Definition/Introduction
In 1825, the "first workable, full-time SCUBA" was invented by an Englishman, William James. His design incorporated a cylindrical belt around the diver's trunk that served as an air reservoir at a pressure of 450 PSI. Air delivery to the diver was by turning a valve on and off as needed. While James is credited with pioneering SCUBA, the act of diving was introduced to the world by Jacques-Yves Cousteau. In 1942, Cousteau and Emile Gagnan developed the demand valve system that became a precursor to modern regulators. The work of Paul Bert and John Scott Haldane, from France and Scotland, respectively, further elucidated the effects of water pressure on the body and defined safe limits for compressed air diving. Cousteau became famous by following these rules, which allowed him to survive dives that killed many others before him.
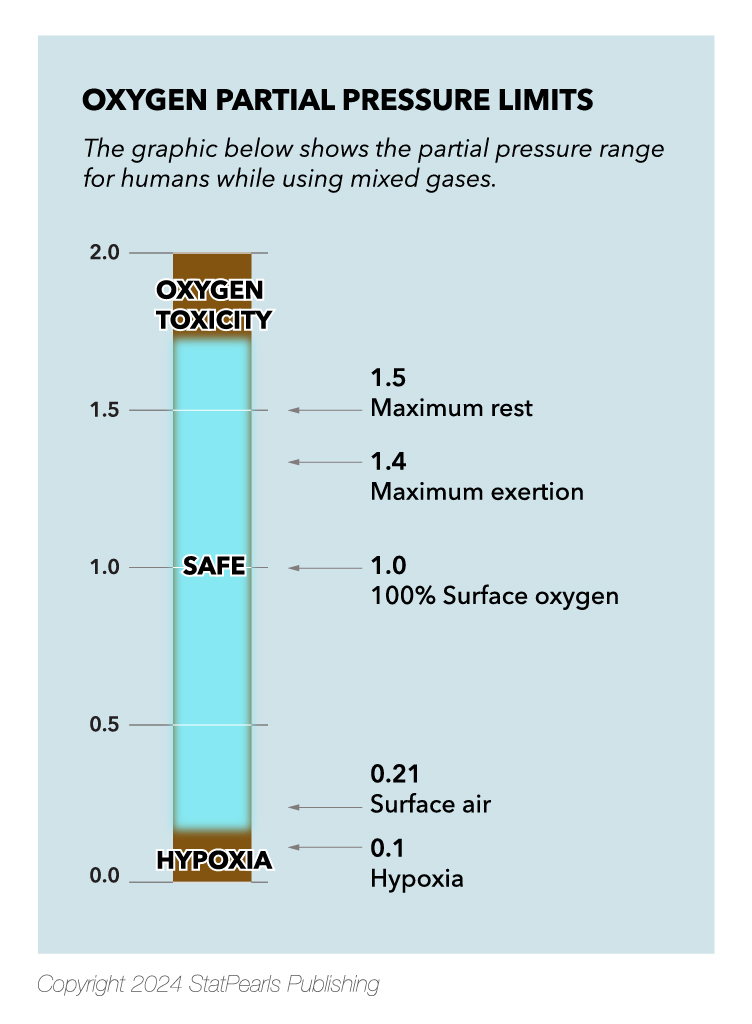
Today, there are 2 principal types of SCUBA: open and closed circuits. Open-circuit vents all expired gas into the water and is the mode predominately used during recreational diving. Closed-circuit systems involve the exhaled gas being rebreathed after carbon dioxide is absorbed and oxygen added. These systems were widely used before open circuits became available, particularly by military divers who wished to avoid showing any air bubbles. This article will review the medical concerns associated with compressed gas diving, including the risks and benefits of air, enhanced oxygen (nitrox) and trimix diving, and the physics and physiology of SCUBA (see Image. Oxygen Partial Pressure Limits).[1][2]
Issues of Concern
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Issues of Concern
Open- Versus Closed-Circuit Diving
Open
Open circuit diving can include compressed air, nitrox, and trimix. As exhaled gas leaves the circuit, the diver is safe if they have sufficient gas during inhalation. The last resort for any problem in open circuit diving is exhaling and ascending to the surface. As a result, open circuit diving accidents usually involve a heart attack, arterial gas embolism from rapid ascent or severe decompression sickness (DCS), insufficient gas, or environmental factors (eg, a diver who is lost or trapped).[3]
Closed
Closed-circuit diving is classified as technical diving, requiring significantly more training. Most sports diving systems are semi-closed circuits, but the military relies on completely closed circuits. As little or no bubbles are released, underwater stealth allows the diver to move close to marine life or enemy vessels without detection. Semi-closed circuit sports diving typically uses nitrox (enhanced oxygenated air) to add gas when computer-controlled oxygen sensors detect low partial pressures. Exhaled air is scrubbed of carbon dioxide by various filter substances depending on the system; barium hydroxide and sodium peroxide are examples.[1]
In addition to the open-circuit dangers mentioned previously, closed-circuit diving is also associated with oxygen seizures, hypoxia, and equipment-induced DCS. Failure of the rebreather can cause carbon dioxide accumulation and injury or death. Oxygen toxicity or hypoxia at depth may occur due to a sensor malfunction, an error in gas selection, or the diver's physiology.
Furthermore, these divers often operate at extreme depths. A safe ascent requires safety stops to off-gas excess nitrogen to avoid DCS, making an immediate ascent to the surface potentially fatal.[1] Off-gassing allows the inert gases (eg, nitrogen, helium) that have dissolved into a diver's body tissues to reenter the bloodstream and be exhaled gradually.
Diving with compressed gas forms nitrogen bubbles in the bloodstream, regardless of circuit type. The body's ability to clear the excess nitrogen depends on many factors and determines whether the diver is injured as they off the gas.
Gas Types Used During SCUBA
Air
Air is the safest and most commonly used gas during sports diving. Above 100 feet of depth, there are virtually no risks to divers using compressed air. However, at depths below 100 feet, the risk of nitrogen narcosis and DCS is increased. Nitrogen narcosis occurs due to high nitrogen partial pressure in the blood, causing impaired vision, drunkenness, disorientation, and an anesthetic effect. At 200 feet, nitrogen narcosis is so severe that the majority of divers are disabled. Fortunately, nitrogen is cleared rapidly during the ascent to shallower depths, and divers can increase their tolerance to nitrogen narcosis by repeated exposure. Oxygen toxicity does not occur until a diver has descended below 218 feet.
Nitrox
Nitrox or enhanced air oxygen (EAN) is a mixed gas with an oxygen percentage that exceeds the 21% present in normal air. Nitrox is commonly mixed with 32% or 36% oxygen, thus lowering the nitrogen content. By reducing the amount of nitrogen, nitrox minimizes the risk of nitrogen narcosis and decompression sickness at depths of 110 feet or less. This mixture is easier to breathe at depth and prolongs the diver's time at shallower depths by reducing nitrogen loading. However, the risk of oxygen toxicity is significant at depths below 100 feet. Oxygen toxicity occurs when excess partial pressures of oxygen enter the tissues. Excessive oxygen causes oxidative damage to the central nervous system, lungs, and eyes, leading to alveolar collapse in the lungs, retinal detachment, and seizures. Initial symptoms typically include cough due to tracheal irritation and myopia.Trimix
Trimix is composed of an oxygen, helium, and nitrogen combination that may be altered based on depth requirements. A standard mix is trimix 20/30, containing 20% oxygen, 30% helium, and 50% nitrogen. Trimix reduces the risk of nitrogen narcosis and oxygen toxicity during dives below 200 feet. The helium reduces breathing resistance, which is essential for the high volumes of gas required at greater depths. Helium also enters and leaves the tissue quickly, which reduces decompression periods in saturation diving. This mixture also reduces hyperbaric arthralgia during descent.
However, helium conducts body heat 6 times faster than normal air, increasing the risk of hypothermia. Also, since helium is rapidly absorbed, it requires deeper decompression stops compared to other mixed gases during ascent. Hypoxia can occur when using trimix at shallower depths. The most significant disadvantage associated with trimix is the cost of training, mixing, and accidents.SCUBA Physics and Physiology
The concept of partial pressure must be firmly grasped to understand the physiological ramifications of breathing various gas mixtures under pressure. The partial pressure of a particular gas constituent in a gas mixture represents the portion of the total pressure exerted by the specific constituent. The sum of all the partial pressures of each mixture component equals the total pressure of the mixture.
To maintain oxygenation at depth, higher volumes of air are necessary. Boyle's law states that the absolute pressure exerted by a given mass of an ideal gas is inversely proportional to the volume it occupies if the temperature and amount of gas remain unchanged within a closed system. For every 33 feet (10 m) of depth in seawater, the ambient pressure increases by an additional 14.7 PSI (1 atm). At a depth of 99 feet (30 m), the ambient pressure is 4 atm, 1 atm caused by the Earth's atmosphere, plus 3 atm for every 33 feet (10 m) of depth. Therefore, a diver must inhale 4 times the air at 100 feet than at the surface to deliver the same amount of oxygen to the tissues.[4]
Henry's law states that "the amount of any given gas that dissolves in a liquid at a given temperature is a function of the partial pressure of that gas in contact with the liquid." What this means for divers is that gas molecules dissolve into the blood in proportion to the partial pressure of that gas in the lungs. As warm-blooded creatures, our core body temperature remains relatively constant; this leaves a specific range in which the partial pressure of oxygen is safe and necessary for consciousness (see Image. Oxygen Partial Pressure Limits).
Similarly, excess dissolved nitrogen in the blood can cause bubble formation, nitrogen narcosis, and decompression sickness during a rapid ascent. The overall volume of compressed gas expands on the ascent, whether the gas is in the lungs, the blood, or the middle ear and sinuses, as described in Boyle's Law.[5]
If a diver holds their breath during the ascent, this causes barotrauma from hyperinflation of the lungs and can result in gas embolism as gas is forced into the pulmonary vasculature. In the sinuses or middle ear, barotrauma occurs when the gas cannot equalize to the external pressure; the overpressure can produce a "squeeze" or epistaxis to compensate. In the blood, the dissolved gas saturates out and forms bubbles. These gas bubbles can embolize, causing myocardial infarctions or cerebral vascular accidents when there is a right to left shunt in the heart (eg, patent foramen ovale or atrial septal defect).
Clinical Significance
Scuba diving has provided humankind with the freedom and opportunity to explore the underwater world. Understanding the basic physiology and application is essential to clinical practice, as the sport has inherent risks that require clinician familiarity.
Nursing, Allied Health, and Interprofessional Team Interventions
The safety and security of scuba divers require an understanding of the material by divers and clinical staff. This will allow providers to discuss scuba diving physiology and the use of various gases and their effects, when to prohibit the activity based on other medical conditions, and when to advise on activity limits.
Nursing, Allied Health, and Interprofessional Team Monitoring
Clinical staff should be aware of the information presented in this article. Providers performing regular scuba diving screening exams should also consult the Underwater and Hyperbaric Medical Society and the Diver's Alert Network for additional information.
Media
References
Dugrenot E, Balestra C, Gouin E, L'Her E, Guerrero F. Physiological effects of mixed-gas deep sea dives using a closed-circuit rebreather: a field pilot study. European journal of applied physiology. 2021 Dec:121(12):3323-3331. doi: 10.1007/s00421-021-04798-y. Epub 2021 Aug 25 [PubMed PMID: 34435274]
Level 3 (low-level) evidenceGemba KL, Nosal EM, Reed TR. Partial dereverberation used to characterize open circuit scuba diver signatures. The Journal of the Acoustical Society of America. 2014 Aug:136(2):623-33. doi: 10.1121/1.4884879. Epub [PubMed PMID: 25096097]
Denoble PJ, Caruso JL, Dear Gde L, Pieper CF, Vann RD. Common causes of open-circuit recreational diving fatalities. Undersea & hyperbaric medicine : journal of the Undersea and Hyperbaric Medical Society, Inc. 2008 Nov-Dec:35(6):393-406 [PubMed PMID: 19175195]
Shopov NG. Study of the changes in respiratory function in self-contained underwater breathing apparatus divers. International maritime health. 2019:70(1):61-64. doi: 10.5603/IMH.2019.0009. Epub [PubMed PMID: 30931519]
Blake DF, Crowe M, Mitchell SJ, Aitken P, Pollock NW. Vibration and bubbles: a systematic review of the effects of helicopter retrieval on injured divers. Diving and hyperbaric medicine. 2018 Dec 24:48(4):235-240. doi: 10.28920/dhm48.4.241-251. Epub [PubMed PMID: 30517957]
Level 1 (high-level) evidence