Introduction
The recurrent artery of Heubner (RAH) was first described in 1872 by Johann Otto Leonhard Heubner (1843–1926), a German pediatrician recognized as the father of German pediatrics.[1] In 1909, H. F. Aitken, an artist at Massachusetts General Hospital, labeled it "Heubner’s artery." In 1920, Joseph Shellshear, an anatomist at St. Bartholomew’s Hospital in London, introduced the more precise and now widely accepted term "recurrent artery of Heubner," emphasizing its characteristic course along the A1 segment of the anterior cerebral artery (ACA) following its origin.[2] Older names for the artery include "anterior striate artery," "long telencephalic artery," "long centralis artery," and "distal medial striate artery."[3]
Structure and Function
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Structure and Function
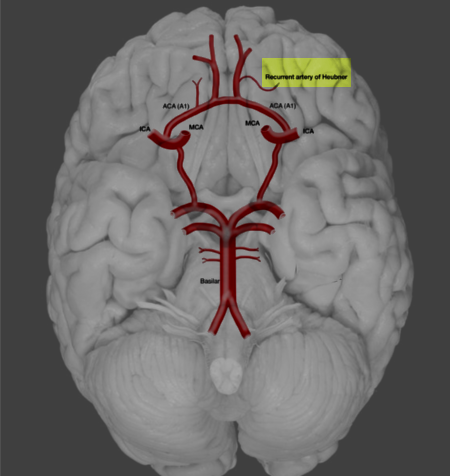
The RAH is typically the largest of the perforating medial lenticulostriate arteries arising from the ACA (see Image. Recurrent Artery of Heubner). This blood vessel may originate from the A1 or A2 segment or the junction of the ACA with the anterior communicating artery (AComA). The artery then characteristically turns posteriorly, courses over the gyrus rectus, and reaches the anterior perforating substance (APS). The RAH then passes inferiorly and laterally to the origin of the olfactory striae before terminating in the APS. One study reported a mean RAH diameter of 0.8 ± 0.04 mm and a mean length of 23.4 ± 1.1 mm.[4]
The RAH primarily supplies the following regions:
- Inferior aspect of the head of the caudate nucleus
- Outer segment of the globus pallidus
- Anterior and inferior portions of the anterior limb of the internal capsule
- Anterior hypothalamus
- Nucleus accumbens (connecting the caudate and putamen)
- Parts of the uncinate fasciculus (connecting the limbic system with the frontal lobe)
- Diagonal band of Broca (connecting the septal area to the amygdala)
- Basal nucleus of Meynert (located in the substantia innominata)
- Anterior 3rd of the putamen
- Olfactory bulbs and tracts
This extensive vascular distribution highlights the RAH's critical role in supplying key regions involved in motor control, cognition, and limbic functions.
Embryology
With the continued growth and development of the telencephalic vesicles, a vascular system emerges from the rostral division of the internal cerebral artery during embryogenesis. This vascular channel gives rise to all the lenticulostriate vessels, including the RAH, and later develops into the adult middle cerebral artery (MCA). This embryological origin accounts for the formation of a network of interconnections (rete communication) among branches of the RAH, the lenticulostriate arteries from the ACA, and the MCA.
According to Abbie’s phylogenetic concept, the RAH represents the remnant of an anastomotic network that once surrounded the paleo-olfactorium.[5] Although the ACA is recognized as the primary blood supply to the RAH, the artery may retain some prenatal connections with the MCA. The presence of anastomosing perforating branches between the MCA and RAH, along with the considerable variations observed in RAH vascularization, supports Abbie’s hypothesis.[6]
Physiologic Variants
In a study of 183 adult human brains (366 hemispheres), the most common origin of the RAH was the A2 segment (47.81%), followed by the ACA-AComA junction (43.4%).
However, another study found that the most frequent origin was the junction of A1 (extending from the ACA origin to the AComA) and A2 (extending from the AComA to the anterior border of the corpus callosum) in 76.2% of cases, followed by the A2 segment in 16.3%. Variations in RAH anatomy were observed, with the artery being absent or duplicated in approximately 6% of cases and triplicated in 0.1%.[7]
The RAH follows a predictable pattern. When it originates from the A1 segment, it arises from the posterior wall. When it originates from the A1/A2 junction or the A2 segment, it typically arises from the lateral wall of the ACA. Regardless of its origin, the RAH must be clearly identified and dissected before clipping to prevent iatrogenic injury, and reimplantation should be considered if it arises from the aneurysm wall.[8]
The recurrent course of the RAH in relation to the A1 segment as it moves toward the APS can be classified into 3 types:
- Type I or the superior course: 63%
- Type II or the anterior course: 34%
- Type III or the posterior course: 3%
The intracerebral course of the RAH is directed toward the head of the caudate nucleus. Within both its extracerebral and intracerebral courses, the RAH may form a network (rete) with the MCA by connecting with the middle group of lenticulostriate arteries or direct junctions.[9]
A persistent primitive olfactory artery (PPOA) results from the failure of the embryonic olfactory artery to regress, leading to an extension along the olfactory tract. This variant appears as an anteriorly oriented, elongated ACA or an AComA with a hairpin turn near the olfactory bulb and typically lacks an ipsilateral RAH. Persistent primitive olfactory arteries are associated with an increased risk of aneurysms and other ACA anomalies.[10]
Surgical Considerations
The primary concern during clipping of an AComA aneurysm is securing the aneurysm neck while fully preserving the RAH and other perforators. Intraoperatively, surgeons can confirm RAH patency by observing green indocyanine dye flow within the artery following permanent clipping.
Newer techniques, such as intraoperative angiography, endoscopy, and Doppler vascular flow assessment, are invaluable when visualizing areas not directly accessible under standard microscopy, as they minimize the risk of inadvertent clipping or kinking of the RAH. Vascular mapping provides both visual and quantitative analysis of flow within perforators, parent vessels, and branching arteries, reducing the risk of postoperative vasospasm. This method can also identify high-risk patients with decreased absorption intensity and delayed dye appearance, allowing for early initiation of antispasmodic interventions, including the "Triple-H" therapy, comprised of hypertension, hypervolemia, and hemodilution, as well as calcium channel blocker administration.[11]
The RAH is usually the first artery encountered during initial frontal lobe retraction, even before the A1 segment, as it courses anterior to A1 in 60% of cases.[12] Distinguishing the RAH from the orbitofrontal artery (OFA) can be challenging during surgery, but their courses provide a reliable distinction. The RAH typically follows the path of the A1 segment, whereas the OFA courses perpendicularly over the gyrus rectus and crosses the olfactory tract.[13] The proximal origin of the RAH, within a few millimeters of the A1-A2 junction, along with its recurrent course along A1, further differentiates it from the OFA.
Clinical Significance
The RAH may be involved in various pathologies, including the following:
- Large artery disease arising from greater than 50% stenosis of a major vessel, such as the carotid artery
- Small vessel disease secondary to hypertension, diabetes, or hypercholesterolemia
- Cardiac emboli due to atrial fibrillation, cardiac hypokinesis, mural thrombus formation, or dilated cardiomyopathies
- Trauma leading to vessel dissection and microthrombi formation
- Vasospasm following a ruptured aneurysm
- Inadvertent clipping during microsurgical clipping of AComA aneurysms
- Dissecting aneurysm of the RAH (eg, in patients with osteogenesis imperfecta) [14]
- Vascular malformations, such as cavernomas
Damage to the RAH diminishes the blood supply to its territory, leading to a spectrum of clinical manifestations, including the following:
- Motor deficits: Hemiparesis, often with faciobrachiocrural predominance
- Speech and swallowing difficulties: Dysarthria due to corticolingual and corticostriatocerebellar pathway involvement and hoarseness or dysphagia from supranuclear disruption of the nucleus ambiguus
- Behavioral changes: Abulia or hyperactivity due to disruption between the associative cortex and deep cortical regions
- Movement disorders: Choreoathetosis
- Cognitive and perceptual symptoms: Aphasia in left-sided involvement and left visual neglect in right-sided involvement
A study analyzing neurological abnormalities following vascular lesions in the caudate region reported the following manifestations in decreasing order of frequency:
- Abulia
- Frontal lobe abnormalities
- Speech deficits in left-sided lesions
- Neglect syndromes in right-sided lesions [15]
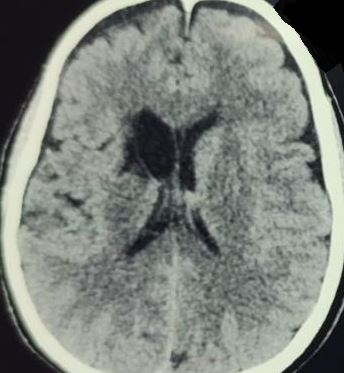
Neuroimaging can help diagnose RAH pathology. Computed tomography usually reveals hypodensity in cases of acute infarction and hyperdensity in cases of acute hemorrhage in the caudate region (see Image. Recurrent Artery of Heubner Stroke on Computed Tomography). Diffusion-weighted imaging and apparent diffusion coefficient sequences enable ultra-early detection of infarction via magnetic resonance imaging, as these sequences are particularly sensitive to ischemic changes. Endovascular approaches offer combined diagnostic and therapeutic benefits, allowing for the detection of vasospasm in affected vessels and providing treatment through stent placement or vasodilator administration.
Other Issues
The differential diagnosis for vascular involvement of the head of the caudate nucleus includes damage to the following arteries:
- RAH: Supplies the inferior part of the head of the caudate nucleus and the anterior limb of the internal capsule
- Anterior lenticulostriate arteries: Originate from the A1 segment of the ACA and supply the anterior portion of the head of the caudate nucleus
- Lateral lenticulostriate arteries: Arise from the MCA and supply a major portion of the head of the caudate nucleus, the anterior limb of the internal capsule, and the putamen
Media
(Click Image to Enlarge)
(Click Image to Enlarge)
References
Haroun RI, Rigamonti D, Tamargo RJ. Recurrent artery of Heubner: Otto Heubner's description of the artery and his influence on pediatrics in Germany. Journal of neurosurgery. 2000 Dec:93(6):1084-8 [PubMed PMID: 11117858]
El Falougy H, Selmeciova P, Kubikova E, Haviarová Z. The variable origin of the recurrent artery of Heubner: an anatomical and morphometric study. BioMed research international. 2013:2013():873434. doi: 10.1155/2013/873434. Epub 2013 Jul 9 [PubMed PMID: 23936853]
Zunon-Kipré Y, Peltier J, Haïdara A, Havet E, Kakou M, Le Gars D. Microsurgical anatomy of distal medial striate artery (recurrent artery of Heubner). Surgical and radiologic anatomy : SRA. 2012 Jan:34(1):15-20. doi: 10.1007/s00276-011-0888-5. Epub 2011 Nov 25 [PubMed PMID: 22116404]
Lee SH, Lee CH, Park IS, Han JW. Bilateral Infarction of the Recurrent Arteries of Heubner Following Clipping of an Anterior Communicating Artery Aneurysm. Journal of cerebrovascular and endovascular neurosurgery. 2018 Mar:20(1):28-34. doi: 10.7461/jcen.2018.20.1.28. Epub 2018 Mar 31 [PubMed PMID: 30370237]
Abbie AA. The Morphology of the Fore-Brain Arteries, with Especial Reference to the Evolution of the Basal Ganglia. Journal of anatomy. 1934 Jul:68(Pt 4):433-70 [PubMed PMID: 17104495]
Gomes F, Dujovny M, Umansky F, Ausman JI, Diaz FG, Ray WJ, Mirchandani HG. Microsurgical anatomy of the recurrent artery of Heubner. Journal of neurosurgery. 1984 Jan:60(1):130-9 [PubMed PMID: 6689705]
Matsuda W, Sonomura T, Honma S, Ohno S, Goto T, Hirai S, Itoh M, Honda Y, Fujieda H, Udagawa J, Ueda S. Anatomical variations of the recurrent artery of Heubner: number, origin, and course. Anatomical science international. 2018 Jun:93(3):317-322. doi: 10.1007/s12565-017-0415-9. Epub 2017 Sep 25 [PubMed PMID: 28948536]
Kashtiara A, Beldé S, Menovsky T. Anatomical Variations and Anomalies of the Anterior Communicating Artery Complex. World neurosurgery. 2024 Mar:183():e218-e227. doi: 10.1016/j.wneu.2023.12.060. Epub 2023 Dec 15 [PubMed PMID: 38104930]
Maga P, Tomaszewski KA, Pasternak A, Zawiliński J, Tomaszewska R, Gregorczyk-Maga I, Skrzat J. Extra- and intracerebral course of the recurrent artery of Heubner. Folia morphologica. 2013 May:72(2):94-9 [PubMed PMID: 23740494]
Wolman DN, Moraff AM, Heit JJ. Anatomy of the Intracranial Arteries: The Anterior Intracranial and Vertebrobasilar Circulations. Neuroimaging clinics of North America. 2022 Aug:32(3):617-636. doi: 10.1016/j.nic.2022.04.007. Epub [PubMed PMID: 35843666]
Munakomi S, Poudel D. A pilot study on assessing the role of intra-operative Flow 800 vascular map model in predicting onset of vasospasm following micro vascular clipping of ruptured intracranial aneurysms. F1000Research. 2018:7():1188. doi: 10.12688/f1000research.15627.1. Epub 2018 Aug 3 [PubMed PMID: 30271586]
Level 3 (low-level) evidencePerlmutter D, Rhoton AL Jr. Microsurgical anatomy of the anterior cerebral-anterior communicating-recurrent artery complex. Journal of neurosurgery. 1976 Sep:45(3):259-72 [PubMed PMID: 948013]
Perlmutter D, Rhoton AL Jr. Microsurgical anatomy of the distal anterior cerebral artery. Journal of neurosurgery. 1978 Aug:49(2):204-28 [PubMed PMID: 671075]
Mansfield K, Rahme R. Dissecting Aneurysm of the Recurrent Artery of Heubner in a Patient With Osteogenesis Imperfecta. The Canadian journal of neurological sciences. Le journal canadien des sciences neurologiques. 2015 Nov:42(6):461-5. doi: 10.1017/cjn.2015.295. Epub [PubMed PMID: 26551090]
Kumral E, Evyapan D, Balkir K. Acute caudate vascular lesions. Stroke. 1999 Jan:30(1):100-8 [PubMed PMID: 9880396]