Introduction
Full-thickness skin grafts (FTSGs) are a critical reconstructive technique in plastic and reconstructive surgery, offering superior aesthetic and functional outcomes compared to split-thickness skin grafts (STSGs) (see Image. Full-Thickness Skin Graft Procedure). A skin graft is a cutaneous free tissue transfer harvested from a donor site and transplanted to a recipient site when healing by primary closure, secondary intention, or flap repair is not feasible.[1] While the ideal graft would provide all skin layers and appendages—including sebaceous and sweat glands, nerves, and pigmentation—current options remain limited to autologous, allogenic, or xenographic tissue.[2] Additionally, engineered acellular and cellular skin products are often used independently or with grafting.
FTSGs consist of the complete epidermis and dermis, offering advantages such as better cosmesis, less contraction, and greater resistance to trauma compared to STSGs, which lack skin appendages and produce a suboptimal functional and aesthetic outcome.[3] FTSGs are particularly useful for reconstructing small functional units like fingers and facial defects, including the nasal tip, dorsum, ala, eyelids, and ears, where their superior color match, reduced contraction, and improved functional integration provide optimal results.[4] FTSGs can be applied to deep defects over underlying structures with intact sheaths, such as perichondrium, perineurium, periosteum, peritenon, fascia, muscle, and granulation.[5] However, their success depends on a well-vascularized wound bed and an appropriate donor site, which should closely match the recipient area in color, thickness, and texture. While FTSGs provide excellent long-term results, they carry a greater risk of graft failure, require primary donor site closure, and are limited by tissue availability.[6][7] This course explores the indications, surgical principles, donor site selection, complications, and clinical outcomes associated with FTSGs.
Anatomy and Physiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Anatomy and Physiology
FTSGs include the epidermis and the entire dermis, preserving skin appendages such as sebaceous glands, sweat glands, and hair follicles. The epidermis, composed primarily of keratinocytes, provides a protective barrier, while the dermis consists of fibroblasts, collagen, elastin, and an extensive vascular network that supports wound healing and graft survival. Unlike STSGs, which lack deeper dermal structures, FTSGs retain a rich dermal matrix, leading to better sensory recovery, durability, and cosmetic outcomes. The ideal donor site for FTSGs should closely match the recipient area's color, thickness, and texture, ensuring optimal functional and aesthetic results.
Graft survival depends on the ingrowth of capillaries from the recipient site and follows 3 key phases: plasmatic imbibition, inosculation, and neovascularization. In the first 1 to 2 days, the graft absorbs transudate from the wound bed in a process called plasmatic imbibition, which provides initial hydration and nutrients. This is followed by inosculation, where capillaries from the recipient site establish connections with those in the graft. By days 4 to 7, neovascularization occurs, leading to full vascular integration, while lymphatic channels are organized within a week. Tissue remodeling and sensory reinnervation begin within 14 to 28 days and continue for months.[8] A vascular network from the wound periphery can supply up to 5 mm of the graft. In cases where initial perfusion is inadequate, allowing secondary healing with granulation tissue formation or using a vascularized tissue flap can enhance graft success.[8][9]
Indications
FTSGs are used to cover smaller soft tissue defects that will not heal by primary closure, secondary intention, or flap repair, including areas of cosmetic and practical importance such as the nasal tip, dorsum, ala, and sidewall, the eyelids, and the ears and small functional units such as the digits.[4]
Contraindications
FTSG success relies on a well-vascularized recipient site, making poorly perfused wounds unsuitable candidates. Irradiated wounds and those lacking underlying structures often have inadequate capillary networks for graft survival. Contraindications include infection, colonization, malignancy, and uncontrolled bleeding. Additionally, smoking impairs tissue oxygenation and should be discontinued before graft placement to optimize outcomes.[10]
Equipment
The following equipment is required when performing an FTSG:
Preoperative
- Local anesthetic (typically lidocaine 0.5% with epinephrine 1:200,000 and buffered with sodium bicarbonate)
- 3 cc syringe(s)
- 30 gauge needle(s)
- Antiseptic scrub
- Surgical marker
- Sterile foil or other material to template the defect
Intraoperative
- Sterile drape
- Scalpel with #15 blade
- Toothed forceps
- Serrated scissors (for trimming away fat from graft)
- Suture scissors
- Needle holders
- Normal saline
- Sterile gauze
- Absorbable suture (for subcutaneous/deep stitches)
- Cutaneous suture (nonabsorbable or absorbable)
- Electrosurgical device for hemostasis
Postoperative
Personnel
Successful FTSG procedures require a multidisciplinary team. The surgeon performs the grafting, assisted by a surgical assistant and scrub nurse who manage instruments and graft handling. Anesthesiologists/nurse anesthetists provide anesthesia and monitor patient stability. Postoperative nurses oversee wound care, while physical/occupational therapists aid in functional recovery. Pharmacists ensure proper medication management and wound care specialists assist with dressing changes and monitoring. Pathologists may assess margins in oncologic cases, and social workers/case managers coordinate follow-up care. This collaborative approach optimizes graft survival, functional outcomes, and patient safety.
Preparation
Proper preparation is essential for FTSG's success. The recipient site must be clean, well-vascularized, and free from infection or necrotic tissue. If necessary, debridement or staged wound bed optimization with negative pressure therapy or biologic dressings may be performed. The donor site should be selected from a well-perfused area with optimal cosmetic match and comparable appendages, such as the supraclavicular, postauricular, or groin regions. A pliable material, such as foil or a surgical drape, can be pressed into the wound to create a template, which is then traced at the donor site with a surgical marker.
Both donor and recipient sites are anesthetized and prepped using a sterile technique. The recipient site is scrubbed and debrided as needed to ensure healthy bleeding tissue and meticulous hemostasis to optimize graft adherence.[12] Preoperative considerations include patient optimization, such as controlling comorbidities, providing adequate nutrition, and discontinuing vasoconstrictive medications. Proper anesthesia planning, aseptic technique, and surgical markings further enhance graft survival and functional outcomes.
Technique or Treatment
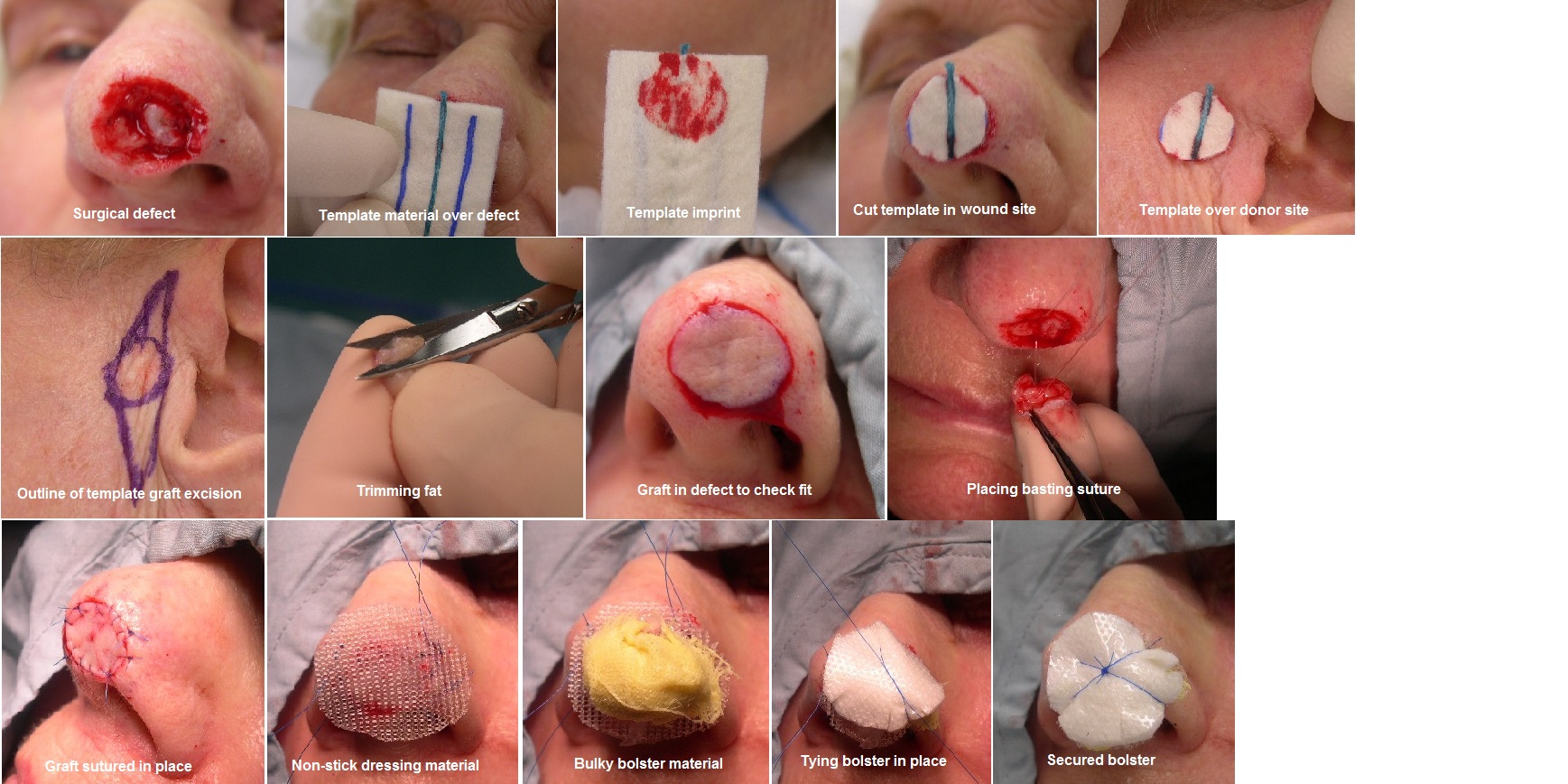
The technique of FTSG involves the precise harvesting, preparation, and transplantation of a graft containing the entire epidermis and dermis to a well-vascularized recipient site (see Image. Full-Thickness Skin Graft Procedure). This method is preferred for its superior cosmetic and functional outcomes, offering better color match, less contraction, and preserving skin appendages. Proper execution of FTSG requires meticulous surgical technique, including careful donor site selection, atraumatic graft handling, secure fixation, and postoperative care to ensure graft viability. Mastery of these technical steps is essential for optimizing graft survival and minimizing complications such as graft failure, infection, and contracture.
The harvesting process begins with selecting an optimal donor site that provides a good cosmetic match. A template of the recipient wound is created using pliable material and transferred to the donor site with a surgical marker. The graft is then incised sharply along the marked outline, and the skin is carefully elevated using a skin hook while being dissected free deep to the dermal plane. Excess adipose tissue is sharply trimmed from the underside to enhance graft take. The surrounding donor skin may be undermined to facilitate primary closure. The donor site is then sutured in layers, with 4-0 vicryl for the deep layer and 5-0 nonabsorbable sutures for skin approximation.
The wound is scrubbed and debrided to healthy bleeding tissue at the recipient site, and hemostasis is ensured. The graft is placed dermal side down, carefully positioned to cover the wound bed, and secured using interrupted 4-0 absorbable sutures at the corners followed by a running suture around the periphery. A sterile, moist gel may be applied to promote healing. A bolster dressing is then placed over the graft and sutured to the surrounding skin using nonabsorbable sutures to ensure immobilization and graft adherence. The bolster dressing remains in place for 1 week, during which the graft must maintain direct contact with the wound bed and remain immobile to optimize inosculation and neovascularization.[13][14][15]
Complications
FTSGs generally yield favorable outcomes but are associated with potential complications that can impact graft survival and aesthetic results. The most significant risk is graft failure, which may result from hematoma, infection, or disruption of graft contact with the wound bed. Poor microcirculation, particularly in patients with diabetes or those who smoke, can also contribute to graft failure. In particular, smoking more than 1 pack per day has been shown to triple the risk of flap necrosis. Hematomas and seromas can accumulate beneath the graft, impairing graft take. The graft may be left as "scaffolding" for epithelialization if necrosis occurs.
Infection is another common complication that can affect donor and recipient healing. Infection may lead to wound breakdown, graft rejection, or systemic complications if left untreated. Graft contraction is less of an issue with FTSGs compared to STSGs but can still occur, particularly in areas under tension, affecting functional range or cosmetic outcomes. Hypertrophic scarring or keloid formation can occur, especially when there is tension at the graft site, negatively impacting aesthetics. In some cases, the grafted area may remain insensate due to a lack of nerve regeneration.
Donor site complications such as dehiscence can occur if not properly closed, requiring additional wound care or secondary closure. Moreover, there is a risk of malignancy developing at donor sites, such as basal cell carcinoma.[16] Results from studies comparing FTSGs with partial-thickness grafts in pediatric hand burns found that FTSGs resulted in less contraction and greater function but increased scar formation, discoloration, and hair growth in the grafted areas.[17] Understanding these complications allows clinicians to tailor their approach to minimize risks and improve patient outcomes.
Clinical Significance
FTSGs provide skin coverage for wounds in which healing by secondary intention, primary closure, or flap repair is not feasible. No synthetic substitute or engineered product provides all components of full-thickness skin, but this is an active area of research. Research focuses on ways to reduce donor site morbidity and provide broader coverage. One such product utilizes pluripotent cells to seed epithelization.[3][18][19][20] Researchers generated skin grafts using pluripotent stem cell-derived epidermis with intact appendages using p63 knockout embryo dermis.[18][21][22]
Cultured epidermal autografts are used to harvest keratinocytes and develop skin layers from cells. Still, this technology lacks appendages or pigmentation and is best used in extensive wounds with limited options for donor sites. Other innovations include a cell therapy that uses an autologous epidermal donor site and includes melanocytes but no appendages, also used to cover large areas. These products can be used in conjunction with skin grafts.[3][23] A skin substitute was used with staged full-thickness grafting to yield improved healing and less contracture in a toddler hand burn.[24][25] Additional research is focused on utilizing xenographic products such as fish grafts.[26]
Enhancing Healthcare Team Outcomes
Effective care for patients undergoing FTSGs relies on coordinated efforts and strong interprofessional communication across various healthcare providers. Surgeons and advanced clinicians lead the procedure by selecting appropriate donor sites, harvesting the graft, and ensuring meticulous application to the recipient site. Nurses are crucial in perioperative care, providing wound management, monitoring for complications such as infection or graft failure, and educating patients on postoperative care and signs of complications. Pharmacists ensure the proper administration of antibiotics and analgesics, preventing infection and managing pain, which is critical for patient comfort and optimal healing.
Effective interprofessional communication is key to ensuring that all team members are aligned with the treatment goals, particularly in managing high-risk patients (eg, those with diabetes or smoking history) who are at higher risk of graft failure. Care coordination is essential in monitoring the patient's recovery, with the team collaboratively adjusting treatment plans based on patient progress. Regular check-ins and updates among the team members help address issues like graft viability, infection, or donor site healing. Fostering a patient-centered approach through clear communication and coordination among healthcare providers enhances patient safety, improves outcomes, and contributes to successful graft survival and cosmetic results.
Nursing, Allied Health, and Interprofessional Team Interventions
The ancillary staff must be familiar with stages of graft healing, bandage and wound assessment, and facets of general clinical care that are important to successful skin grafting. Any concerns should be relayed immediately to the surgical staff.
Media
(Click Image to Enlarge)
References
Adams DC, Ramsey ML. Grafts in dermatologic surgery: review and update on full- and split-thickness skin grafts, free cartilage grafts, and composite grafts. Dermatologic surgery : official publication for American Society for Dermatologic Surgery [et al.]. 2005 Aug:31(8 Pt 2):1055-67 [PubMed PMID: 16042930]
Ratner D. Skin grafting. Seminars in cutaneous medicine and surgery. 2003 Dec:22(4):295-305 [PubMed PMID: 14740962]
Palmieri TL. Emerging Therapies for Full-Thickness Skin Regeneration. Journal of burn care & research : official publication of the American Burn Association. 2023 Jan 2:44(Suppl_1):S65-S67. doi: 10.1093/jbcr/irac102. Epub [PubMed PMID: 36567471]
Bogdanov SB, Gilevich IV, Melkonyan KI, Sotnichenko AS, Alekseenko SN, Porhanov VA. Total full-thickness skin grafting for treating patients with extensive facial burn injury: A 10-year experience. Burns : journal of the International Society for Burn Injuries. 2021 Sep:47(6):1389-1398. doi: 10.1016/j.burns.2020.12.003. Epub 2020 Dec 10 [PubMed PMID: 33358397]
Trufant JW, Marzolf S, Leach BC, Cook J. The utility of full-thickness skin grafts (FTSGs) for auricular reconstruction. Journal of the American Academy of Dermatology. 2016 Jul:75(1):169-76. doi: 10.1016/j.jaad.2016.01.028. Epub 2016 Mar 2 [PubMed PMID: 26944598]
Moors JJE, Xu Z, Xie K, Rashad A, Egger J, Röhrig R, Hölzle F, Puladi B. Full-thickness skin graft versus split-thickness skin graft for radial forearm free flap donor site closure: protocol for a systematic review and meta-analysis. Systematic reviews. 2024 Feb 26:13(1):74. doi: 10.1186/s13643-024-02471-x. Epub 2024 Feb 26 [PubMed PMID: 38409059]
Level 1 (high-level) evidenceS AP, R S, C B. Comparative Retrospective Analysis of Cross-Finger Flap Outcomes: A Study on Split-Thickness Skin Graft vs. Full-Thickness Skin Graft in Donor Fingers. Cureus. 2024 Oct:16(10):e72013. doi: 10.7759/cureus.72013. Epub 2024 Oct 21 [PubMed PMID: 39574994]
Level 2 (mid-level) evidenceFader DJ, Wang TS, Johnson TM. Nasal reconstruction utilizing a muscle hinge flap with overlying full-thickness skin graft. Journal of the American Academy of Dermatology. 2000 Nov:43(5 Pt 1):837-40 [PubMed PMID: 11050590]
Hanna TC, McKenzie WS, Holmes JD. Full-thickness skin graft from the neck for coverage of the radial forearm free flap donor site. Journal of oral and maxillofacial surgery : official journal of the American Association of Oral and Maxillofacial Surgeons. 2014 Oct:72(10):2054-9. doi: 10.1016/j.joms.2014.05.015. Epub 2014 May 27 [PubMed PMID: 25053568]
Davis M, Baird D, Hill D, Layher H, Akin R. Management of full-thickness skin grafts. Proceedings (Baylor University. Medical Center). 2021:34(6):683-686. doi: 10.1080/08998280.2021.1953867. Epub 2021 Jul 29 [PubMed PMID: 34732986]
Dev Measuria H, Haj-Basheer M. A quick and easy method of templating full-thickness skin grafts. JPRAS open. 2020 Sep:25():1-3. doi: 10.1016/j.jpra.2019.12.002. Epub 2020 Feb 26 [PubMed PMID: 32462073]
Zhao CX, Scher M, Hanks JE, McLean SA. Full-thickness versus split-thickness skin graft reconstruction of scalp defects with and without calvarium exposure. Journal of plastic, reconstructive & aesthetic surgery : JPRAS. 2024 Oct:97():275-281. doi: 10.1016/j.bjps.2023.10.053. Epub 2023 Oct 14 [PubMed PMID: 39178692]
Cabeza-Martínez R, Leis V, Campos M, de la Cueva P, Suárez R, Lázaro P. Burow's grafts in the facial region. Journal of the European Academy of Dermatology and Venereology : JEADV. 2006 Nov:20(10):1266-70 [PubMed PMID: 17062044]
Level 3 (low-level) evidenceLee KK, Mehrany K, Swanson NA. Fusiform elliptical Burow's graft: a simple and practical esthetic approach for nasal tip reconstruction. Dermatologic surgery : official publication for American Society for Dermatologic Surgery [et al.]. 2006 Jan:32(1):91-5 [PubMed PMID: 16393606]
Steele L, Brown A, Xie F. Full-thickness Skin Graft Fixation Techniques: A Review of the Literature. Journal of cutaneous and aesthetic surgery. 2020 Jul-Sep:13(3):191-196. doi: 10.4103/JCAS.JCAS_184_19. Epub [PubMed PMID: 33208994]
Shen AY, Seth I, Marcaccini G, Rozen WM, Ross RJ. Basal Cell Carcinoma Arising in a Previous Full-Thickness Graft Donor Site: A Case Report and Comprehensive Literature Review. Journal of clinical medicine. 2025 Jan 17:14(2):. doi: 10.3390/jcm14020591. Epub 2025 Jan 17 [PubMed PMID: 39860596]
Level 3 (low-level) evidenceAlsaif A, Karam M, Hayre A, Abul A, Aldubaikhi A, Kahlar N. Full thickness skin graft versus split thickness skin graft in paediatric patients with hand burns: Systematic review and meta-analysis. Burns : journal of the International Society for Burn Injuries. 2023 Aug:49(5):1017-1027. doi: 10.1016/j.burns.2022.09.010. Epub 2022 Sep 29 [PubMed PMID: 36280545]
Level 1 (high-level) evidenceFuchs C, Pham L, Henderson J, Stalnaker KJ, Anderson RR, Tam J. Multi-faceted enhancement of full-thickness skin wound healing by treatment with autologous micro skin tissue columns. Scientific reports. 2021 Jan 18:11(1):1688. doi: 10.1038/s41598-021-81179-7. Epub 2021 Jan 18 [PubMed PMID: 33462350]
Blome-Eberwein SA. Emerging Technologies. Clinics in plastic surgery. 2024 Jul:51(3):355-363. doi: 10.1016/j.cps.2024.02.002. Epub 2024 Mar 19 [PubMed PMID: 38789145]
Zhong C, He S, Huang Y, Yan J, Wang J, Liu W, Fang J, Ren F. Scaffold-based non-viral CRISPR delivery platform for efficient and prolonged gene activation to accelerate tissue regeneration. Acta biomaterialia. 2024 Jan 1:173():283-297. doi: 10.1016/j.actbio.2023.10.029. Epub 2023 Oct 31 [PubMed PMID: 37913843]
Li F, Zhang JY, Guo YL, Zhang W. Absorbable Meek skin graft material transplantation: A preliminary experimental study. Burns : journal of the International Society for Burn Injuries. 2025 Apr:51(3):107387. doi: 10.1016/j.burns.2025.107387. Epub 2025 Jan 9 [PubMed PMID: 39842062]
Zhao X, Li X, Wang Y, Guo Y, Huang Y, Lv D, Lei M, Yu S, Luo G, Zhan R. Stability and biosafety of human epidermal stem cell for wound repair: preclinical evaluation. Stem cell research & therapy. 2023 Jan 5:14(1):4. doi: 10.1186/s13287-022-03202-6. Epub 2023 Jan 5 [PubMed PMID: 36600269]
Song YT, Liu PC, Zhou XL, Chen YM, Wu W, Zhang JY, Li-Ling J, Xie HQ. Extracellular matrix-based biomaterials in burn wound repair: A promising therapeutic strategy. International journal of biological macromolecules. 2024 Dec:283(Pt 3):137633. doi: 10.1016/j.ijbiomac.2024.137633. Epub 2024 Nov 15 [PubMed PMID: 39549816]
Yuen JC, Wallace JL, Steelman SC. Recalcitrant Bilateral Volar Hand Burn Contracture in a Toddler Treated With Integra and Staged Full-thickness Skin Graft: A Literature Review. Plastic and reconstructive surgery. Global open. 2025 Jan:13(1):e6430. doi: 10.1097/GOX.0000000000006430. Epub 2025 Jan 17 [PubMed PMID: 39830439]
Gil-Pallares P, Almenara-Blasco M, Navarro-Bielsa A, Gilaberte Y. Single-Stage Reconstruction of Large Scalp Surgical Defects With Exposed Bone Using Matriderm Flex and Full-Thickness Skin Grafts, a Case Report. The Australasian journal of dermatology. 2025 Feb:66(1):25-26. doi: 10.1111/ajd.14405. Epub 2024 Dec 30 [PubMed PMID: 39736104]
Level 3 (low-level) evidence. . :(): [PubMed PMID: 39868970]