hall WD, Hurst JW, Philp JR. Allergic Drug Reactions. Clinical Methods: The History, Physical, and Laboratory Examinations. 1990:(): [PubMed PMID: 21250169]
Chandran C, Santra M, Rubin E, Geary ML, Yam GH. Regenerative Therapy for Corneal Scarring Disorders. Biomedicines. 2024 Mar 14:12(3):. doi: 10.3390/biomedicines12030649. Epub 2024 Mar 14 [PubMed PMID: 38540264]
NaPier E, Camacho M, McDevitt TF, Sweeney AR. Neurotrophic keratopathy: current challenges and future prospects. Annals of medicine. 2022 Dec:54(1):666-673. doi: 10.1080/07853890.2022.2045035. Epub [PubMed PMID: 35243932]
Nieuwlaat R, Wilczynski N, Navarro T, Hobson N, Jeffery R, Keepanasseril A, Agoritsas T, Mistry N, Iorio A, Jack S, Sivaramalingam B, Iserman E, Mustafa RA, Jedraszewski D, Cotoi C, Haynes RB. Interventions for enhancing medication adherence. The Cochrane database of systematic reviews. 2014 Nov 20:2014(11):CD000011. doi: 10.1002/14651858.CD000011.pub4. Epub 2014 Nov 20 [PubMed PMID: 25412402]
Level 1 (high-level) evidenceOakley AM, Krishnamurthy K. Stevens-Johnson Syndrome. StatPearls. 2025 Jan:(): [PubMed PMID: 29083827]
Gurnani B, Natarajan R, Mohan M, Kaur K. Breaking-Down Barriers: Proposal of Using Cellulose Biosynthesis Inhibitors and Cellulase Enzyme as a Novel Treatment Modality for Vision Threatening Pythium Insidiosum Keratitis. Clinical ophthalmology (Auckland, N.Z.). 2024:18():765-776. doi: 10.2147/OPTH.S450665. Epub 2024 Mar 11 [PubMed PMID: 38495678]
Udell IJ, Kenyon KR, Sawa M, Dohlman CH. Treatment of superior limbic keratoconjunctivitis by thermocauterization of the superior bulbar conjunctiva. Ophthalmology. 1986 Feb:93(2):162-6 [PubMed PMID: 3951822]
Golhait P, Peseyie R. Persistent Epithelial Defect. StatPearls. 2025 Jan:(): [PubMed PMID: 34424633]
Messmer EM, Ahmad S, Benitez Del Castillo JM, Mrukwa-Kominek E, Rolando M, Vitovska O, Baudouin C, a panel of European dry eye disease experts. Management of inflammation in dry eye disease: Recommendations from a European panel of experts. European journal of ophthalmology. 2023 May:33(3):1294-1307. doi: 10.1177/11206721221141481. Epub 2022 Dec 5 [PubMed PMID: 36471573]
Aremu TO, Oluwole OE, Adeyinka KO, Schommer JC. Medication Adherence and Compliance: Recipe for Improving Patient Outcomes. Pharmacy (Basel, Switzerland). 2022 Aug 28:10(5):. doi: 10.3390/pharmacy10050106. Epub 2022 Aug 28 [PubMed PMID: 36136839]
Thulasi P, Djalilian AR. Update in Current Diagnostics and Therapeutics of Dry Eye Disease. Ophthalmology. 2017 Nov:124(11S):S27-S33. doi: 10.1016/j.ophtha.2017.07.022. Epub [PubMed PMID: 29055359]
Sekeroglu HT, Simsek F, Erdem E, Harbiyeli II, Yagmur M, Ersoz R. Coexistence of ocular cicatricial pemphigoid with Stevens Johnson syndrome. International journal of ophthalmology. 2013:6(3):411-2. doi: 10.3980/j.issn.2222-3959.2013.03.30. Epub 2013 Jun 18 [PubMed PMID: 23826544]
Austin A, Lietman T, Rose-Nussbaumer J. Update on the Management of Infectious Keratitis. Ophthalmology. 2017 Nov:124(11):1678-1689. doi: 10.1016/j.ophtha.2017.05.012. Epub 2017 Sep 21 [PubMed PMID: 28942073]
Vaidyanathan U, Hopping GC, Liu HY, Somani AN, Ronquillo YC, Hoopes PC, Moshirfar M. Persistent Corneal Epithelial Defects: A Review Article. Medical hypothesis, discovery & innovation ophthalmology journal. 2019 Fall:8(3):163-176 [PubMed PMID: 31598519]
Slovis TL, Shkolnik A, Haller JO. Focal areas of increased echogenicity within the brain. Journal of ultrasound in medicine : official journal of the American Institute of Ultrasound in Medicine. 1989 Jan:8(1):31-7 [PubMed PMID: 2644440]
Sharif Z, Sharif W. Corneal neovascularization: updates on pathophysiology, investigations & management. Romanian journal of ophthalmology. 2019 Jan-Mar:63(1):15-22 [PubMed PMID: 31198893]
Cameron JA. Shield ulcers and plaques of the cornea in vernal keratoconjunctivitis. Ophthalmology. 1995 Jun:102(6):985-93 [PubMed PMID: 7777308]
Level 2 (mid-level) evidenceIsamat F, Miranda AM, Bartumeus F, Prat J. Genetic implications of familial brain tumors. Journal of neurosurgery. 1974 Nov:41(5):573-5 [PubMed PMID: 4424633]
Swarup A, Ta CN, Wu AY. Molecular mechanisms and treatments for ocular symblephara. Survey of ophthalmology. 2022 Jan-Feb:67(1):19-30. doi: 10.1016/j.survophthal.2021.04.008. Epub 2021 Apr 29 [PubMed PMID: 33932469]
Level 3 (low-level) evidenceMoshirfar M, Masud M, Harvey DH, Payne C, Bruce E, Ronquillo YC, Hoopes PC. The Multifold Etiologies of Limbal Stem Cell Deficiency: A Comprehensive Review on the Etiologies and Additional Treatment Options for Limbal Stem Cell Deficiency. Journal of clinical medicine. 2023 Jun 30:12(13):. doi: 10.3390/jcm12134418. Epub 2023 Jun 30 [PubMed PMID: 37445454]
Gurnani B, Feroze KB, Patel BC. Neurotrophic Keratitis. StatPearls. 2025 Jan:(): [PubMed PMID: 28613758]
Galor A, Levitt RC, Felix ER, Sarantopoulos CD. What can photophobia tell us about dry eye? Expert review of ophthalmology. 2016:11(5):321-324. doi: 10.1080/17469899.2016.1222905. Epub 2016 Aug 23 [PubMed PMID: 28845186]
Fu L, Jones S. Peripheral Ulcerative Keratitis. StatPearls. 2025 Jan:(): [PubMed PMID: 34662070]
Gurnani B, Kaur K. Therapeutic Keratoplasty. StatPearls. 2025 Jan:(): [PubMed PMID: 37276297]
Abdelghany AA, Alio JL, AttaAllah HR. Role of Anterior Segment Optical Coherence Tomography in Staging and Evaluation of Treatment Response in Infectious Keratitis. Cornea. 2024 Oct 1:43(10):1216-1222. doi: 10.1097/ICO.0000000000003466. Epub 2024 Jan 30 [PubMed PMID: 38289764]
Liu SH, Saldanha IJ, Abraham AG, Rittiphairoj T, Hauswirth S, Gregory D, Ifantides C, Li T. Topical corticosteroids for dry eye. The Cochrane database of systematic reviews. 2022 Oct 21:10(10):CD015070. doi: 10.1002/14651858.CD015070.pub2. Epub 2022 Oct 21 [PubMed PMID: 36269562]
Level 1 (high-level) evidenceHorn R, Hendrix JM, Kramer J. Postoperative Pain Control. StatPearls. 2025 Jan:(): [PubMed PMID: 31335018]
Alebrahim MA, Bakkar MM, Al Darayseh A, Msameh A, Jarrar D, Aljabari S, Khater W. Awareness and Knowledge of the Effect of Ultraviolet (UV) Radiation on the Eyes and the Relevant Protective Practices: A Cross-Sectional Study from Jordan. Healthcare (Basel, Switzerland). 2022 Nov 30:10(12):. doi: 10.3390/healthcare10122414. Epub 2022 Nov 30 [PubMed PMID: 36553938]
Level 2 (mid-level) evidenceFazal MI, Patel BC. Blepharoconjunctivitis. StatPearls. 2025 Jan:(): [PubMed PMID: 32644328]
Malhotra A, Mars JA, Baker J. Group Therapy. StatPearls. 2025 Jan:(): [PubMed PMID: 31747219]
Geerling G, Maclennan S, Hartwig D. Autologous serum eye drops for ocular surface disorders. The British journal of ophthalmology. 2004 Nov:88(11):1467-74 [PubMed PMID: 15489495]
Gurnani B, Kaur K. Changing drug regimen during COVID-19 pandemic lockdown: An experience from a Tertiary Eye Care Hospital as a cornea specialist. Indian journal of pharmacology. 2020 Sep-Oct:52(5):435-436. doi: 10.4103/ijp.IJP_688_20. Epub [PubMed PMID: 33283776]
Harigai M, Kaneko Y, Tanaka E, Hirata S, Kameda H, Kaneko K, Kishimoto M, Kohno M, Kojima M, Kojima T, Morinobu A, Nakajima A, Sugihara T, Fusama M, Yajima N, Yanai R, Kawahito Y. 2024 Update of the Japan College of Rheumatology Clinical Practice Guidelines for the Management of Rheumatoid Arthritis - secondary publication. Modern rheumatology. 2025 Jan 17:():. pii: roaf006. doi: 10.1093/mr/roaf006. Epub 2025 Jan 17 [PubMed PMID: 39820350]
Level 1 (high-level) evidenceAlosaimi B, Awadalla M, Alturaiki W, Chen Z, Zhang Z, Zhu A, Rebh F, Alshukairi AN, Zhao J, Alkadi HS. A 10 Year Long-Lived Cellular and Humoral MERS-CoV Immunity Cross-Recognizing the Wild-Type and Variants of SARS-CoV-2: A Potential One-Way MERS-CoV Cross-Protection Toward a Pan-Coronavirus Vaccine. Journal of medical virology. 2025 Jan:97(1):e70071. doi: 10.1002/jmv.70071. Epub [PubMed PMID: 39822038]
Haskell Hanisch BK, Frohm ML. A Review of Current Treatment Options for Stage III and Stage IV Metastatic Melanoma. South Dakota medicine : the journal of the South Dakota State Medical Association. 2024 Oct:77(10):466-469 [PubMed PMID: 39821259]
Qi Y, Zhang Y, Liu X, Li Y, Zheng R. Preliminary comparison of net gain in final adult height of girls with early menarche treated with or without gonadotropin-releasing hormone agonist. Translational pediatrics. 2024 Dec 31:13(12):2204-2213. doi: 10.21037/tp-24-348. Epub 2024 Dec 27 [PubMed PMID: 39823015]
Level 2 (mid-level) evidenceKamenov K, Twomey C, Cabello M, Prina AM, Ayuso-Mateos JL. The efficacy of psychotherapy, pharmacotherapy and their combination on functioning and quality of life in depression: a meta-analysis. Psychological medicine. 2017 Feb:47(3):414-425. doi: 10.1017/S0033291716002774. Epub 2016 Oct 26 [PubMed PMID: 27780478]
Level 1 (high-level) evidenceMohamed-Noriega K, Loya-Garcia D, Vera-Duarte GR, Morales-Wong F, Ortiz-Morales G, Navas A, Graue-Hernandez EO, Ramirez-Miranda A. Ocular Rosacea: An Updated Review. Cornea. 2025 Jan 14:():. doi: 10.1097/ICO.0000000000003785. Epub 2025 Jan 14 [PubMed PMID: 39808113]
Romm KL, Skoge M, Barrett EA, Berentzen LC, Bergsager D, Fugelli P, Bjella T, Gardsjord ES, Kling K, Kruse SH, Kværner KJ, Melle I, Mork E, Ihler HM, Rognli EB, Simonsen C, Værnes TG, Aminoff SR. A Mobile Health Intervention to Support Collaborative Decision-Making in Mental Health Care: Development and Usability. JMIR formative research. 2025 Jan 17:9():e57614. doi: 10.2196/57614. Epub 2025 Jan 17 [PubMed PMID: 39823632]
Lievens CW, Cilimberg KC, Moore A. Contact lens care tips for patients: an optometrist's perspective. Clinical optometry. 2017:9():113-121. doi: 10.2147/OPTO.S139651. Epub 2017 Aug 11 [PubMed PMID: 30214367]
Level 3 (low-level) evidenceCong Y, Zhang Y, Han Y, Wu Y, Wang D, Zhang B. Recommendations for nutritional supplements for dry eye disease: current advances. Frontiers in pharmacology. 2024:15():1388787. doi: 10.3389/fphar.2024.1388787. Epub 2024 May 30 [PubMed PMID: 38873421]
Level 3 (low-level) evidenceHochfelder JL, Ponda P. Allergen immunotherapy: routes, safety, efficacy, and mode of action. ImmunoTargets and therapy. 2013:2():61-71. doi: 10.2147/ITT.S31467. Epub 2013 Jul 22 [PubMed PMID: 27471689]
Tabi K, Randhawa AS, Choi F, Mithani Z, Albers F, Schnieder M, Nikoo M, Vigo D, Jang K, Demlova R, Krausz M. Mobile Apps for Medication Management: Review and Analysis. JMIR mHealth and uHealth. 2019 Sep 11:7(9):e13608. doi: 10.2196/13608. Epub 2019 Sep 11 [PubMed PMID: 31512580]
Ricci L, Villegente J, Loyal D, Ayav C, Kivits J, Rat AC. Tailored patient therapeutic educational interventions: A patient-centred communication model. Health expectations : an international journal of public participation in health care and health policy. 2022 Feb:25(1):276-289. doi: 10.1111/hex.13377. Epub 2021 Nov 24 [PubMed PMID: 34816546]
Gurnani B, Kaur K. Recent Advances in Refractive Surgery: An Overview. Clinical ophthalmology (Auckland, N.Z.). 2024:18():2467-2472. doi: 10.2147/OPTH.S481421. Epub 2024 Sep 2 [PubMed PMID: 39246558]
Level 3 (low-level) evidenceSchnider C, Yuen L, Rampat R, Zhu D, Dhallu S, Trinh T, Gurnani B, Abdelmaksoud A, Bhogal-Bhamra G, Wolffsohn JS, Naroo SA. BCLA CLEAR presbyopia: Management with intraocular lenses. Contact lens & anterior eye : the journal of the British Contact Lens Association. 2024 Aug:47(4):102253. doi: 10.1016/j.clae.2024.102253. Epub 2024 Jul 2 [PubMed PMID: 39068141]
Solano D, Fu L, Czyz CN. Viral Conjunctivitis. StatPearls. 2025 Jan:(): [PubMed PMID: 29262100]
Paulbuddhe V, Addya S, Gurnani B, Singh D, Tripathy K, Chawla R. Sympathetic Ophthalmia: Where Do We Currently Stand on Treatment Strategies? Clinical ophthalmology (Auckland, N.Z.). 2021:15():4201-4218. doi: 10.2147/OPTH.S289688. Epub 2021 Oct 20 [PubMed PMID: 34707340]
Kaur K, Gurnani B, Gireesh P. Hearing Loss and Decreasing Visual Acuity. Journal of pediatric ophthalmology and strabismus. 2019 Jul 1:56(4):212. doi: 10.3928/01913913-20190520-01. Epub [PubMed PMID: 31322708]
Kaur K, Gurnani B, Devy N. Atypical optic neuritis - a case with a new surprise every visit. GMS ophthalmology cases. 2020:10():Doc11. doi: 10.3205/oc000138. Epub 2020 Feb 27 [PubMed PMID: 32269909]
Level 3 (low-level) evidenceByber K, Radtke T, Norbäck D, Hitzke C, Imo D, Schwenkglenks M, Puhan MA, Dressel H, Mutsch M. Humidification of indoor air for preventing or reducing dryness symptoms or upper respiratory infections in educational settings and at the workplace. The Cochrane database of systematic reviews. 2021 Dec 10:12(12):CD012219. doi: 10.1002/14651858.CD012219.pub2. Epub 2021 Dec 10 [PubMed PMID: 34891215]
Level 1 (high-level) evidencePassali D, Cingi C, Staffa P, Passali F, Muluk NB, Bellussi ML. The International Study of the Allergic Rhinitis Survey: outcomes from 4 geographical regions. Asia Pacific allergy. 2018 Jan:8(1):e7. doi: 10.5415/apallergy.2018.8.e7. Epub 2018 Jan 25 [PubMed PMID: 29423374]
Level 3 (low-level) evidenceLevine J, Snyder RW. Practical ophthalmic microbiology. Journal of ophthalmic nursing & technology. 1999 Mar-Apr:18(2):50-9; quiz 74-5 [PubMed PMID: 10409996]
Jefferis J, Perera R, Everitt H, van Weert H, Rietveld R, Glasziou P, Rose P. Acute infective conjunctivitis in primary care: who needs antibiotics? An individual patient data meta-analysis. The British journal of general practice : the journal of the Royal College of General Practitioners. 2011 Sep:61(590):e542-8. doi: 10.3399/bjgp11X593811. Epub [PubMed PMID: 22152728]
Level 1 (high-level) evidenceCooke L, Gemmill R, Grant M. Advanced practice nurses core competencies: a framework for developing and testing an advanced practice nurse discharge intervention. Clinical nurse specialist CNS. 2008 Sep-Oct:22(5):218-25. doi: 10.1097/01.NUR.0000325366.15927.2d. Epub [PubMed PMID: 18753879]
Vos JFJ, Boonstra A, Kooistra A, Seelen M, van Offenbeek M. The influence of electronic health record use on collaboration among medical specialties. BMC health services research. 2020 Jul 22:20(1):676. doi: 10.1186/s12913-020-05542-6. Epub 2020 Jul 22 [PubMed PMID: 32698807]
Wong CL, Chan CWH, Zhang M, Cheung YT, Chow KM, Li CK, Li WHC, Brauer E, Chen Y. Care coordination models for transition and long-term follow-up among childhood cancer survivors: a scoping review. BMJ open. 2024 Aug 19:14(8):e087343. doi: 10.1136/bmjopen-2024-087343. Epub 2024 Aug 19 [PubMed PMID: 39160096]
Level 2 (mid-level) evidenceEdgman-Levitan S, Schoenbaum SC. Patient-centered care: achieving higher quality by designing care through the patient's eyes. Israel journal of health policy research. 2021 Mar 5:10(1):21. doi: 10.1186/s13584-021-00459-9. Epub 2021 Mar 5 [PubMed PMID: 33673875]
Level 2 (mid-level) evidenceBalamurugan S, Das D, Hasanreisoglu M, Toy BC, Akhter M, Anuradha VK, Anthony E, Gurnani B, Kaur K. Interleukins and cytokine biomarkers in uveitis. Indian journal of ophthalmology. 2020 Sep:68(9):1750-1763. doi: 10.4103/ijo.IJO_564_20. Epub [PubMed PMID: 32823391]
Gurnani B, Kaur K. Leap forward in wet lab surgical training and simulation using goat's eyeball during COVID-19 pandemic. Indian journal of ophthalmology. 2022 Mar:70(3):1059-1060. doi: 10.4103/ijo.IJO_41_22. Epub [PubMed PMID: 35225576]
Kaur K, Gurnani B, Venkatesh D, Sinha A, Tejaswini A, Niharika J, Morya AK. Commentary: Changing era of modern cataract surgery - The role of virtual reality-based simulators in manual small-incision cataract surgery training modules. Indian journal of ophthalmology. 2022 Nov:70(11):4016-4017. doi: 10.4103/ijo.IJO_2233_22. Epub [PubMed PMID: 36308147]
Level 3 (low-level) evidenceGurnani B, Tripathy K. Minimally Invasive Glaucoma Surgery. StatPearls. 2025 Jan:(): [PubMed PMID: 35881761]