Introduction
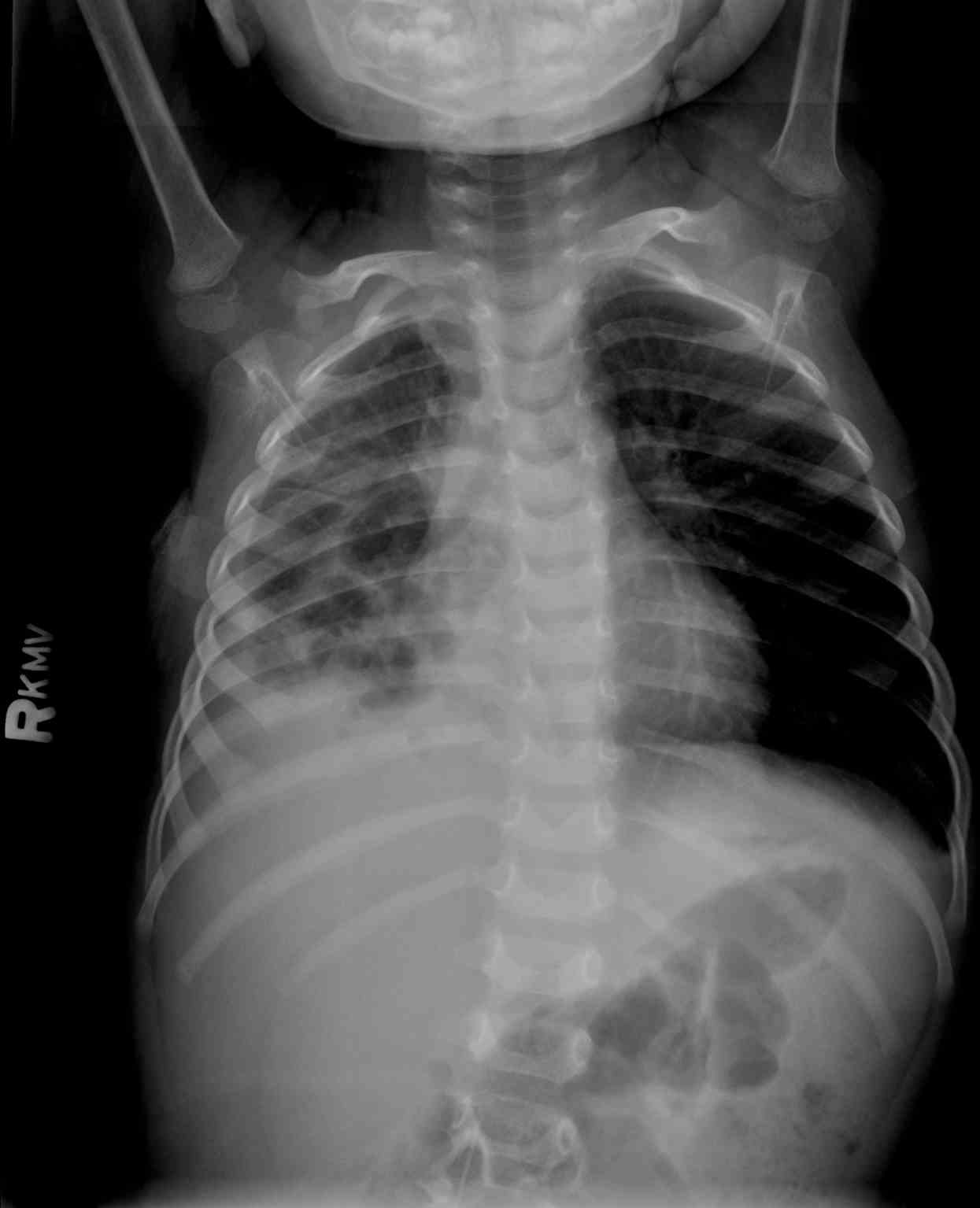
Pneumatoceles, also called pulmonary pseudocysts, are thin-walled, air-filled cystic lesions that form within the lung interstitium, often as a complication of infections, trauma, or mechanical ventilation, particularly in young and immunocompromised individuals (see Image. Pneumatoceles on Chest Radiograph). These lesions are typically air-filled but may also contain air-fluid levels, with sizes ranging from small cysts to those occupying a significant portion of the thoracic cavity. Although they lack an epithelial lining, which distinguishes them as pseudocysts, pneumatoceles can be single or, more commonly, multiple, sparing the lung apices.
Fraser and Pare first clearly defined the term pneumatocele in 1977, describing cavities occupying up to one-third of lung volume. Pneumatoceles can be associated with conditions such as traumatic pulmonary contusions, pneumothoraces, and pneumomediastinum. Rare cases of cervical pneumatoceles in infants may mimic laryngoceles, presenting diagnostic challenges.[1] Though often resolving spontaneously, particularly when caused by infections or trauma, effective pneumatocele management may require intervention based on severity, complications, and underlying pathology, such as in cases of desquamative interstitial pneumonia.
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
The primary mechanism of pneumatocele formation involves interstitial or alveolar air trapping, forming thin-walled, air-filled cysts. Pneumatoceles arise due to various underlying etiologies, most commonly due to lung injury, infection, or mechanical forces. These include:
- Infections
- Pneumatoceles frequently occur following bacterial infections, with Staphylococcus aureus being a leading pathogen, especially in children.
- Other infectious agents include tuberculosis, Streptococcus pneumoniae, Proteus mirabilis, Escherichia coli, Acinetobacter calcoaceticus, Klebsiella pneumoniae, and Pneumocystis jirovecii.[2]
- Notably, intravenous (heroin users are also at risk of developing infection-related pneumatoceles due to aspiration and associated lung damage.
- Infections induce necrosis and airway damage, promoting air trapping.
- Trauma
- Pneumatoceles can develop following blunt or penetrating chest trauma. In traumatic cases, they often result from pulmonary contusions, where the force of impact causes alveolar rupture, trapping air in the interstitial space. This may be seen with pneumothorax, pneumomediastinum, or pulmonary lacerations.
- Mechanical ventilation
- Other causes
- Underlying lung diseases
- Desquamative interstitial pneumonia can lead to the formation of multiple cysts or pneumatoceles.
- Persistent or diffuse interstitial pulmonary emphysema is another cause.
- Congenital disorders
- Cystic adenomatoid malformations may also be linked to the development of similar air-filled cysts.
- Hydrocarbon inhalation [2][5]
- Kerosene ingestion [6][7]
- Burns [8][6][9]
- Endobronchial valve-related injuries
- These also lead to pneumatocele formation.[9]
- Underlying lung diseases
The etiology of pneumatoceles reflects underlying alveolar or interstitial damage, with varying severity and clinical implications based on the root cause.
Epidemiology
Pneumatoceles are air-filled cystic lesions in the lungs due to trauma, pneumonia, or certain infections. They are particularly common among infants, children, adolescents, and individuals with weakened immune systems, such as those with acquired immunodeficiency syndrome. Traumatic pneumatoceles, in particular, are most often seen in children and young adults and are generally the result of nonpenetrating chest trauma, such as motor vehicle accidents or falls. These rare lesions occur in less than 3% of patients with pulmonary parenchymal injuries. Results from a study in Milan, Italy, reported 10 cases over 9 years, and other reports indicate that about 85% of traumatic pneumatoceles occur in individuals aged younger than 30. Male patients are more commonly affected due to the higher rates of trauma in men.[10][11][12][13] Pneumonia complicated by pneumatoceles is seen in 2.4% to 8.3% of hospitalized children with pneumonia.[14]
The incidence of pneumatoceles is notably higher in certain regions. For instance, results from a study in Brazil found that 9.5% of hospitalized children with pneumonia developed pneumatoceles. This high incidence has been linked to malnutrition, which can delay the development of Kohn pores in the lungs, leading to the formation of pneumatoceles when there is valvular obstruction.[14] Traumatic pneumatoceles in children and adolescents occur due to the greater compressibility of their thoracic skeleton and the elastic recoil of their lungs, which increases the negative intrathoracic pressure and leads to laceration of the lung parenchyma. Only about 20% of these patients have associated rib fractures. The formed cavities filled with fluid, blood, or air continue to increase in size until lung pressures are balanced between the cavities and the surrounding tissue.[4][15][16] A survey of 10,229 trauma admissions found that 12.3% of children with pulmonary contusions developed a pneumatocele. The largest pneumatocele reported in this study was 3.7 cm in diameter, and most cases resolved spontaneously without intervention.[12]
Pneumatoceles are also reported in patients recovering from viral infections. For example, results from a study of H7N9 influenza survivors showed that 9.8% developed pneumatoceles; a similar occurrence was noted in 10% of those after COVID-19 infection.[17][18] Additionally, pneumatoceles have been observed in patients with pulmonary metastases treated with chemotherapy, such as paclitaxel, where pulmonary nodules regressed into multiple pneumatoceles.[19] These various causes highlight the diverse etiologies of pneumatoceles and their clinical significance in both infectious and traumatic settings.
Pathophysiology
Pneumatoceles are air-filled cavities within the lung parenchyma that can arise from various causes, including trauma, infection, and inflammatory processes. The pathogenesis of pneumatocele formation is complex and involves mechanical and inflammatory mechanisms. The formation of a pneumatocele is commonly attributed to an endobronchial check-valve mechanism. This process leads to air trapping and the subsequent development of cystic air space in the distal regions of the lung. Another theory involves inflammation and necrosis of an airway segment, resulting in direct communication between the airway and the bronchovascular interstitium. This communication allows air to travel through the bronchovascular connective tissue, accumulating beneath the pleura and forming a recognizable pneumatocele.[20]
Pneumatoceles are also associated with various infectious agents, including:
- Streptococcus pneumonia [21]
- Staphylococcus aureus
- In cases involving this pathogen, 60% result in pneumatocele.
- This bacteria is more commonly found in children.
- In an analysis of burns patients, those who develop staphylococcal pneumonia also develop pneumatocele.[22][23]
- A case report of S aureus and candida colonization leading to multiple pneumatoceles is also reported.
- Pneumocystis carinii in human immunodeficiency virus [23][24][25][26]
- Pseudomonas aeruginosa
- Group A streptococci
- Klebsiella pneumoniae [27]
- Adenovirus
- Mycobacterium tuberculosis
- Pneumatoceles can develop during or after treatment of tuberculosis.[28]
- Proteus mirabilis and extended-spectrum beta-lactamase Proteus mirabilis [29][30]
- Escherichia coli
- Cavitary pneumonia from this pathogen has been associated with the formation of pneumatocele.[31]
- Acinetobacter calcoaceticus [32]
- Hemophilus influenzae B [33]
- Bacteroides species, Peptostreptococcus asaccharolyticus, and Fusobacterium species [34]
These pathogens can trigger airway inflammation and obstruction. The resulting inflammatory exudates can create the ball-valve effect, contributing to pneumatocele formation. In postpneumonia cases, inflammation and narrowing of the bronchus often lead to an endobronchial ball-valve mechanism, causing distal dilation of the bronchi and alveoli. Air enters the cystic space but cannot escape due to inflammatory exudates within the airway lumen or wall. This mechanism results in pneumatocele enlargement, combined with pressure exerted by adjacent pneumatoceles or accumulating exudates.
After trauma, pseudocysts form due to shearing forces that cause pulmonary lacerations, leading to the escape of air or fluid into the surrounding tissue. The exact mechanisms underlying traumatic pneumatocele formation remain uncertain, but several theories have been proposed:
- Concussive waves and shearing forces
- Blunt trauma generates concussive waves, which create shearing forces capable of tearing the lung parenchyma. The location and type of injury depend on the impact characteristics:
- High-velocity impacts with minimal chest wall displacement produce peripheral pseudocysts.
- Low-velocity impacts with significant chest wall displacement result in central pseudocysts.
- Both scenarios involve intraparenchymal pulmonary lacerations, with airway disruptions allowing air to leak into the pulmonary parenchyma.[35]
- Blunt trauma generates concussive waves, which create shearing forces capable of tearing the lung parenchyma. The location and type of injury depend on the impact characteristics:
- Explosive pressure and "bursting lesions"
- Sudden, severe compression of a segment of the peripheral bronchial tree can obstruct the bronchus and transmit explosive pressure distally, creating "bursting lesions." This process occurs in 2 steps:
- Step 1
- Compressive forces increase intrapulmonary pressure, causing the lung parenchyma to rupture and form lacerations.
- Step 2
- Subsequent chest decompression and increased negative intrathoracic pressure allow the lung's elastic tissue to recoil, forming small cavities filled with air or fluid. These cavities expand until the internal pressure equilibrates with the surrounding parenchyma.
- Step 1
- Sudden, severe compression of a segment of the peripheral bronchial tree can obstruct the bronchus and transmit explosive pressure distally, creating "bursting lesions." This process occurs in 2 steps:
- Check-valve mechanism
- In cases of penetrating trauma or airway injury, a one-way or check-valve effect may occur. This allows air to enter a laceration in the pulmonary parenchyma while the defect seals quickly, trapping the air and preventing its escape. Similar mechanisms have been observed in patients mechanically ventilated receiving continuous positive airway pressure.[4]
Traumatic pneumatoceles typically result from significant chest trauma involving a pliable chest wall, where kinetic energy is efficiently transmitted to the underlying pulmonary parenchyma. The condition is more common in young people and children due to the increased elasticity of their chest walls. Radiologically, traumatic pneumatoceles appear as pulmonary cavities devoid of epithelial lining and filled with air, fluid, or blood. They often coexist with other injuries, such as pneumothorax or pneumomediastinum. A pneumatocele indicates extensive tissue disruption, signifying a more severe injury than a simple pulmonary contusion.[10][36][37]
The location of the pneumatocele may not correspond to the site of the chest wall injury. In some cases, traumatic forces are transmitted in a contrecoup manner. The velocity and nature of the impact influence the lesion's location. High-velocity impacts tend to cause peripheral alveolar injuries. In contrast, low-velocity impacts with substantial chest wall displacement lead to central parenchymal injuries. In studies of high-speed accidents, results showed all traumatic pneumatoceles occurred peripherally.
Environmental exposures, such as hydrocarbons, can induce lung injuries characterized by rapid progression of respiratory difficulty without causative factors and the presence of ground-glass opacities, spontaneous air leak syndrome resulting in pneumatoceles, and consolidation with later diffuse centrilobular nodular opacity on chest computed tomography.[5] Hydrocarbons contain polyhexamethylene guanidine (PHMG), oligo (2-[2-ethoxy]ethoxyethyl) guanidinium chloride (PGH), 5-chloro-2-methyl-4-isothiazolin-3-one (CMIT), or 2-methyl-4-isothiazolin-3-one (MIT). PHMG and PGH are commonly thought to cause hydrocarbon-associated lung injuries. Animal study results have not established definitive links between exposure to hydrocarbons containing CMIT and/or MIT, even though patients exposed to hydrocarbons containing only CMIT and/or MIT showed clinically similar lung injury to that of hydrocarbons containing PHMG and/or PGH.[38][39]
Persistent interstitial pulmonary emphysema (PIPE), when diffuse, can be associated with pneumatocele formation. PIPE is a rare entity related to mechanical ventilation-induced lung damage in premature infants with respiratory distress syndrome. Although PIPE localized in 1 lobe has the potential for curative resection, diffuse PIPE affecting both lungs carries a poor prognosis. Rarely, extrathoracic pneumatocele formation has been documented, such as in untreated tuberculosis patients where old scars provide a pathway for air accumulation.[28][40][41]
Results from a Brazilian study proposed that the Kohn pores theory contributes to pneumatocele formation. Kohn pores, small interalveolar connections, play a critical role in equalizing pressure within the lung. According to this theory, delayed development of Kohn pores, often due to undernutrition, leaves the lungs of malnourished children functionally similar to those of newborns. In this scenario, when a valvular obstruction occurs, air enters a lung segment but cannot escape, forming a pneumatocele. The absence of well-developed Kohn pores in these patients allows air trapping, as there are no alternative pathways for air redistribution within the alveolar network. This mechanism might explain the higher prevalence of pneumatoceles in children compared to adults, in whom fully developed Kohn pores facilitate better intrapulmonary pressure equilibration.[35]
Histopathology
Pneumatoceles are air-filled cystic lesions that arise within the lung parenchyma, and their histopathological features are characterized by the lack of epithelial lining and varying degrees of surrounding inflammatory and fibrotic changes. The specific histopathology of pneumatoceles depends on the underlying etiology (eg, trauma, infection, or inflammatory conditions) but generally includes the following features:
- Lack of epithelial lining
- The most distinguishing feature of pneumatoceles is that they are not lined by epithelium, distinguishing them from true cysts. The cavity is essentially a space within the lung parenchyma filled with air or fluid, but it lacks the typical epithelial lining seen in other types of cysts. This absence of epithelial lining is a key histological feature of pneumatoceles, especially when compared to congenital cysts or other cystic lesions that might have a well-developed epithelial or mesothelial lining.
- Air or fluid-filled cavities
- The cystic spaces within pneumatoceles contain air, fluid, or blood, depending on the underlying cause. These spaces are often filled with purulent exudate or inflammatory debris in infectious cases. The fluid component may appear as a proteinaceous exudate; in some cases, it may contain cellular debris, inflammatory cells (such as neutrophils), and fibrotic material in chronic cases.
- Surrounding inflammatory infiltrate
- In many cases, a marked inflammatory infiltrate will surround the pneumatocele, particularly those related to infection or inflammation (such as pneumonia). This infiltrate often includes neutrophils, lymphocytes, and macrophages, depending on the inflammation stage and the underlying condition's nature. In bacterial infections, the inflammatory response may be more acute, while in viral or chronic infections, the infiltrate may be more chronic and predominantly lymphocytic.
- Fibrotic changes and granulation tissue
- Chronic pneumatoceles, especially those associated with trauma or long-standing inflammation, may show varying degrees of fibrosis surrounding the cystic space. This fibrosis is typically a response to the injury or inflammation and may be seen as dense collagen deposition or fibrous tissue around the pneumatocele. In cases of traumatic pneumatoceles, the fibrotic changes may be a result of the healing response to pulmonary laceration and tissue disruption. Granulation tissue may also be observed in the surrounding lung parenchyma, especially in the early stages after trauma or infection.
- Necrosis and hemorrhage
- In some cases, particularly in traumatic pneumatoceles, hemorrhage may occur within the cyst or surrounding tissue. The cyst may contain blood or hemosiderin-laden macrophages, indicating previous bleeding. Necrotic tissue may also be present, particularly in severe trauma or infectious processes, where the injury or infection has resulted in tissue death.
- Absence of a bronchial lining
- Pneumatoceles, particularly those caused by trauma, often lack the bronchial or airway structure that would be expected in other types of pulmonary cysts. The lesion typically represents an area of pulmonary parenchyma dissected by air, blood, or fluid following mechanical trauma, infection, or other pathologic processes without forming a discrete bronchial or ductal system inside the cyst.
- Presence of septations (in some cases)
- Some pneumatoceles, particularly those of infectious or inflammatory origin, may develop internal septations. These septations are thin, fibrous partitions that can arise from the inflammatory or reparative processes occurring within the cyst. These partitions may be more prominent in cases of chronic or long-standing pneumatoceles.
A documented case of pneumonectomy performed for multiple pneumatoceles revealed findings consistent with cystic adenomatoid malformation in a patient with hyper-immunoglobulin (Ig)E syndrome. This malformation was characterized by multiple cysts of varying sizes, all lined by epithelium, within the affected lung tissue.[42] Hyper-IgE syndrome, a rare immunodeficiency disorder, is often associated with recurrent infections, chronic inflammation, and structural lung abnormalities, including pneumatocele formation. In this case, the coexistence of multiple pneumatoceles and cystic adenomatoid malformation highlights a potential link between the chronic inflammatory state, immune dysfunction, and the development of complex pulmonary abnormalities in such patients.
History and Physical
The history and physical examination findings of a patient with a pneumatocele vary based on the size, location, underlying etiology, and associated complications. Pneumatoceles are typically asymptomatic but may cause symptoms in cases of significant size or secondary complications such as infection, rupture, or compression of adjacent structures.
History
Pneumatoceles are thin-walled, air-filled cystic lesions within the lung parenchyma that arise from various causes, including bacterial pneumonia (notably Staphylococcus aureus and Streptococcus species), blunt chest trauma, chemical toxin exposure (eg, hydrocarbons), or positive-pressure ventilation. Although more commonly seen in infants, children, and adolescents, pneumatoceles are relatively uncommon in adults, with limited cases documented in this population.[43] Risk factors for their formation include young age, postinfectious or posttraumatic conditions, human immunodeficiency virus, intravenous drug use, and immune dysfunctions such as hyper-IgE syndrome.[14][44][45] The clinical presentation varies based on the size of the lesion, the underlying cause, and associated complications, ranging from asymptomatic findings to significant respiratory symptoms.
A detailed history often reveals preceding acute illnesses or events contributing to pneumatocele development. Many cases follow prior infections, particularly Staphylococcus aureus pneumonia in children or necrotizing pneumonia caused by organisms such as Klebsiella pneumoniae, Streptococcus pneumoniae, or Pseudomonas aeruginosa.[14][46] Patients typically report a history of fever, cough, dyspnea, chest pain, or sputum production linked to the initial infectious episode. Viral pneumonia, including those caused by influenza or respiratory syncytial virus, may also precede pneumatocele formation.
In some cases, trauma or iatrogenic factors play a significant role. Blunt or penetrating chest trauma, barotrauma from mechanical ventilation, or postoperative thoracic procedures such as thoracentesis or biopsy may predispose patients to pneumatocele development. Toxic shock syndrome associated with Staphylococcus aureus can also lead to pneumatocele formation, presenting with rash, fever, chills, and mucositis. Traumatic pneumatoceles often appear within 24 to 48 hours after injury, with hemoptysis occurring in 56% of cases, alongside cough and chest pain due to pulmonary parenchymal injury.[47] However, acute respiratory failure in these scenarios typically stems from associated pulmonary contusion rather than the pneumatocele itself.[48]
Complications of pneumatoceles can lead to specific symptoms. Sudden-onset chest pain or acute respiratory distress may indicate rupture, resulting in pneumothorax or tension pneumothorax. Secondary infection of the pneumatocele may present with fever, worsening respiratory symptoms, or systemic signs like malaise or fatigue. Underlying medical conditions, including immunosuppressive states, cystic fibrosis, or chronic granulomatous disease, increase pneumatocele susceptibility. A history of recurrent infections or chronic respiratory symptoms may further suggest structural lung abnormalities predisposing to their formation.
Physical
On physical examination, findings in patients with pneumatoceles vary depending on complications or associated conditions. In uncomplicated cases, patients may appear well or show mild respiratory symptoms. At the same time, those with complications often present with respiratory distress characterized by tachypnea, hypoxia, or the use of accessory muscles. Vital signs provide essential diagnostic clues, with tachypnea, fever, and hypoxia pointing to infection, respiratory compromise, hypotension, and tachycardia suggesting tension pneumothorax or sepsis.
Inspection may reveal decreased chest wall movement on the affected side, especially in significant pneumatocele or pneumothorax cases, with cyanosis or labored breathing apparent in severe cases. Palpation findings include decreased tactile fremitus over large pneumatoceles or pneumothorax and subcutaneous emphysema in rare cases of rupture. Percussion may reveal hyperresonance over air-filled spaces, such as a pneumatocele or pneumothorax.
Auscultation findings include diminished or absent breath sounds over affected lung areas. Adventitious sounds such as crackles or rhonchi may occur when infection or inflammation surrounds the pneumatocele. In cases of rupture causing tension pneumothoraces, mediastinal shift, and muffled heart sounds may occur, indicating a critical need for immediate intervention.
Mild fever or leukocytosis appearing 12 to 36 hours after trauma often results from the absorption of damaged lung tissue or blood clots rather than infection and should not be misinterpreted.[10] Advanced cases or those with complications, such as secondary infection, may also present with systemic signs like fever, chills, and leukocytosis. Careful monitoring of pneumatoceles, whether caused by trauma or illness, is crucial to address potential complications, including rupture, secondary infection, or compression of adjacent structures.
Evaluation
Evaluating pneumatoceles involves integrating clinical history, physical examination, laboratory findings, and imaging studies to confirm the diagnosis, identify underlying causes, and assess complications. While typically nonspecific, laboratory investigations play a supportive role in evaluating pneumatoceles. Blood tests may reveal leukocytosis, abnormal liver function, anemia, and thrombocytopenia, indicating an underlying infection or systemic inflammatory response. Although often negative, blood cultures can be positive for anaerobic organisms such as Bacteroides fragilis when infection is present. Elevated markers like c-reactive protein and procalcitonin may further indicate the severity of inflammation or infection. In certain cases, specific serum markers like cancer antigen-125 can help predict disease severity, particularly in complicated infectious conditions.
Imaging studies are paramount in diagnosing pneumatoceles and assessing their characteristics. Chest radiographs are frequently the first imaging modality used, especially in children, with 90% of pneumatoceles visible on the first chest radiograph after pneumonia (see Image. Pneumatoceles on Chest Radiograph). Radiologic evidence typically appears on the fifth to sixth day of hospitalization. However, chest radiographs have a low sensitivity of around 24% for diagnosing traumatic pneumatoceles, especially when obtained in the supine position or when the lesion is smaller than 2 cm. Despite the yield of false-negative findings, they are still used as the first diagnostic modality in patients with thoracic trauma.[10]
The diagnostic accuracy of chest radiography ranges from 24% to 50%; however, serial chest radiographs taken over several days can enhance detection, particularly after the fifth post-trauma day when pulmonary contusions begin to resolve and pneumatoceles become more apparent. The typical radiographic presentation of a traumatic pneumatocele includes a round or oval shadow usually surrounded by pulmonary contusion, with air-fluid levels often present due to bleeding into the pneumatocele.[49][50] Most traumatic pneumatoceles are found in the lower lobes.[10]
Computed tomography (CT) is the gold standard for evaluating pneumatoceles, offering a much higher sensitivity of approximately 96% compared to radiography. CT scans provide detailed images that precisely define the pneumatocele's location, size, and extent and identify any associated complications, such as pulmonary contusions or abscess formation. The typical appearance of a traumatic pneumatocele on CT is a round or oval thin-walled cavitary lesion with air-fluid levels commonly found in the lower lobes. The main advantage
s of CT include earlier identification, visualization of small lesions, and the ability to detect pneumatoceles even in the presence of pulmonary contusion. Recent study results report an increased incidence of pneumatoceles due to the more widespread use of CT rather than an actual rise in occurrence.[51] Additionally, CT-guided aspiration can be performed to sample the abscess and place an indwelling drain, facilitating diagnosis and treatment.
In pediatric populations, lung ultrasound has emerged as a useful adjunct to chest radiographs, enhancing diagnostic accuracy without radiation exposure.[52] The size of traumatic pseudocysts reported in the literature ranges from 1 to 14 cm in diameter, with larger pseudocysts more frequently occurring in patients with multiple injuries, bilateral pulmonary contusions, or significant respiratory involvement. Serial imaging studies are often necessary to monitor changes in size, shape, and composition of pneumatoceles within days following trauma, helping to differentiate them from other lesions.[53]
Advanced diagnostic techniques, such as CT-guided needle aspiration and bronchoscopy, may be employed in cases where secondary infection or abscess formation is suspected. These procedures allow microbiological sampling and direct intervention, particularly in recurrent or antibiotic-resistant collections. Emerging technologies, like optical property measurements and methylene blue uptake for photodynamic therapy, are being explored to improve the management of refractory pneumatoceles.
Treatment / Management
Treating pneumatoceles involves various approaches tailored to their etiology, size, complications, and clinical presentation. Accurate diagnosis is crucial to avoid unnecessary interventions and potential complications. Misdiagnosing a pneumatocele as a different cavitary pulmonary lesion, such as a pulmonary abscess or pneumothorax, can lead to harmful procedures like thoracostomy tube placement or inappropriate drainage. Below is a comprehensive description of management strategies, incorporating national and international guidelines.
Conservative Management
Conservative management is the cornerstone of treatment for most cases of pneumatocele, emphasizing observation, appropriate antimicrobial therapy, and supportive care. Most pneumatoceles resolve spontaneously within weeks to 6 to 12 months, with larger lesions sometimes requiring up to 6 months or more for complete resolution. In children, spontaneous resolution occurs in up to 90% of cases without invasive intervention. Regular clinical evaluations and imaging are essential for monitoring the progression or resolution of these lesions, with the average radiological resolution time being approximately 3 months.
Ventilation strategies are vital in managing pneumatocele-associated respiratory compromise. High-frequency oscillatory ventilation and unilateral main bronchus ventilation are effective techniques for maintaining adequate respiratory function while minimizing the risk of pneumatocele enlargement.[3][54] Avoiding high-pressure ventilation is critical to preventing further expansion of the lesion. Additionally, decubitus positioning with the affected side down can alleviate symptoms and improve clinical outcomes in some cases.(B3)
Antimicrobial therapy plays a key role in managing infected pneumatoceles. Empiric antibiotic regimens are initiated for suspected infections and tailored based on culture results to address common pathogens such as Staphylococcus aureus, Streptococcus pneumoniae, and mixed anaerobic organisms. Routine prophylactic antibiotic use is discouraged unless indicated by concomitant injuries or surgical interventions, as it may promote resistant strains and unnecessary colonization.
While conservative management remains the first-line approach for uncomplicated cases, it is insufficient in patients with cardiorespiratory instability or extensive disease.[55][56][57] Early escalation to invasive or surgical intervention is crucial in such situations to stabilize the patient and effectively address underlying complications. Therefore, careful clinical judgment and ongoing monitoring are essential to achieving favorable outcomes.(B3)
Minimally Invasive Interventions
- Percutaneous drainage
- Percutaneous needle decompression and drainage is a therapeutic option for managing pneumatocele in select cases where complications arise, or conservative management proves insufficient. This technique is indicated under the following circumstances:
- When the pneumatocele occupies more than 50% of the hemithorax, leading to compression of adjacent structures.[58]
- In cases of significant atelectasis caused by the pneumatocele's mass effect.
- Development of bronchopleural fistulae, which can lead to persistent air leaks.
- Presence of a tension pneumatocele, where intrathoracic pressure compromises cardiorespiratory function.[59]
- Persistent signs of a chest infection or infection within the cyst that fails to respond to antibiotic therapy.
- Difficulty in ensuring adequate outpatient follow-up, raising concerns about monitoring progression or complications.
- Decompression by percutaneous pigtail catheter placement in children who develop pneumatoceles as a result of a complication of pulmonary interstitial emphysema with superimposing respiratory syncytial virus infection has been described in a preterm infant in a case report.[60][61][62][63]
- While effective, percutaneous needle decompression carries risks, including the potential development of bronchopleural fistulae, which may complicate the clinical course and necessitate further intervention.[64] Careful patient selection and meticulous procedural techniques are critical to minimizing complications and optimizing outcomes.
- In some cases, patients are not suitable for earlier intervention with percutaneous drainage of the pneumatoceles due to the extent of lung disease and the high risk of complications associated with full heparinization and extracorporeal membrane oxygenation support.
- Percutaneous needle decompression and drainage is a therapeutic option for managing pneumatocele in select cases where complications arise, or conservative management proves insufficient. This technique is indicated under the following circumstances:
- Injection therapy
- The management of pneumatocele can also involve minimally invasive interventions, such as injecting fibrin sealant via a pigtail catheter, particularly in cases with persistent air leaks.[65] Fibrin sealant, a biodegradable material, is favored for effectively sealing air leaks by promoting natural tissue adhesion. This surgical hemostatic and adhesive material has applications in cardiac, vascular, hepatic, and maxillofacial surgeries.
- Sclerosants used in pleurodesis, such as talc, antibiotics, antineoplastic drugs, and autologous blood, can also help manage air leaks. A study by Andres et al demonstrated the efficacy of autologous blood infusion, reporting 100% success in treating persistent air leaks within 24 hours.
- The use of fibrin sealant, guided by CT-fluoroscopy, allows for precise catheter manipulation and targeted closure of fistulas. This approach is both rapid and safe, reducing the duration of hospitalization and potentially avoiding more invasive surgical interventions. These techniques underscore the importance of tailored, patient-specific strategies in effectively managing complicated pneumatocele cases.
- Bronchoscopy
- Diagnostic and therapeutic bronchoscopy are crucial in managing traumatic pneumatoceles, particularly in cases with specific complications or diagnostic uncertainties. The indications for bronchoscopy include:
- Endobronchial bleeding
- Thick sputum
- Large air leak
- Mediastinal emphysema
- Lobar collapse
- Bronchoscopy in these scenarios serves diagnostic and therapeutic purposes, aiding in accurately identifying underlying issues while enabling immediate intervention to stabilize the patient's respiratory status.
(B2) - Diagnostic and therapeutic bronchoscopy are crucial in managing traumatic pneumatoceles, particularly in cases with specific complications or diagnostic uncertainties. The indications for bronchoscopy include:
Surgical Interventions
Surgical intervention is generally reserved for patients with pneumatocele who do not respond to conservative or less invasive treatments. Indications for surgery include persistent air leaks, progressive enlargement of the pneumatocele, significant compression of adjacent lung parenchyma, recurrent infections, or cardiorespiratory instability. Additionally, failure of catheter drainage, bronchoscopy, or other minimally invasive approaches may necessitate surgical management. The extent of the disease determines the choice of procedure, the patient’s clinical stability, and the potential to preserve functional lung tissue.
Pneumatoceles larger than 6 cm or those unresponsive to conservative treatment are considered strong candidates for surgical resection. Procedures such as video-assisted thoracoscopic surgery (VATS) lobectomy or traditional lobectomy are generally associated with improved outcomes, particularly when performed early in refractory cases. However, pneumonectomy remains a last-resort option, reserved for extensive disease with no salvageable lung tissue, offering a potentially life-saving solution. Optimizing outcomes while minimizing complications requires careful, individualized decision-making based on the patient’s disease progression and overall clinical status.
- VATS
- VATS is preferred for smaller, localized lesions and is associated with reduced postoperative morbidity compared to open thoracotomy. This minimally invasive approach avoids rib displacement, making it a favorable option in stable patients who can tolerate double-lumen tube placement for thoracic exploration. However, in unstable patients with massive chest wall disruption, significant air loss, or hemoptysis, open thoracotomy may be more appropriate.[66]
- Lobectomy
- Lobectomy is a definitive surgical option for large, infected, or necrotic lesions not amenable to conservative management. This procedure is particularly effective in reducing the risks of infection, parenchymal necrosis, and the need for additional surgeries. Early lobectomy may be indicated in cases of:
- Persistent air leaks
- Progressive pneumatocele enlargement
- Compression of functional lung parenchyma
- Extensive lung abscess surrounded by necrotic tissue
- Failure of conservative treatment or bronchoscopic interventions.
- Despite its benefits, lobectomy is unsuitable if the amount of salvageable lung tissue is negligible or if retaining necrotic tissue poses risks.
- Lobectomy is a definitive surgical option for large, infected, or necrotic lesions not amenable to conservative management. This procedure is particularly effective in reducing the risks of infection, parenchymal necrosis, and the need for additional surgeries. Early lobectomy may be indicated in cases of:
- Pneumonectomy
- Pneumonectomy is reserved for cases of extensive disease with no salvageable lung tissue or when less invasive approaches fail. This procedure aims to remove large pneumatoceles or necrotic lung tissue, improving ventilation and cardiorespiratory function. Pediatric pneumonectomy has been documented, with long-term outcomes generally favorable despite potential complications like scoliosis or mild spirometric changes.[67] Removing as little lung parenchyma as possible is ideal, although extensive disease may necessitate a more aggressive approach.
(B3)
Adjunctive Measures
Surgical options may still be viable after stabilization in cases where patients are unsuitable for early intervention, such as those requiring extracorporeal membrane oxygenation (ECMO). ECMO can serve as a bridge to definitive treatments like pneumonectomy. Additionally, late thoracotomy has been reported up to 6 months after trauma due to pneumonic infiltration and persistent cavitary size.
Special considerations
- Traumatic pneumatoceles
- The management of traumatic pneumatoceles is primarily conservative, as most resolve spontaneously over time, though complications may necessitate surgical intervention. Radiological resolution varies depending on lesion size and content, typically averaging 3 months but ranging from 4 weeks to 6 months. Larger lesions or those filled with blood may require significantly longer periods, with an average resolution time of 5 months for those exceeding 2 cm in diameter or containing blood, as reported by Chon et al.[11][68] Notably, some pneumatoceles may transiently enlarge during the first 2 weeks posttrauma, potentially prompting unnecessary diagnostic or therapeutic procedures that could increase the risk of complications.
- Traumatic pneumatoceles complicated by secondary infection or associated with respiratory compromise demand more active management. Positive-pressure ventilation may exacerbate lesion enlargement, hypoxemia, and inadequate ventilation, necessitating surgical interventions such as VATS or open thoracotomy with tube decompression. In cases of secondary infection, antibiotic therapy tailored to sputum culture and sensitivity is the first step. Percutaneous drainage is indicated if an infected pneumatocele is larger than 2 cm or sepsis persists beyond 72 hours of antibiotics. Early CT-guided catheter drainage is recommended if no clinical improvement is observed, with escalation to thoracotomy or thoracoscopy for refractory cases.
- Long-term follow-up with chest radiography or other imaging modalities is essential to ensure resolution.[11] While the prognosis for traumatic pneumatoceles is excellent in most cases, with lesions typically disappearing within 3 months, late thoracotomy may occasionally be required for persistent cavitary lesions or associated pneumonic infiltration up to 6 months posttrauma. Cases like the giant hemopneumatocele described by Romero et al, which resolved conservatively, underscore the importance of individualized treatment strategies, particularly in patients with multiple thoracic injuries who may not tolerate surgery.
- Pediatric pneumatoceles
- Pneumatoceles in pediatric populations present unique challenges due to the distinct physiology and vulnerabilities of children compared to adults. Ventilator-induced pneumatoceles are a subset of intrathoracic air leaks commonly observed in premature neonates and are considered markers of ventilator-induced lung injury. These lesions arise from alveolar overdistension and rupture, often caused by high mean airway pressures during mechanical ventilation. The fragile and immature lung parenchyma of preterm neonates predisposes them to such complications, making this condition a significant concern in neonatal intensive care settings.
- Historically, neonatal pneumatoceles were frequently described as complications of ventilator-induced air leak conditions, particularly during the 1970s and 1980s. During this era, high-pressure ventilation was more common, leading to increased incidence of such complications. However, advances in neonatal care, including the introduction of surfactant replacement therapy and the adoption of lung-protective ventilation strategies, have significantly reduced the incidence of all intrathoracic air leaks, including pneumatoceles, in neonates.
- The clinical significance of ventilator-induced pneumatoceles lies in their association with increased morbidity and mortality in premature infants. Despite this, the condition typically responds well to conservative management. Reducing mean airway pressure is the cornerstone of treatment, as it minimizes further lung damage and promotes spontaneous resolution of the pneumatocele. Modern lung-protective strategies, such as permissive hypercapnia and high-frequency oscillatory ventilation, further enhance the outcomes by preventing overdistension and reducing ventilator-induced injury.
- Surgical intervention is rarely necessary for ventilator-induced pneumatoceles, as most resolve with conservative measures.[4][16][69] The focus remains on optimizing ventilatory support to allow for natural healing while minimizing the risk of additional complications, such as pneumothorax or secondary infection.
- Pediatric cases of pneumatoceles are often associated with infections, such as pneumonia, and may be complicated by conditions like bronchopulmonary fistulae. In severe cases involving extensive disease, management may require advanced interventions. For example, venovenous (V-V) ECMO can be employed as a salvage measure in children with extensive disease affecting multiple lobes.[67] In rare, refractory cases, pneumonectomy may be necessary to manage enlarging pneumatoceles or severe bronchopulmonary fistulae. While preoperative care in such cases has not been well-defined in the literature, outcomes following pneumonectomy are generally positive.[67]
- Long-term complications after pediatric pneumonectomy are infrequent and typically mild. Reported issues include scoliosis and changes in spirometry values, likely attributable to the anatomical adaptations following lung resection. Despite these potential changes, most children demonstrate no significant impairments in respiratory function or exercise capacity. These outcomes underscore the resilience of pediatric patients and highlight the importance of tailoring management strategies to the unique needs of this population.
(B3)
Differential Diagnosis
Differential Diagnosis
The differential diagnosis for a pneumatocele includes a wide range of conditions that can mimic its radiological appearance or clinical presentation. These include:
- Infectious and inflammatory causes
- Lung abscess
- Postinflammatory pneumatocele
- Tuberculosis
- Mycosis
- Coccidioidomycosis
- Fungus ball
- Aspergilloma
- Empyema
- Pneumonia
- Tuberculoma
- Congenital and structural anomalies
- Bronchial cyst
- Pulmonary sequestration
- Cystic adenomatoid malformation
- Congenital cysts
- Cystic conditions
- Cystic fibrosis
- Primary ciliary dyskinesia
- Primary immunodeficiencies
- Systemic and genetic disorders
- Marfan syndrome
- Proteus syndrome
- Neurofibromatosis (pulmonary cystic changes, blebs, bullae, giant bulla)
- Neoplastic and granulomatous conditions
- Granulomatosis with polyangiitis
- Langerhans cell histiocytosis
- Hydatid cysts
- Lung cancer
- Metastatic neoplasm
- Trauma and other structural conditions
- Herniation of viscera
- Esophageal rupture
Radiological and Pathological Considerations
Cysts are thin-walled spaces (<2 mm) that lack associated pulmonary emphysema on CT scans and are distinguished from bullae by their greater wall thickness and location. Bullae, in contrast, are airspaces larger than 1 cm with an imperceptibly thin wall (<1 mm) and are typically subpleural, often occurring in the context of emphysema. Cavitary lesions, on the other hand, are gas-filled spaces characterized by thick, irregular walls and are commonly seen within areas of pulmonary consolidation, masses, or nodules. These cavities are distinct from cysts due to their irregular shape and thicker walls and are frequently associated with infections, chronic systemic diseases, or malignancies.[70][71] A thorough clinical history, imaging studies, and microbiological or histopathological investigations are crucial to narrowing the differential diagnosis and guiding appropriate management.
Prognosis
The prognosis of pneumatoceles generally depends on the underlying cause, size, and complications. In most cases, pneumatoceles are self-limiting and resolve spontaneously without significant long-term effects. This is particularly true for small, traumatic pneumatoceles, which typically undergo spontaneous resolution within weeks to months, especially when associated with minor injuries. For instance, many children with traumatic pneumatoceles show complete resolution within 6 months without the need for invasive treatment. However, larger pneumatoceles or those associated with severe trauma or pneumonia may persist for longer periods and pose greater risks, such as rupture or infection.
Pneumatoceles resulting from pneumonia, especially when complicated by bacterial infections like Streptococcus pneumoniae or Staphylococcus aureus, can have a more guarded prognosis. Infected pneumatoceles are at a higher risk of leading to more severe complications, including lung abscess, empyema, and pyopneumothorax, which can increase morbidity and mortality rates. These cases may require surgical intervention, including drainage or resection and extended antibiotic therapy. Additionally, pneumatoceles associated with conditions such as cystic fibrosis or immunocompromised states (eg, acquired immunodeficiency syndrome) may have a more complicated course due to the underlying disease process and the risk of recurrent infections.
For patients with pneumatoceles caused by mechanical ventilation, such as in premature infants or those with acute respiratiry distress syndrome, the prognosis is influenced by the severity of the lung injury and the effectiveness of ventilation management. In these cases, the development of a pneumatocele can be indicative of significant pulmonary trauma, which may lead to ongoing respiratory issues or the need for long-term respiratory support. The overall prognosis of pneumatoceles is more favorable in young, otherwise healthy individuals, particularly if the pneumatocele is small, noninfected, and does not lead to significant complications.
However, in the presence of large pneumatoceles, secondary infections, or complications like pneumothorax, the prognosis can be more serious and may require more intensive management. Follow-up care with imaging and clinical monitoring is essential, as unresolved or enlarging pneumatoceles may require surgical intervention to prevent further complications. In general, with appropriate management, most patients experience a good outcome, although severe cases can result in long-term respiratory issues or, in rare instances, death.
Complications
Pneumatoceles, though often self-limited and generally resolving without the need for invasive treatment, can lead to a range of complications. These complications can arise from the nature of the pneumatocele itself or from errors in diagnosis and subsequent intervention, which can exacerbate the condition. Below are the key complications associated with pneumatoceles:
- Infection
- Infected pneumatoceles are one of the most common complications, particularly in traumatic or post-pneumonia cases. Infection may lead to the development of empyema or lung abscess. Infected traumatic pulmonary pneumatoceles have higher mortality rates compared to typical lung abscesses. Pulmonary contusion impairs bacterial clearance and can increase the risk of secondary infections. The treatment for infected traumatic pulmonary pneumatoceles is similar to that of lung abscesses and typically includes antibiotics, drainage of pus if necessary, and supportive care.[72]
- Rupture
- Rupture of a pneumatocele is a critical complication that may result in pneumothorax or even tension pneumothorax. In this life-threatening condition, the pressure from trapped air compresses the lung and mediastinal structures, leading to severe respiratory distress and cardiovascular instability. The rupture may occur spontaneously or due to external trauma. Tension pneumothorax requires emergency treatment, typically with conventional tube thoracostomy (chest tube placement) to relieve the trapped air and reexpand the lung. If left untreated, this can lead to a mediastinal shift and severe hemodynamic instability.
- Pneumomediastinum
- Pneumomediastinum, the presence of air in the mediastinum, is another potential complication. This can occur due to pneumatocele rupture or air leakage from the cystic space into the surrounding tissues. Pneumomediastinum may lead to chest pain, dyspnea, and subcutaneous emphysema. In some cases, it can progress to life-threatening complications such as tension pneumomediastinum.[73]
- Pyopneumothorax
- Pyopneumothorax, a combination of pneumothorax and infection (pus accumulation in the pleural space), can develop when a pneumatocele becomes infected. The accumulation of pus and air in the pleural space can cause further respiratory distress and necessitate drainage. This condition is also associated with a higher mortality rate compared to a simple pneumothorax, making early detection and intervention crucial.
- Hemothorax and hemopneumothorax
- Trauma-related pneumatoceles may also be complicated by hemothorax (blood in the pleural space) or hemopneumothorax (air and blood in the pleural space). These conditions often result from pulmonary laceration and the escape of blood into the pleural cavity. Both conditions can cause significant respiratory distress and require urgent treatment, typically in the form of chest tube placement or surgical intervention, depending on the severity of the bleeding.
- Fistulous pleural-extrathoracic communication
- In untreated or inadequately treated tuberculosis cases, pneumatoceles may develop a fistulous communication between the pleura and the extrathoracic environment. This is more likely to occur in patients who have not completed anti tuberculosis treatment. Such fistulas may cause persistent air leaks or fluid drainage from the pleural cavity to the outside of the body, necessitating surgical intervention for closure.
- Continuous positive airway pressure complications
- In those who are mechanically ventilated, particularly those on continuous positive airway pressure, pneumatocele enlargement can occur, leading to compression of adjacent lung tissue. This increased compression can lead to worsening hypoxemia, which may result in cardiopulmonary instability.[11] Monitoring pneumatocele size and managing ventilation settings is essential to avoid these complications, especially in critically ill individuals.
- Obstruction of adjacent airway
- Large pneumatoceles can exert pressure on nearby bronchial structures, potentially leading to airway obstruction. This can result in atelectasis (collapse of a part of the lung) or impaired ventilation, further compromising respiratory function. Airway obstruction also increases the risk of infection, as secretions become trapped in the obstructed region of the lung.
- Fibrosis and scarring
- Chronic pneumatoceles, especially those that result from repeated infections or trauma, can lead to fibrosis or scarring of the lung tissue. This process may reduce lung compliance and capacity, leading to chronic respiratory insufficiency. If left untreated, this could result in restrictive lung disease, which would require ongoing medical management.
- Chronic respiratory symptoms
- Chronic respiratory symptoms such as cough, dyspnea, and wheezing may develop in some cases, especially those with multiple or large pneumatoceles. These persistent symptoms can impair quality of life and may require long-term monitoring or surgical intervention if the symptoms worsen or lung function deteriorates.
Consultations
Management of pneumatoceles often requires coordinated care between multiple specialists to ensure optimal outcomes. In pediatric cases, a pediatrician and pediatric pulmonologist typically lead the planning and management of treatment. A cardiothoracic surgeon plays a crucial role when surgical intervention is required, such as for a lobectomy, chest tube placement, pigtail catheter insertion, or fibrin sealant injections.
In adult cases, management decisions are more variable and depend on the patient's condition and the underlying cause of the pneumatocele. Surgeons, including trauma and cardiothoracic specialists, work with pulmonologists to tailor treatment strategies. Critically ill or hemodynamically unstable patients necessitate close involvement of the intensive care unit team for advanced monitoring and supportive care. Effective interprofessional collaboration is essential for optimizing outcomes for pediatric and adult patients.
Deterrence and Patient Education
Proper deterrence and patient education are crucial in preventing complications and recurrence of pneumatoceles and optimizing patient outcomes. Pneumatoceles, which can result from trauma, pneumonia, or mechanical ventilation, require comprehensive management strategies that include preventive measures and clear communication with patients and their families. For trauma-related pneumatoceles, the primary prevention strategy involves reducing the risk of chest injuries. This includes promoting motor vehicle safety by ensuring the use of seatbelts and appropriate child safety seats, encouraging protective gear in sports, and fall prevention in vulnerable populations such as older adults and children. Educating individuals who have experienced trauma about the importance of early medical evaluation can help detect pulmonary injuries like pneumatoceles, even in the absence of immediate symptoms. After trauma, patients should also be informed that early intervention can prevent complications like infection, pneumothorax, or further tissue damage.
Regarding pneumonia, vaccination is key in preventing infections that can lead to pneumatoceles. Vaccines like the pneumococcal vaccine and flu shot should be emphasized, particularly for high-risk groups such as young children, older adults, and those with underlying health conditions. Additionally, educating patients on the importance of completing prescribed antibiotic courses can help prevent complications like pneumatoceles from persistent infection. Recognizing pneumonia symptoms early, such as cough, fever, and difficulty breathing, is vital for timely treatment and reducing risks associated with pneumatocele formation.
Proper ventilator settings and monitoring are essential for patients undergoing mechanical ventilation or continuous positive airway pressure therapy. Patients and caregivers should be educated on the potential for pneumatocele formation, especially in neonates or premature infants, and the need for regular follow-up to detect early signs of complications. Those on mechanical ventilation should understand the importance of continued monitoring, particularly when new respiratory symptoms arise.
After trauma or surgery, patients should be educated on the need for follow-up care, including imaging studies to detect pneumatocele formation. Even if they feel better postinjury, pneumatocele development can occur weeks later, and early imaging can detect and address any complications before they become severe. Patients should also be instructed to seek immediate medical care if they experience worsening symptoms, such as sudden chest pain, difficulty breathing, or coughing up blood.
For individuals with traumatic or infectious pneumonia-associated pneumatoceles, regular follow-up appointments with their healthcare provider are necessary. Imaging studies like chest x-rays or CT scans should be used to monitor for recurrence or enlargement of existing pneumatoceles. Patients should be advised on lifestyle modifications, such as smoking cessation, especially for those with existing lung disease, as smoking can worsen lung health and increase the risk of further pulmonary complications.
In terms of preventing infections, patients should be educated on the importance of hand hygiene, cough etiquette, and avoiding exposure to respiratory pathogens, particularly in hospital settings or areas with high-risk individuals. Pneumatoceles are susceptible to secondary infections, especially in the context of pulmonary contusions, which impair bacterial clearance. Infected pneumatoceles may require more intensive treatment and carry a higher mortality rate than typical lung abscesses. Patients should recognize signs of infection, such as fever or worsening cough, to seek prompt medical attention.
Finally, patients recovering from traumatic or infectious pneumatocele formation should be encouraged to seek psychosocial support, as the condition can have significant emotional and psychological impacts, particularly in children or those who have experienced trauma. Support from caregivers, counselors, or support groups can help manage anxiety and improve adherence to follow-up care. Educating patients about the need for physical and psychological care can help them cope with the challenges of recovery and reduce the risk of complications, ensuring better long-term outcomes.
In conclusion, a multifaceted approach to deterrence and patient education is essential for preventing and managing pneumatoceles. By focusing on trauma prevention, vaccination, early detection of pneumonia, proper mechanical ventilation care, and regular monitoring after trauma or surgery, healthcare providers can reduce the incidence and severity of pneumatocele complications. Empowering patients with knowledge about the importance of prevention, recognizing symptoms, and adhering to follow-up care will contribute to better outcomes and reduce the risks associated with this condition.
Pearls and Other Issues
Pneumatoceles are critical complications that can arise following trauma, infection, burns, or chemical-induced lung injury. Accurate diagnosis and differentiation from other pathological conditions are crucial, as the management strategies for pneumatoceles differ significantly based on their etiology. Misdiagnosis or delayed recognition can result in inappropriate interventions, potentially exacerbating patient outcomes and leading to further harm. Early and precise identification is essential to guide appropriate treatment and ensure optimal care.
Enhancing Healthcare Team Outcomes
Effective management of pneumatoceles, whether caused by trauma, infection, or underlying conditions, requires a multidisciplinary approach to optimize patient-centered care, safety, and outcomes. Clinicians play a central role in diagnosis, employing imaging and clinical assessments to determine severity and underlying etiology. Advanced clinicians, such as nurse practitioners and physician assistants, extend this role by monitoring the patient's progress, coordinating follow-ups, and educating the patient and family about the condition and its management. Nurses ensure continuity of care by assessing vital signs, managing oxygen therapy, and providing wound care or monitoring for signs of infection. Pharmacists contribute by reviewing medication regimens, including antibiotics for infection-induced pneumatoceles or analgesics for pain management, ensuring appropriate dosing, and monitoring for adverse effects.
Interprofessional communication and care coordination are crucial to enhancing patient safety and team performance. Regular multidisciplinary rounds provide a platform to share insights, adjust care plans, and address potential complications, such as pneumothorax or superimposed infections. Clear documentation and standardized protocols help ensure seamless care transitions, particularly when transferring patients between hospital units or discharging them to outpatient settings. Collaborative decision-making empowers all team members to contribute their expertise, fostering trust and improving outcomes. A patient-centered approach that prioritizes shared decision-making and education further enhances adherence to treatment plans and promotes optimal recovery.
Media
(Click Image to Enlarge)
References
Devgan BK, Brodeur AE. Apical pneumatocele. Archives of otolaryngology (Chicago, Ill. : 1960). 1976 Feb:102(2):121-3 [PubMed PMID: 1247420]
Level 3 (low-level) evidenceChen Z, Lian XH, Ye WQ, Wen KY, Chen JY, Rong XB, Wang XB. [Spiral CT findings of pulmonary infection induced by intravenous injection of heroin]. Di 1 jun yi da xue xue bao = Academic journal of the first medical college of PLA. 2005 Jun:25(6):742-4 [PubMed PMID: 15958328]
Level 2 (mid-level) evidencede Bie HM, van Toledo-Eppinga L, Verbeke JI, van Elburg RM. Neonatal pneumatocele as a complication of nasal continuous positive airway pressure. Archives of disease in childhood. Fetal and neonatal edition. 2002 May:86(3):F202-3 [PubMed PMID: 11978755]
Level 3 (low-level) evidenceAl-Ghafri M, Al-Hanshi S, Al-Ismaily S. Two Cases of Pneumatoceles in Mechanically Ventilated Infants. Oman medical journal. 2015 Jul:30(4):299-302. doi: 10.5001/omj.2015.59. Epub [PubMed PMID: 26366266]
Level 3 (low-level) evidenceBahceci Erdem S, Nacaroglu HT, Isgüder R, Unsal Karkiner CS, Alper H, Can D. Pulmonary complications of chemical pneumonía: a case report. Archivos argentinos de pediatria. 2016 Aug 1:114(4):e245-8 [PubMed PMID: 27399025]
Level 3 (low-level) evidenceVerma SK, Kapoor N, Bhaskar R, Upadhyay R. Pyopneumothorax following suicidal kerosene ingestion. BMJ case reports. 2012 Dec 18:2012():. doi: 10.1136/bcr-2012-007795. Epub 2012 Dec 18 [PubMed PMID: 23257648]
Level 3 (low-level) evidenceAziz AA, Abdullah AF, Mahmud A. Lung abscess rather than pneumatocele following kerosene ingestion. British journal of hospital medicine (London, England : 2005). 2007 Nov:68(11):616-7 [PubMed PMID: 18087856]
Level 3 (low-level) evidenceSergiacomi GL, Mazzetti di Pietralata G, Moscone A, Ciorra DM, Motta MR, Palmisano P, Simonetti G. [Role of diagnostic imaging in the assessment of lung complications of burns]. La Radiologia medica. 1998 Nov:96(5):454-61 [PubMed PMID: 10051868]
Skowasch D, Pizarro C, Valipour A, Wilhelm KE, Thomas D, Nickenig G. Endobronchial valve-induced pneumatocele: a case report. Pneumologie (Stuttgart, Germany). 2013 Nov:67(11):639-40. doi: 10.1055/s-0033-1344640. Epub 2013 Sep 2 [PubMed PMID: 23999694]
Level 3 (low-level) evidenceMatuszczak E, Oksiuta M, Hermanowicz A, Dębek W. Traumatic pneumatocele in an 11-year-old boy - report of a rare case and review of the literature. Kardiochirurgia i torakochirurgia polska = Polish journal of cardio-thoracic surgery. 2017 Mar:14(1):59-62. doi: 10.5114/kitp.2017.66934. Epub 2017 Mar 31 [PubMed PMID: 28515753]
Level 3 (low-level) evidencePhillips B, Shaw J, Turco L, McDonald D, Carey J, Balters M, Wagner M, Bertellotti R, Cornell DL, Agrawal DK, Asensio JA. Traumatic pulmonary pseudocyst: An underreported entity. Injury. 2017 Feb:48(2):214-220. doi: 10.1016/j.injury.2016.12.006. Epub 2016 Dec 12 [PubMed PMID: 27986273]
Armstrong LB, Mooney DP. Pneumatoceles in pediatric blunt trauma: Common and benign. Journal of pediatric surgery. 2018 Jul:53(7):1310-1312. doi: 10.1016/j.jpedsurg.2017.08.002. Epub 2017 Aug 7 [PubMed PMID: 28917584]
Aissaoui O, Alharrar R. Traumatic pulmonary pseudocyst: a rare complication of blunt thoracic injury. The Pan African medical journal. 2019:32():180. doi: 10.11604/pamj.2019.32.180.18592. Epub 2019 Apr 11 [PubMed PMID: 31312294]
Kunyoshi V, Cataneo DC, Cataneo AJ. Complicated pneumonias with empyema and/or pneumatocele in children. Pediatric surgery international. 2006 Feb:22(2):186-90 [PubMed PMID: 16362309]
Level 2 (mid-level) evidenceKaira K, Ishizuka T, Yanagitani N, Sunaga N, Hisada T, Mori M. Pulmonary traumatic pneumatocele and hematoma. Japanese journal of radiology. 2009 Feb:27(2):100-2. doi: 10.1007/s11604-008-0296-x. Epub 2009 Mar 12 [PubMed PMID: 19373539]
Level 3 (low-level) evidenceAmitai I, Mogle P, Godfrey S, Aviad I. Pneumatocele in infants and children. Report of 12 cases. Clinical pediatrics. 1983 Jun:22(6):420-2 [PubMed PMID: 6839622]
Level 3 (low-level) evidenceChen J, Wu J, Hao S, Yang M, Lu X, Chen X, Li L. Long term outcomes in survivors of epidemic Influenza A (H7N9) virus infection. Scientific reports. 2017 Dec 8:7(1):17275. doi: 10.1038/s41598-017-17497-6. Epub 2017 Dec 8 [PubMed PMID: 29222500]
Sugimoto H, Era Y, Sugimoto K. Pneumatocele after recovering from COVID-19. BMJ case reports. 2021 Apr 19:14(4):. doi: 10.1136/bcr-2021-242729. Epub 2021 Apr 19 [PubMed PMID: 33875514]
Level 3 (low-level) evidenceGuandalini M, Steinke K, Francesconi A. Pulmonary metastases from cervical adenocarcinoma regress to a 'hole' lot of nothing. Journal of thoracic oncology : official publication of the International Association for the Study of Lung Cancer. 2009 Apr:4(4):552-4. doi: 10.1097/JTO.0b013e3181952947. Epub [PubMed PMID: 19333074]
Level 3 (low-level) evidenceHamad AM, El-Saka HA. Is it a bulla or a pneumatocele? European journal of cardio-thoracic surgery : official journal of the European Association for Cardio-thoracic Surgery. 2021 Jul 14:60(1):203. doi: 10.1093/ejcts/ezaa481. Epub [PubMed PMID: 33480415]
Purdy GD, Cullen M, Yedlin S, Bedard MP. An unusual neonatal case presentation: Streptococcus pneumoniae pneumonia with abscess and pneumatocele formation. Journal of perinatology : official journal of the California Perinatal Association. 1987 Fall:7(4):378-81 [PubMed PMID: 3333168]
Level 3 (low-level) evidenceCaksen H, Oztürk MK, Uzüm K, Yüksel S, Ustünbaş HB. Pulmonary complications in patients with staphylococcal sepsis. Pediatrics international : official journal of the Japan Pediatric Society. 2000 Jun:42(3):268-71 [PubMed PMID: 10881583]
Level 2 (mid-level) evidenceIshiguchi S, Nishioka K, Kanetake Y, Sugiyama K, Shibayama M. [Pneumatocele accompanying staphylococcal pneumonia]. Rinsho hoshasen. Clinical radiography. 1966 Aug:11(8):644-53 [PubMed PMID: 5182446]
Sandhu JS, Goodman PC. Pulmonary cysts associated with Pneumocystis carinii pneumonia in patients with AIDS. Radiology. 1989 Oct:173(1):33-5 [PubMed PMID: 2789413]
Gurney JW, Bates FT. Pulmonary cystic disease: comparison of Pneumocystis carinii pneumatoceles and bullous emphysema due to intravenous drug abuse. Radiology. 1989 Oct:173(1):27-31 [PubMed PMID: 2789412]
Albitar HAH, Abu Saleh OM. Pneumocystis Pneumonia Complicated by Extensive Diffuse Pneumatoceles. The American journal of medicine. 2019 May:132(5):e562-e563. doi: 10.1016/j.amjmed.2018.12.029. Epub 2019 Jan 16 [PubMed PMID: 30659808]
Papageorgiou A, Bauer CR, Fletcher BD, Stern L. Klebsiella pneumonia with pneumatocele formation in a newborn infant. Canadian Medical Association journal. 1973 Dec 15:109(12):1217-9 [PubMed PMID: 4586073]
Wan-Hsiu L, Sheng-Hsiang L, Tsu-Tuan W. Pneumatocele formation in adult pulmonary tuberculosis during antituberculous chemotherapy: a case report. Cases journal. 2009 Sep 9:2():8570. doi: 10.4076/1757-1626-2-8570. Epub 2009 Sep 9 [PubMed PMID: 19918388]
Level 3 (low-level) evidenceLim GY, Kim OH, Kim HW, Lee KS, Kang KH, Song HR, Cho TJ. Pulmonary manifestations in Proteus syndrome: pulmonary varicosities and bullous lung disease. American journal of medical genetics. Part A. 2011 Apr:155A(4):865-9. doi: 10.1002/ajmg.a.33926. Epub 2011 Mar 15 [PubMed PMID: 21412980]
Level 3 (low-level) evidenceLysy J, Werczberger A, Globus M, Chowers I. Pneumatocele formation in a patient with Proteus mirabilis pneumonia. Postgraduate medical journal. 1985 Mar:61(713):255-7 [PubMed PMID: 3885205]
Level 3 (low-level) evidenceColling J, Allaouchiche B, Floccard B, Pilleul F, Monneuse O, Tissot E. Pneumatocele formation in adult Escherichia coli pneumonia revealed by pneumothorax. The Journal of infection. 2005 Oct:51(3):e109-11 [PubMed PMID: 16230186]
Level 3 (low-level) evidenceHunt JP, Buechter KJ, Fakhry SM. Acinetobacter calcoaceticus pneumonia and the formation of pneumatoceles. The Journal of trauma. 2000 May:48(5):964-70 [PubMed PMID: 10823547]
Level 3 (low-level) evidenceChitayat D, Diamant S, Lazevnick R, Spirer Z. Hemophilus influenzae type B pneumonia with pneumatocele formation. Clinical pediatrics. 1980 Feb:19(2):151-2 [PubMed PMID: 6965366]
Level 3 (low-level) evidenceKim SH, Chung YT, Lee KD, Seon KY, Lee JH, Lee SH, Choi SH. Infected pneumatocele following anaerobic pneumonia in adult. The Korean journal of internal medicine. 2005 Dec:20(4):343-5 [PubMed PMID: 16491835]
Level 3 (low-level) evidenceStulz P, Schmitt HE, Hasse J, Grädel E. Traumatic pulmonary pseudocysts and paramediastinal air cyst: two rare complications of blunt chest trauma. The Journal of trauma. 1984 Sep:24(9):850-3 [PubMed PMID: 6481838]
Level 3 (low-level) evidenceSantos GH. Posttraumatic pneumatocele. Journal of the American College of Surgeons. 2005 Aug:201(2):319; author reply 319-20 [PubMed PMID: 16038834]
Level 3 (low-level) evidenceYang TC, Huang CH, Yu JW, Hsieh FC, Huang YF. Traumatic pneumatocele. Pediatrics and neonatology. 2010 Apr:51(2):135-8. doi: 10.1016/S1875-9572(10)60024-1. Epub [PubMed PMID: 20417466]
Level 3 (low-level) evidenceLee E, Son SK, Yoon J, Cho HJ, Yang SI, Jung S, Do KH, Cho YA, Lee SY, Park DU, Hong SJ. Two Cases of Chloromethylisothiazolinone and Methylisothiazolinone-associated Toxic Lung Injury. Journal of Korean medical science. 2018 Apr 16:33(16):e119. doi: 10.3346/jkms.2018.33.e119. Epub 2018 Apr 16 [PubMed PMID: 29651817]
Level 3 (low-level) evidenceCampbell JB. Pneumatocele formation following hydrocarbon ingestion. The American review of respiratory disease. 1970 Mar:101(3):414-8 [PubMed PMID: 5414060]
Level 3 (low-level) evidenceDuttaroy DD, Jagtap J, Bansal U, Duttaroy B. Tuberculous pulmonary pneumatocele communicating extrathoracically. Thorax. 2006 Aug:61(8):738 [PubMed PMID: 16877697]
Level 3 (low-level) evidenceHong SJ, Hwang S, Hwang SH, Kim DY, Kong SJ. Extrathoracic communication of persistent pulmonary tuberculous pneumatocele. Respiration; international review of thoracic diseases. 2011:81(3):247-8. doi: 10.1159/000321251. Epub 2010 Nov 5 [PubMed PMID: 21051873]
Level 3 (low-level) evidenceTeixeira J, Silva T, Correia-Pinto J, Gonçalves A. Pneumatocele or something more? BMJ case reports. 2016 Jan 12:2016():. doi: 10.1136/bcr-2015-213241. Epub 2016 Jan 12 [PubMed PMID: 26759441]
Level 3 (low-level) evidenceLewars J. Pneumatocele Manifestation in Early Recovery Period of COVID-19: A Case Study. Cureus. 2023 Jul:15(7):e41259. doi: 10.7759/cureus.41259. Epub 2023 Jul 1 [PubMed PMID: 37529801]
Level 3 (low-level) evidenceHsieh MS, Chen CK, Wong WW, Huang CS. Rapid-growth pneumatocele mimics massive pneumothorax in a HIV-positive patient. Thorax. 2013 Mar:68(3):307-8. doi: 10.1136/thoraxjnl-2012-201990. Epub 2012 Jun 26 [PubMed PMID: 22735688]
Level 3 (low-level) evidenceShamberger RC, Wohl ME, Perez-Atayde A, Hendren WH. Pneumatocele complicating hyperimmunoglobulin E syndrome (Job's Syndrome). The Annals of thoracic surgery. 1992 Dec:54(6):1206-8 [PubMed PMID: 1449312]
Level 3 (low-level) evidenceALMKLOV JR, HATOFF A. Pneumatocele during the course of pneumonia in children. American journal of diseases of children (1911). 1946 Nov:72(5):521-8 [PubMed PMID: 20276997]
Hazer S, Orhan Söylemez UP. Clinical features, diagnosis, and treatment of traumatic pulmonary pseudocysts. Ulusal travma ve acil cerrahi dergisi = Turkish journal of trauma & emergency surgery : TJTES. 2018 Jan:24(1):49-55. doi: 10.5505/tjtes.2017.56023. Epub [PubMed PMID: 29350368]
Joseph LJ, Bromiker R, Toker O, Schimmel MS, Goldberg S, Picard E. Unilateral lung intubation for pulmonary air leak syndrome in neonates: a case series and a review of the literature. American journal of perinatology. 2011 Feb:28(2):151-6. doi: 10.1055/s-0030-1263298. Epub 2010 Aug 11 [PubMed PMID: 20703979]
Level 3 (low-level) evidenceDesrues B, Delaval P, Motreff C, Kernec J, Dormoy C, Pencolé C, Bergeron D, Malledan Y, Saint-Marc C. [Post-traumatic pneumatocele and hemato-pneumatocele of the lung. Apropos of 3 cases]. Revue des maladies respiratoires. 1988:5(1):67-70 [PubMed PMID: 3368637]
Level 3 (low-level) evidenceGarcía Herrera AN, Barber Ansón M. Post-traumatic haemato-pneumatocele. Medicina clinica. 2018 Sep 21:151(6):253. doi: 10.1016/j.medcli.2017.09.028. Epub 2017 Dec 13 [PubMed PMID: 29246563]
Expert Panel on Pediatric Imaging, Chan SS, Kotecha MK, Rigsby CK, Iyer RS, Alazraki AL, Anupindi SA, Bardo DME, Brown BP, Chandra T, Dorfman SR, Garber MD, Moore MM, Nguyen JC, Shet NS, Siegel A, Valente JH, Karmazyn B. ACR Appropriateness Criteria® Pneumonia in the Immunocompetent Child. Journal of the American College of Radiology : JACR. 2020 May:17(5S):S215-S225. doi: 10.1016/j.jacr.2020.01.033. Epub [PubMed PMID: 32370966]
Yan JH, Yu N, Wang YH, Gao YB, Pan L. Lung ultrasound vs chest radiography in the diagnosis of children pneumonia: Systematic evidence. Medicine. 2020 Dec 11:99(50):e23671. doi: 10.1097/MD.0000000000023671. Epub [PubMed PMID: 33327356]
Level 1 (high-level) evidenceBarbick B, Cothren CC, Zimmerman MA, Moore EE. Posttraumatic pneumatocele. Journal of the American College of Surgeons. 2005 Feb:200(2):306-7 [PubMed PMID: 15664109]
Level 3 (low-level) evidenceShen HN, Lu FL, Wu HD, Yu CJ, Yang PC. Management of tension pneumatocele with high-frequency oscillatory ventilation. Chest. 2002 Jan:121(1):284-6 [PubMed PMID: 11796465]
Level 3 (low-level) evidenceDe Bisschop B, Peeters L, Sonnaert M. Successful conservative managements of extensive pneumatoceles in a preterm girl: A case report. Journal of neonatal-perinatal medicine. 2021:14(1):139-142. doi: 10.3233/NPM-190382. Epub [PubMed PMID: 31903998]
Level 3 (low-level) evidenceMaranella E, Conte E, Di Natale C, Coclite E, Di Fabio S. Disseminated, large-sized neonatal pneumatoceles: the wait-and-see strategy. Pediatric pulmonology. 2014 Mar:49(3):E69-71. doi: 10.1002/ppul.22831. Epub 2013 Jun 21 [PubMed PMID: 23794463]
Level 3 (low-level) evidenceSchimpl G, Schneider U. Traumatic pneumatoceles in an infant: case report and review of the literature. European journal of pediatric surgery : official journal of Austrian Association of Pediatric Surgery ... [et al] = Zeitschrift fur Kinderchirurgie. 1996 Apr:6(2):104-6 [PubMed PMID: 8740134]
Level 3 (low-level) evidenceYao CW, Shen TC, Tu CY. Giant pneumatocele with lung herniation. Internal medicine (Tokyo, Japan). 2013:52(20):2377-8 [PubMed PMID: 24126406]
Level 3 (low-level) evidenceGholap PM, Anuroop Shankar S. A rare case of tension pneumatocele. The Indian journal of tuberculosis. 2017 Oct:64(4):327-329. doi: 10.1016/j.ijtb.2016.09.020. Epub 2016 Dec 28 [PubMed PMID: 28941858]
Level 3 (low-level) evidenceFujii AM, Moulton S. Percutaneous catheter evacuation of a pneumatocele in an extremely premature infant with respiratory failure. Journal of perinatology : official journal of the California Perinatal Association. 2003 Sep:23(6):516-8 [PubMed PMID: 13679944]
Level 3 (low-level) evidenceKu SW, Yu TC, Chan KW. Decompression of multiple tension pneumatoceles in a child using computed tomography-guided percutaneous catheter placement. Canadian respiratory journal. 2011 Nov-Dec:18(6):e82-5 [PubMed PMID: 22187691]
Level 3 (low-level) evidenceZuhdi MK, Spear RM, Worthen HM, Peterson BM. Percutaneous catheter drainage of tension pneumatocele, secondarily infected pneumatocele, and lung abscess in children. Critical care medicine. 1996 Feb:24(2):330-3 [PubMed PMID: 8605809]
Level 2 (mid-level) evidenceKumar J, Mukhopadhyay K, Bhatia A. Successful percutaneous drainage of pneumatoceles in an extremely low-birthweight infant. BMJ case reports. 2018 Jan 26:2018():. pii: bcr-2017-222630. doi: 10.1136/bcr-2017-222630. Epub 2018 Jan 26 [PubMed PMID: 29374641]
Level 3 (low-level) evidenceKogutt MS, Lutrell CA, Puyau FA, Tieman EK. Decompression of pneumatocele in a neonate by percutaneous catheter placement. Pediatric radiology. 1999 Jun:29(6):488-9 [PubMed PMID: 10369915]
Level 3 (low-level) evidencePark TH, Kim JK. Nonsurgical management of an enlarging pneumatocele by fibrin sealant injection via pigtail catheter. Pediatric pulmonology. 2016 Feb:51(2):E5-7. doi: 10.1002/ppul.23311. Epub 2015 Sep 4 [PubMed PMID: 26340670]
Karmy-Jones R. Commentary on "Large traumatic pneumatocele treated using video-assisted thoracoscopic surgery". The journal of trauma and acute care surgery. 2019 Jun:86(6):1041. doi: 10.1097/TA.0000000000002274. Epub [PubMed PMID: 31124905]
Level 3 (low-level) evidenceGerdung CA, Ross BC, Dicken BJ, Bjornson CL. Pneumonectomy in a Child with Multilobar Pneumatocele Secondary to Necrotizing Pneumonia: Case Report and Review of the Literature. Case reports in pediatrics. 2019:2019():2464390. doi: 10.1155/2019/2464390. Epub 2019 Jul 17 [PubMed PMID: 31396429]
Level 3 (low-level) evidenceFagkrezos D, Giannila M, Maniatis P, Papailiou J, Triantopoulou C. Post-traumatic pulmonary pseudocyst with hemopneumothorax following blunt chest trauma: a case report. Journal of medical case reports. 2012 Oct 19:6():356. doi: 10.1186/1752-1947-6-356. Epub 2012 Oct 19 [PubMed PMID: 23083130]
Level 3 (low-level) evidenceFatimi SH, Jafferani A, Ashfaq A. Giant pulmonary bulla with mediastinal shift in a 12 1/2 year old girl. JPMA. The Journal of the Pakistan Medical Association. 2012 May:62(5):503-4 [PubMed PMID: 22755321]
Level 3 (low-level) evidenceChiarenza A, Esposto Ultimo L, Falsaperla D, Travali M, Foti PV, Torrisi SE, Schisano M, Mauro LA, Sambataro G, Basile A, Vancheri C, Palmucci S. Chest imaging using signs, symbols, and naturalistic images: a practical guide for radiologists and non-radiologists. Insights into imaging. 2019 Dec 4:10(1):114. doi: 10.1186/s13244-019-0789-4. Epub 2019 Dec 4 [PubMed PMID: 31802270]
Lee KC, Kang EY, Yong HS, Kim C, Lee KY, Hwang SH, Oh YW. A Stepwise Diagnostic Approach to Cystic Lung Diseases for Radiologists. Korean journal of radiology. 2019 Sep:20(9):1368-1380. doi: 10.3348/kjr.2019.0057. Epub [PubMed PMID: 31464115]
Haddad E. Infected pneumatocele. Journal of hospital medicine. 2006 Sep:1(5):311 [PubMed PMID: 17219517]
Level 3 (low-level) evidenceTortosa A, Boza R, Romeo A, Guillén-Solà A. [Pneumomediastinum caused by rupture of pneumatoceles]. Medicina clinica. 2016 Aug 5:147(3):137. doi: 10.1016/j.medcli.2015.11.036. Epub 2016 Jan 29 [PubMed PMID: 26832982]