Introduction
Pityriasis rotunda is an uncommon acquired dermatosis related to a keratinization disorder described first in 1906 by Jean Baptiste Emil Vidal. This dermatosis was called pityriasis circinata and pseudo-ichtyose acquise en taches circulaires. Pityriasis rotunda, uncommonly encountered in some regions of the world, is not always easy to diagnose.[1][2][3] Two variants of the condition have been identified. Type I is observed in adult Asian or Black individuals and is frequently linked to an underlying malignancy, typically stomach or hepatocellular carcinoma. Type II pityriasis rotunda manifests before age 40 and frequently has a familial pattern. This condition as not been linked to internal diseases or malignant tumors. The clinical pictures of type I and type II lesions are indistinguishable. The lesions appear as pink or light brown circular areas, finely delineated with dry scaling, and measure between 0.5 and 20 cm at their largest dimension; they typically develop on the trunk and proximal parts of the limbs. Individuals with dark skin typically exhibit hyperpigmented lesions, while those with a light complexion typically exhibit hypopigmented lesions.[4]
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
No established etiology for pityriasis rotunda exists. Authors have attached it to various disorders of keratinization, such as ichthyosis vulgaris, late-onset congenital ichthyosis, and acquired ichthyosis. Type I pityriasis rotunda, also known as paraneoplastic pityriasis rotunda, primarily appears in conjunction with hepatic and gastric malignancies, but it can also manifest in the following malignancies:
- Immunoglobulin G myeloma, multiple myeloma
- Acute myeloid leukemia
- Chronic myeloid leukemia
- Adenocarcinoma of the prostate
- Esophagus carcinoma
- Gastric carcinoma
- Squamous cell carcinoma of the palate
Pityriasis rotunda is also linked to several systemic diseases, including:
- Malnutrition
- Diabetes mellitus
- Infections (tuberculosis, leprosy, and osteitis)
- Hepatic diseases such as cirrhosis and hepatitis C virus [5]
- Chronic obstructive pulmonary disease and recurrent pulmonary infections
- Heart disease
- Female genital tract anomalies (ovarian cysts and fibroids)
- Glucose-6-phosphate dehydrogenase deficiency (favism)
- Chronic diarrhea
- Scleroderma
- Sarcoidosis
- Hormonal disorders such as hyperprolactinemia
- Acquired immune deficiency syndrome [6]
However, researchers have not eliminated the idea that the association of some of these conditions associated with pityriasis rotunda, especially favism, could be due to chance.[7][8][9][10]
Epidemiology
Pityriasis rotunda affects all sexes equally, although certain authors noticed a slight predominance in women. The age for the diagnosis ranges from 20 to 45 years, with extremes of 2 and 87 years. There is a particular racial and geographic distribution for pityriasis rotunda. Most cases are reported in Japan and among West Indian and Black individuals in South Africa. Case reports of pityriasis rotunda in Black Americans are less common, and the frequency of the disease on the North American continent is not known. Cases of pityriasis rotunda among White individuals are also rare. Familial cases may also be observed and likely have a hereditary determinism, which may be an autosomal dominant transmission. Researchers have documented a familial incidence among 42 individuals in Sardinia, thereby corroborating the theory of genetic influences.[7] A small number of cases involving children have also been documented.[1][11]
Researchers have developed a categorization encompassing all known pityriasis rotunda cases, proposing 2 subcategories. The first group consists of Asian or dark-complected patients with less than 30 hyperpigmented lesions. This condition does not run in families and is linked to 30% of cases of cancer or systemic diseases. White patients with a familial history manifest the second kind; additionally, the lesions are typically hypopigmented and numerous (>30), and this subtype is not associated with a chronic condition. This subtype emerges between ages 25 and 45 and remains constant throughout life.[12]
Pathophysiology
Lesions of pityriasis rotunda occasionally manifest disturbance of the epidermal filaggrin.[9] Hashimoto et al described a diminished expression of loricrin and filaggrin in pityriasis rotunda lesions.[13] Yoneda et al reported a complete absence of the profilaggrin-N terminus domain in the lesional skin.[14] Further, Makino et al reported a decrease in the expression of epidermal filaggrin 2, as seen in ichthyosis vulgaris, atopic dermatitis, and psoriasis vulgaris.[9]
Histopathology
The histopathology of pityriasis rotunda lesions can reveal various findings, such as hyperkeratosis, which is primarily orthokeratotic but also parakeratotic. Plugs are observed within hair follicles, elongated rete ridges, a diminished or absent stratum granulosum, slight spongiosis, enhanced pigmentation of the basal layer, pigmentary incontinence, and perivascular infiltrate of small lymphocytes. Periodic acid-Schiff stain for fungus is negative. These changes are similar to those observed in ichthyosis vulgaris. Type I pityriasis rotunda may be a biological variation of paraneoplastic ichthyosis vulgaris.[4]
Grimalt et al performed an ultrastructural study on pityriasis rotunda lesional skin biopsies. They noted the existence within the epidermis of vacuoles containing lipids and crystal-like inclusions. The granules of keratohyalin remained normal, while the number of keratinosomes increased. The stratum granulosum was reduced to a single layer. The cementing substance connecting corneocytes was abundant. These findings indicate a stronger relationship between pityriasis rotunda and congenital ichthyosis than with ichthyosis vulgaris.[15]
History and Physical
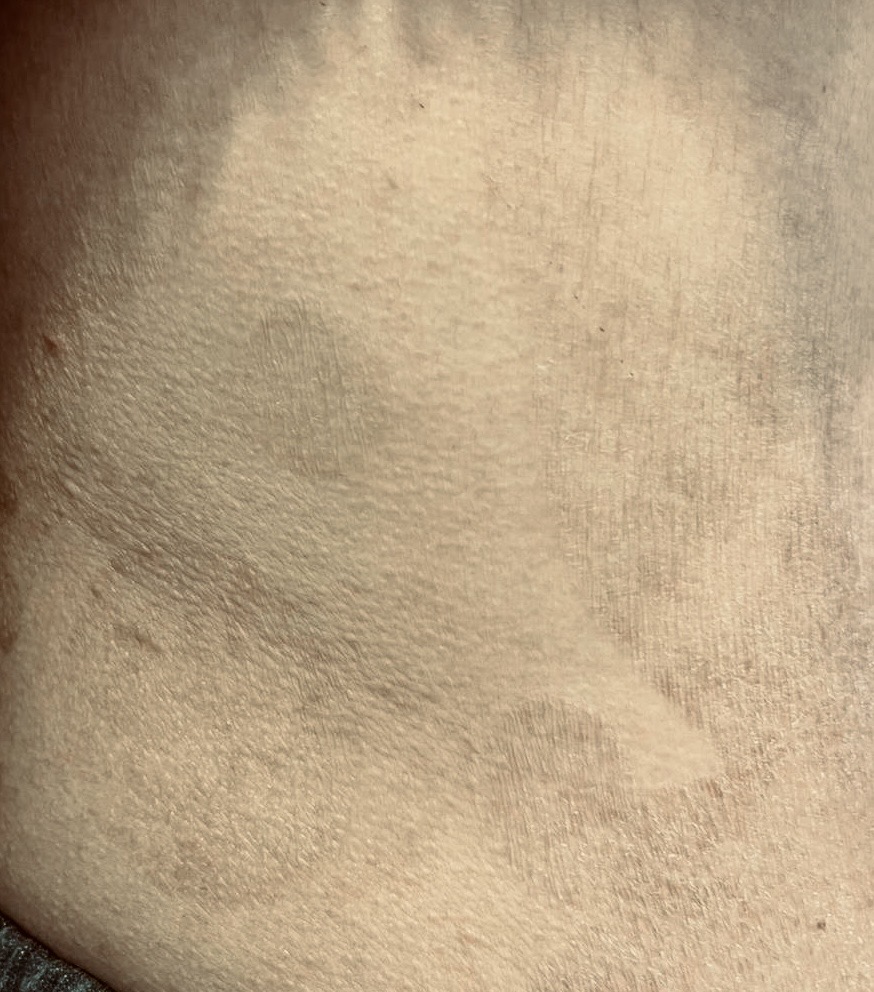
The diagnosis of pityriasis rotunda is usually based on clinical examination. Skin lesions are generally asymptomatic or mildly symptomatic. Pityriasis rotunda plaques are scaly, dry, sharply defined, and round, with prominent hair follicles but no inflammatory erythema. Scales may be highlighted by scratching the plaques. The lesions appear hypopigmented in fair-skinned patients and hyperpigmented in dark-skinned patients. The plaques of pityriasis rotunda may be confluent; coalescence of lesions may result in a polycyclic formation. Their size is variable (1 cm to more than 20 cm), and their number ranges from 1 to dozens but rarely exceeds 100. Pityriasis rotunda affects mainly the trunk (back and abdomen), buttocks, and proximal parts of the limbs (thighs and arms)(see Image. Pityriasis Rotunda). The face, hands, and feet are usually not involved. Some patients may exhibit associated skin lesions of ichthyosis vulgaris. A familial history of ichthyosis vulgaris has been reported in certain patients with pityriasis rotunda.[16][17]
Grimalt et al proposed a classification of pityriasis rotunda. The first subtype comprises less than 30 hyperpigmented patches, mainly in Black and Asian persons. No similar familial cases are seen in this subtype, but associated diseases are frequently described, such as malignancies (solid tumors and hemopathies), systemic diseases, and infections. A second subtype is characterized by more than 30 hypopigmented patches, occurring mainly in white patients with a familial history of similar skin lesions. There usually is no underlying disease. This classification is schematic since overlap cases have been reported.[15]
Evaluation
The diagnosis relies on clinical suspicion.[18] Mycological examination of scaly material removed from lesional skin (direct microscopy using potassium hydroxide and fungal culture) is useful in ruling out dermatomycosis. Wood light examination is used if erythrasma is suspected. On dermoscopy, white polygonal streaks with a homogeneous brown background were observed. The polygonal scales over clearly defined pigmented brown patches represent hyperkeratosis and a pigmented basal layer in histological analysis, respectively.[1][19]
Treatment / Management
Management of pityriasis rotunda is often challenging. Topical corticosteroids, antifungal agents, emollients, tars, and keratolytic topicals (salicylic acid and lactic acid) have proven ineffective; retinoids, topical (retinoic acid and tazarotene) or oral (etretinate), have been used with little or no success in pityriasis rotunda. Treatment with vitamin D3 analogs such as calcipotriol leads to a progressive improvement in the lesions in a few patients. However, for patients with comorbidities, successful treatment of the underlying disease often leads to remission of pityriasis rotunda lesions.[18]
Differential Diagnosis
Annular lesions are ring-shaped formations that arise from macules, papules, or plaques. While the annular pattern typically indicates lesion enlargement, annular lesions may not continue to expand. Various subtypes of annular lesions can be identified. These subtypes encompass a diverse array of inflammatory and infectious dermatoses, including:
- Erythrasma
- Leprosy
- Parapsoriasis
- Pityriasis alba
- Pityriasis rosea
- Plaque psoriasis
- Sarcoidosis
- Tinea corporis
- Tinea cruris
- Tinea versicolor
- Elastosis perforans serpiginosa
- Nummular eczema
- Early-stage mycosis fungoides
- Fixed drug reaction
- Figurate erythema
Prognosis
Once lesions manifest, they typically remain static throughout life unless the underlying internal disease or malignancy is managed, resulting in resolution or improvement. Moreover, childhood-onset has been associated with a more favorable prognosis, with remission occurring in nearly 50% of cases throughout later childhood.[11] Pityriasis rotunda was associated with a quantitatively and statistically significant reduction in mortality risk among all documented skin abnormalities of hepatocellular carcinoma.[20]
Complications
The manifestation of pityriasis rotunda in an older adult, without a familial history of the disorder, is significantly correlated with an underlying malignancy.[4]
Deterrence and Patient Education
Patients with pityriasis rotunda must undergo medical and physical examinations, as well as appropriate laboratory and radiographic tests, to detect internal diseases or malignancies.
Pearls and Other Issues
Skin lesions of pityriasis rotunda have a chronic course and can persist for months to years, with alternating periods of exacerbation (especially during winter or pregnancy) and remission. Since underlying diseases, mainly malignancies, have been associated with pityriasis rotunda, clinicians should know the importance of ruling out such diseases based on anamnesis, physical examination, and appropriate investigations. There is no definitive treatment for pityriasis rotunda. Lesions typically improve or resolve with the therapy of the underlying internal disease or cancer. Topical retinoids, salicylic acid ointment, and lactic acid lotion have been employed for the symptomatic treatment of lesions. Follow-up of pityriasis rotunda skin lesions without treatment is the other option because of possible spontaneous resolution.
Enhancing Healthcare Team Outcomes
Pityriasis rotunda is a rare skin disorder that is not simple to diagnose. The primary care clinician may see the condition, but because of the complexity of the diagnosis, it is best managed by a dermatologist. Once diagnosed, the management of pityriasis rotunda is often challenging. Topical corticosteroids, antifungal agents, emollients, tars, and keratolytic topicals (salicylic acid and lactic acid) have proven ineffective.
Retinoids, topical (retinoic acid and tazarotene) or oral (etretinate), have been used with little or no success in pityriasis rotunda. Treatment with vitamin D3 analogs such as calcipotriol led to a progressive improvement of the lesions in a few patients. The skin condition can take months or even years to disappear. However, for patients with comorbidities, successful treatment of the underlying disease often leads to remission of pityriasis rotunda lesions. Because of the few cases, no evidence-based guidelines on managing this disorder are available.
Media
(Click Image to Enlarge)
References
Al-Refu K, Al-Tarawneh A, Odeibat H. Pityriasis rotunda. A clinical study in Jordan: experience of 10 years. International journal of dermatology. 2018 Jul:57(7):759-762. doi: 10.1111/ijd.13994. Epub 2018 May 17 [PubMed PMID: 29774541]
Suzuki Y, Aoshima M, Fujiyama T, Ito T, Tokura Y. Pityriasis rotunda associated with acute myeloid leukemia. The Journal of dermatology. 2018 Jan:45(1):105-106. doi: 10.1111/1346-8138.13802. Epub 2017 Mar 29 [PubMed PMID: 28370261]
Vaccaro M, Salpietro C, Foti A, Borgia F, Lentini M, Cannavò SP. Pityriasis rotunda with recurrent respiratory infections. Giornale italiano di dermatologia e venereologia : organo ufficiale, Societa italiana di dermatologia e sifilografia. 2017 Apr:152(2):190-191. doi: 10.23736/S0392-0488.16.05327-X. Epub [PubMed PMID: 28166620]
Wick MR, Patterson JW. Cutaneous paraneoplastic syndromes. Seminars in diagnostic pathology. 2019 Jul:36(4):211-228. doi: 10.1053/j.semdp.2019.01.001. Epub 2019 Jan 31 [PubMed PMID: 30736994]
Bonito F, Costin A, Cunha H, Bártolo E. Pityriasis rotunda. The Australasian journal of dermatology. 2020 Aug:61(3):275-276. doi: 10.1111/ajd.13261. Epub 2020 Mar 3 [PubMed PMID: 32126584]
Mbamulu V, Otokpa GA, Ukonu BA, Ibekwe PU. Disseminated AIDS-associated Kaposi sarcoma without cutaneous involvement: a case report and review of literature. International cancer conference journal. 2024 Jul:13(3):245-249. doi: 10.1007/s13691-024-00671-9. Epub 2024 Apr 2 [PubMed PMID: 38962034]
Level 3 (low-level) evidenceLefkowitz EG, Natow AJ. Pityriasis Rotunda: A Case Report of Familial Disease in an American-Born Black Patient. Case reports in dermatology. 2016 Jan-Apr:8(1):71-4. doi: 10.1159/000445043. Epub 2016 Mar 18 [PubMed PMID: 27065844]
Level 3 (low-level) evidencePinos-León V, Núñez M, Salazar M, Solís-Bowen V. Pityriasis Rotunda and Hyperprolactinemia. Actas dermo-sifiliograficas. 2016 Jul-Aug:107(6):535-7. doi: 10.1016/j.ad.2015.10.016. Epub 2016 Mar 19 [PubMed PMID: 27006018]
Makino T, Mizawa M, Seki Y, Hayashi M, Shimizu T. Decreased filaggrin-2 expression in the epidermis in a case of pityriasis rotunda. Clinical and experimental dermatology. 2016 Mar:41(2):215-7. doi: 10.1111/ced.12716. Epub 2015 Jul 20 [PubMed PMID: 26189536]
Level 3 (low-level) evidencevan Heerden T, Webb MJ, Barrett CL. Pityriasis rotunda as an incidental paraneoplastic finding in two patients with multiple myeloma. Clinical and experimental dermatology. 2014 Aug:39(6):734-7. doi: 10.1111/ced.12396. Epub 2014 Jul 1 [PubMed PMID: 24985090]
Level 3 (low-level) evidenceEna P, Cerimele D. Pityriasis rotunda in childhood. Pediatric dermatology. 2002 May-Jun:19(3):200-3 [PubMed PMID: 12047637]
Li G, Xu Z, Huang L, Xue R. Pityriasis rotunda. International journal of dermatology. 2023 Aug:62(8):1098-1100. doi: 10.1111/ijd.16369. Epub 2022 Aug 7 [PubMed PMID: 35933687]
Hashimoto Y, Suga Y, Chikenji T, Matsuba S, Mizoguchi M, Kakuta M, Ogawa H. Immunohistological characterization of a Japanese case of pityriasis rotunda. The British journal of dermatology. 2003 Jul:149(1):196-8 [PubMed PMID: 12890220]
Level 3 (low-level) evidenceYoneda K, Presland RB, Demitsu T, Shimizu T, Kubota Y. The profilaggrin N-terminal domain is absent in pityriasis rotunda. The British journal of dermatology. 2012 Jan:166(1):227-9. doi: 10.1111/j.1365-2133.2011.10528.x. Epub [PubMed PMID: 21777219]
Grimalt R, Gelmetti C, Brusasco A, Tadini G, Caputo R. Pityriasis rotunda: report of a familial occurrence and review of the literature. Journal of the American Academy of Dermatology. 1994 Nov:31(5 Pt 2):866-71 [PubMed PMID: 7962738]
Zur RL, Shapero J, Shapero H. Pityriasis rotunda diagnosed in Canada: case presentation and review of the literature. Journal of cutaneous medicine and surgery. 2013 Nov-Dec:17(6):426-8 [PubMed PMID: 24138982]
Level 3 (low-level) evidenceMirsky L, Watters K, Jafarian F. Chronic hyperpigmented scaly plaques. Pityriasis rotunda (PR). Archives of dermatology. 2012 Sep:148(9):1073-8. doi: 10.1001/archdermatol.2012.1494. Epub [PubMed PMID: 22986863]
Level 3 (low-level) evidenceWollina U, Fölster-Holst R, Karadağ AS. Miscellaneous annular diseases. Clinics in dermatology. 2023 May-Jun:41(3):413-421. doi: 10.1016/j.clindermatol.2023.07.010. Epub 2023 Jul 13 [PubMed PMID: 37453713]
Chamli A, Kouki C, Belhadj O, Helal I, Hammami H, Fenniche S, Zaouak A. Pityriasis rotunda associated with hepatocellular carcinoma. Journal of the European Academy of Dermatology and Venereology : JEADV. 2023 Dec:37(12):e1438-e1440. doi: 10.1111/jdv.19353. Epub 2023 Jul 24 [PubMed PMID: 37458682]
Al-Showbaki L, Toubasi AA, Jaber DZ, Shdifat MA, Al-Maani N, Qudah O, Fararjeh F, Amir E. Paraneoplastic Cutaneous Manifestations of Hepatocellular Carcinoma. A Systematic Review and Meta-analysis. Journal of Cancer. 2024:15(4):1021-1029. doi: 10.7150/jca.88931. Epub 2024 Jan 1 [PubMed PMID: 38230223]
Level 1 (high-level) evidence