Introduction
Spondylolisthesis, a condition characterized by the displacement of one vertebra relative to its adjacent counterpart, can be a significant cause of axial spine pain and radicular symptoms. Spondylolisthesis is derived from the Greek words spondylosis, which means vertebra, and olisthisis, which means slippage. The displacement of the cranial vertebra can be anterior (also known as anterolisthesis), lateral, or posterior (retrolisthesis) to the more caudal vertebra. Spondylolisthesis can happen at any anatomical level of the spine and can arise from various etiologies, including congenital, acquired, or idiopathic factors, and is graded based on the degree of vertebral slippage.[1] The anterolisthesis in the lumbar spine and the lumbosacral junction (L5-S1) are the most clinically relevant types.[2]
The clinical presentation of spondylolisthesis encompasses a broad spectrum, ranging from asymptomatic cases to those involving debilitating pain, neural compromise, and functional limitations. Given its complexity, a nuanced understanding of its pathophysiology, diagnostic evaluation, and management is essential for optimizing patient outcomes.
The natural history of spondylolisthesis is variable. In some cases, it reflects a progression influenced by underlying biomechanical instability and neural compromise, underscoring the importance of early and accurate condition classification. An intricate knowledge of the relevant spinal anatomy and the mechanisms contributing to vertebral instability and symptomatology forms the foundation for effective diagnosis and treatment.
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
The Wiltse classification offers a structured framework for understanding the etiology of spondylolisthesis, grouping spondylolisthesis into the following 5 principal categories based on distinct causative mechanisms:
- Dysplastic spondylolisthesis (Type I) is characterized by congenital malformations in the lumbosacral spine (eg, dysplastic upper sacrum or spina bifida occulta) that inherently compromise spinal stability. These structural abnormalities increase shear forces at the lumbosacral junction, often leading to early anterior displacement of one vertebra over another.
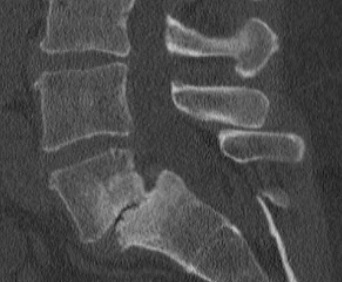
- Isthmic spondylolisthesis (Type II): This type arises from pathology in the pars interarticularis and is further subdivided based on the nature of the lesion (see Image. Isthmic Spondylolisthesis). Type IIA typically involves fatigue fractures resulting from repetitive hyperextension, frequently observed in young athletes in sports such as wrestling, football, and gymnastics. Type IIB is marked by elongated pars secondary to microtrauma and suboptimal healing. Type IIC involves acute traumatic fractures of the pars. Individuals with anatomically thinner pars cortices may exhibit a heightened predisposition to these stress-related injuries.
- Degenerative spondylolisthesis (Type III): Degenerative spondylolisthesis is closely linked to progressive degeneration of the intervertebral discs and facet joints, most commonly at the L4 to L5 level (see Image. Degenerative Spondylolisthesis). Age-related changes in bone density and altered spinal mechanics disrupt segmental stability and are frequently seen in older adults, particularly postmenopausal women.
- Traumatic spondylolisthesis (Type IV): Type IV results from acute fractures or dislocations of the posterior spinal elements, excluding the pars interarticularis. High-energy injuries, such as those sustained in motor vehicle accidents, are often implicated in this form of vertebral slippage.
- Pathologic spondylolisthesis (Type V): This type of spondylolisthesis is associated with systemic or localized disease processes, including neoplasms, infections, and metabolic bone disorders, that weaken the vertebral architecture and render the spine susceptible to displacement.
Additionally, iatrogenic spondylolisthesis can result postoperatively as a result of aggressive bone decompression. Taken together, the Wiltse classification underscores the multifactorial nature of spondylolisthesis etiology. While repetitive biomechanical stress factors prominently in isthmic and degenerative types, congenital anomalies in dysplastic spondylolisthesis illustrate the influence of developmental irregularities. Degenerative changes with advancing age highlight the cumulative impact of wear and tear on spinal stability. Traumatic and pathologic variations emphasize the roles of high-energy injury and disease processes, respectively, in undermining vertebral integrity.[3]
Epidemiology
Spondylolisthesis most commonly occurs in the lower lumbar spine but can also occur in the cervical spine and rarely, except for trauma, in the thoracic spine. Isthmic spondylolisthesis can be present in the adolescent and young adult population but may go unrecognized until symptoms develop in adulthood. A higher prevalence of isthmic spondylolisthesis in males has been observed. In the general adult population, the prevalence of spondylolysis ranges from 5% to 11.5%, while isthmic spondylolisthesis is observed in approximately 4% to 8% of individuals.[4]
Degenerative spondylolisthesis (Type III) predominantly affects older adults, with a marked predilection for females. A cross-sectional epidemiological survey involving over 4,000 patients reported a prevalence of 2.7% in men and 8.4% in women, underscoring a significant sex disparity.[5] The most common site for degenerative spondylolisthesis is the L4 to L5 level, whereas isthmic spondylolisthesis frequently involves the L5 to S1 level.
Pathophysiology
Spondylolisthesis is driven by a combination of biomechanical stressors, degenerative changes, and congenital abnormalities that compromise spinal stability. It can result from defects in the pars interarticularis, wear and tear in the intervertebral discs and facet joints, or congenital malformations of the posterior spinal elements. Each of these factors contributes to a series of pathological events that lead to the onset and progression of spondylolisthesis.
In isthmic spondylolisthesis, the central initiating factor is a defect or fracture in the pars interarticularis, often caused by repetitive mechanical stress. This lesion, termed spondylolysis, undermines the stability of the posterior vertebral elements, enabling the vertebral body to shift forward. Stress-related injuries frequently occur in adolescents and young adults who engage in activities featuring repeated hyperextension of the lumbar spine, eg, gymnastics or football.
Degenerative spondylolisthesis, in contrast, arises from the gradual deterioration of the spine's support structures, particularly the intervertebral discs and facet joints. Loss of disc height, facet joint arthritis, and diminished posterior column support transfer mechanical load to the anterior column, promoting vertebral translation. This form of spondylolisthesis most commonly affects the L4 to L5 level and is prevalent among older adults, especially women, due to age-related changes in bone density and spinal biomechanics.
Dysplastic spondylolisthesis occurs when congenital malformations of the lumbosacral spine, eg, underdeveloped facets or a dysplastic sacrum, increase susceptibility to vertebral slippage. These anomalies alter the biomechanics of the lumbosacral junction, leading to high-grade slips and significant deformities. Dysplastic features often create kyphotic deformities and sagittal imbalances, further intensifying spinal instability.
Muscles and ligaments also significantly influence spondylolisthesis pathophysiology. Atrophy of the multifidus muscle—common in isthmic spondylolisthesis—and compensatory hypertrophy of the erector spinae can undermine dynamic spinal stabilization and exacerbate vertebral slip.[6] Ligamentous compromise, including annular and posterior longitudinal ligament failure, further contributes to the progressive loss of stability over time.
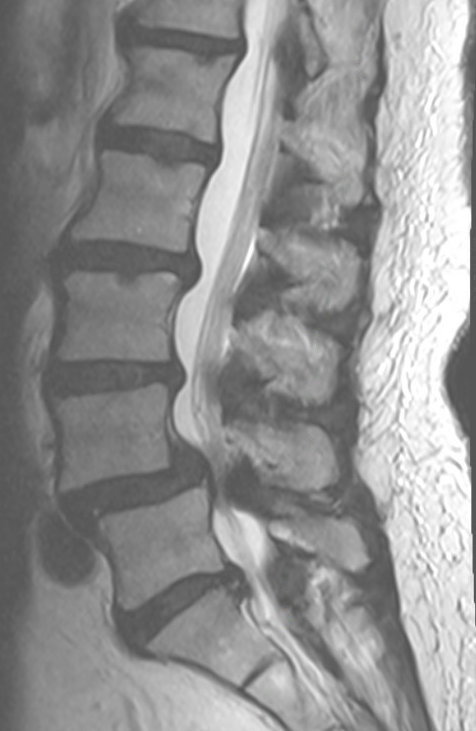
Additionally, spondylolisthesis often narrows the neural foramen and central canal, potentially compressing nerve roots. This can manifest as radiculopathy, neurogenic claudication, and other neurological symptoms, particularly in severe slips or advanced degenerative disease. Neuroforaminal stenosis is frequently aggravated by diminished disc height, forward vertebral translation, and osteophyte growth. In the presence of spinal instability, the stenosis can be static or deteriorate with movement (dynamic). Alterations in normal spinal anatomy, occasionally accompanied by instability, can trigger axial low back pain usually associated with movement.
The pelvic parameters, including pelvic incidence, sacral slope, and pelvic tilt, affect the spinal biomechanics associated with spondylolisthesis. A higher pelvic incidence magnifies shear forces at the lumbosacral junction, increasing the likelihood of slippage and deformity. Progressive vertebral translation can lead to sagittal imbalance, prompting compensatory changes in posture that further influence the spine's mechanical environment.
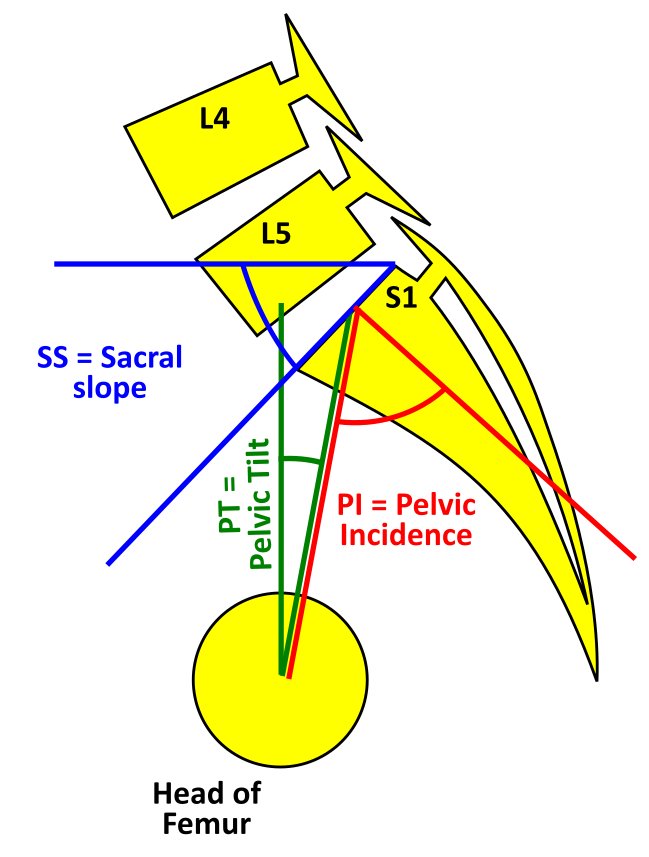
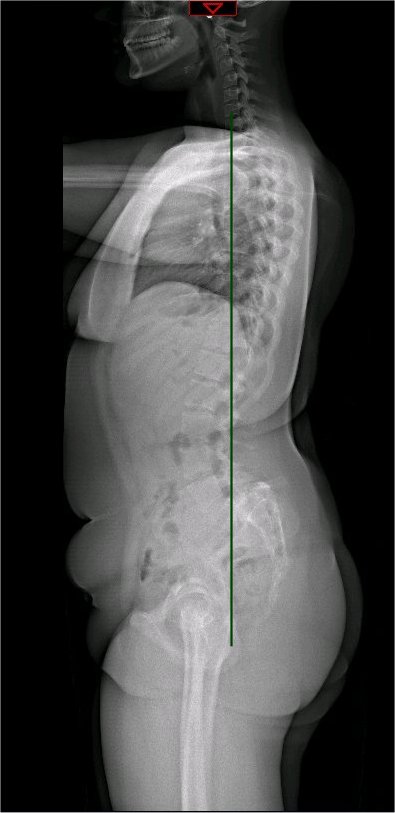
The spinopelvic balance and the global spinal alignment are essential in understanding the etiology, grading, and treatment protocol planning.[7] These parameters are measured on a standing lateral radiograph. The main parameters and their definitions are as follows (see Images. Spinopelvic Parameters and Whole Spine Radiograph):
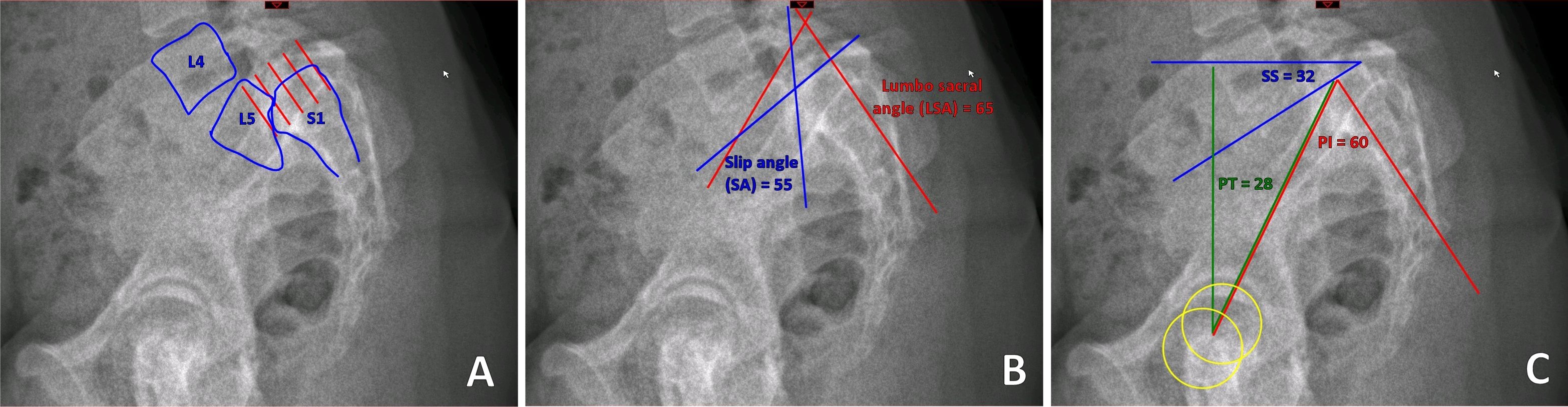
- Pelvic tilt (PT): PT is the angle measured between a line drawn from the center of the superior endplate of S1 to the center of rotation of the femoral head and the vertical reference line.[7]
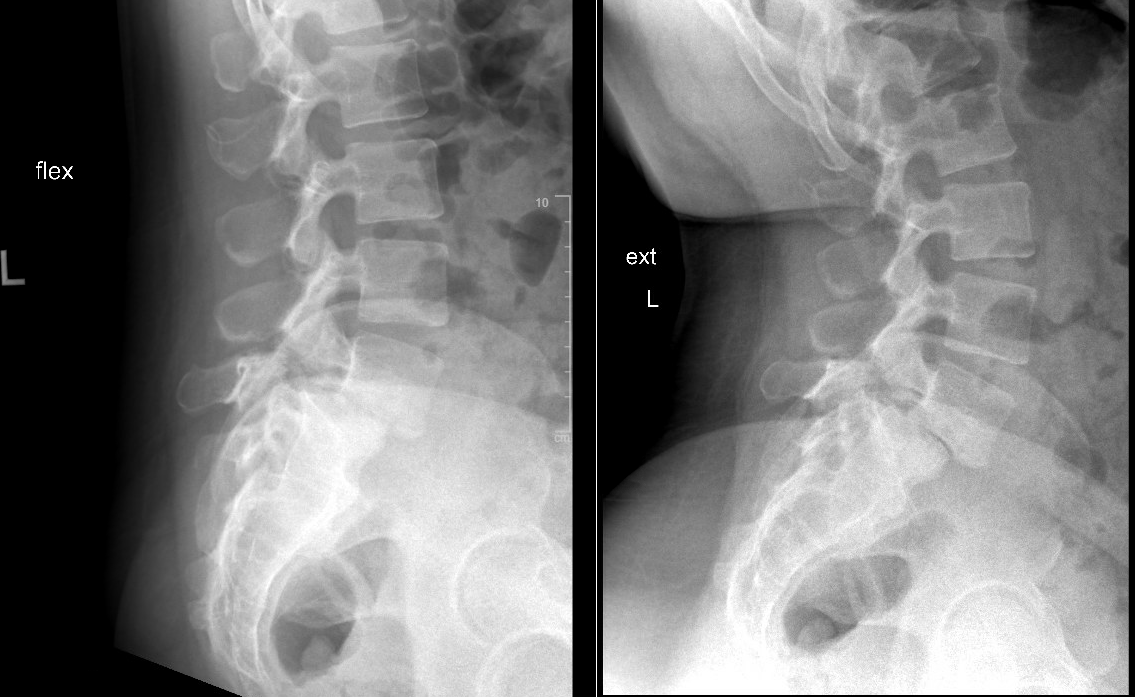
- Sacral slope (SS): SS is the angle between the line drawn along the superior end of the S1 endplate and the horizontal reference line.[7] The PT and the SS are referenced to the vertical and horizontal planes and, hence, can vary based on the position of the pelvis. Though the standard measurements are on a standing radiograph, the values of PT and SS alter between sitting and standing.
- Pelvic incidence (PI): PI is the angle between a line starting at the midline between centers of rotation of each femoral head drawn towards the midpoint of S1 superior endplate and a line perpendicular to the line drawn along the superior endplate of S1. PI = PT + SS (see Image. Spondylolisthesis Classification). The normal PI value is 50 degrees. An increased PI is associated with a higher severity of slips.[8] Compared to the PT and SS, the PI is a fixed value and does not change with the position of the pelvis or in adult life. A PI value of 70 to 80 degrees is present in patients with significant spondylolisthesis.
Iatrogenic spondylolisthesis can result after an operation where an aggressive bone decompression needs to be performed. This usually happens in cases where the laminectomy was extensive and significantly thinned the pars interarticularis, predisposing to iatrogenic spondylolysis. In summary, spondylolisthesis arises from a multifaceted interplay of mechanical, degenerative, and sometimes congenital factors that erode the structural and functional integrity of the spinal column. These processes shape the disorder's clinical presentation, progression, and neurologic complications.
History and Physical
Clinical History
Patients often report localized low back pain in lumbar spondylolisthesis or neck pain in cervical spondylolisthesis. This pain is typically intermittent and may worsen with flexion and extension of the affected spinal segments due to mechanical instability. Direct palpation of the affected area may also exacerbate the pain.
Radicular pain, characterized by sharp or shooting sensations radiating to the lower extremities, occurs when nerve roots are compressed by foraminal, lateral, recess, or central stenosis with or without disc protrusion. Symptoms may improve in specific positions, eg, lying supine, which reduces instability and decompresses neural structures. Associated symptoms include buttock pain, leg numbness or weakness, difficulty ambulating, and, in rare cases, bowel or bladder dysfunction.
Physical Examination
The physical examination should include inspection for step-off deformities or exaggerated lumbar lordosis, palpation for tenderness, and evaluation of lumbar range of motion, particularly noting pain during extension. Neurological assessment includes testing motor strength, sensory function, and reflexes, which may reveal deficits consistent with nerve root involvement. Provocative maneuvers, eg, the Stork test or hyperextension, may reproduce the pain and suggest mechanical instability. On this test, the patient stands on a leg with the other leg flexed at the knee and then extends the lumbar spine. If the lumbar pain is exacerbated, then the test is positive. The physical also includes gait examination and screening for concurrent pathologies, such as hip or sacroiliac joint dysfunction.
Evaluation
Routine laboratory investigations are generally not required unless systemic symptoms raise concerns about infection or metabolic disorders. In such cases, laboratory studies, including complete blood count, erythrocyte sedimentation rate, and C-reactive protein, can help identify infection, while metabolic tests, eg, calcium, vitamin D, and parathyroid hormone, help evaluate bone health.
Anteroposterior, lateral, and flexion-extension radiographs are first-line diagnostic tools, allowing assessment of vertebral alignment, dynamic instability, and grading using the Meyerding classification (see Images. Meyerding's Grading and Lumbar Spine Lateral X-Ray). Meyerding's classification of spondylolisthesis is based on the percentage of anterior translation of the slipped vertebra relative to the adjacent vertebra. Grade I spondylolisthesis is 1% to 25% slippage, grade II is 26% to 50% slippage, grade III is 51% to 75% slippage, and grade IV is 76% to 100% slippage (see Image. Grade II Spondylolisthesis). Grade V spondylolisthesis or spondyloptosis is >100% slippage.
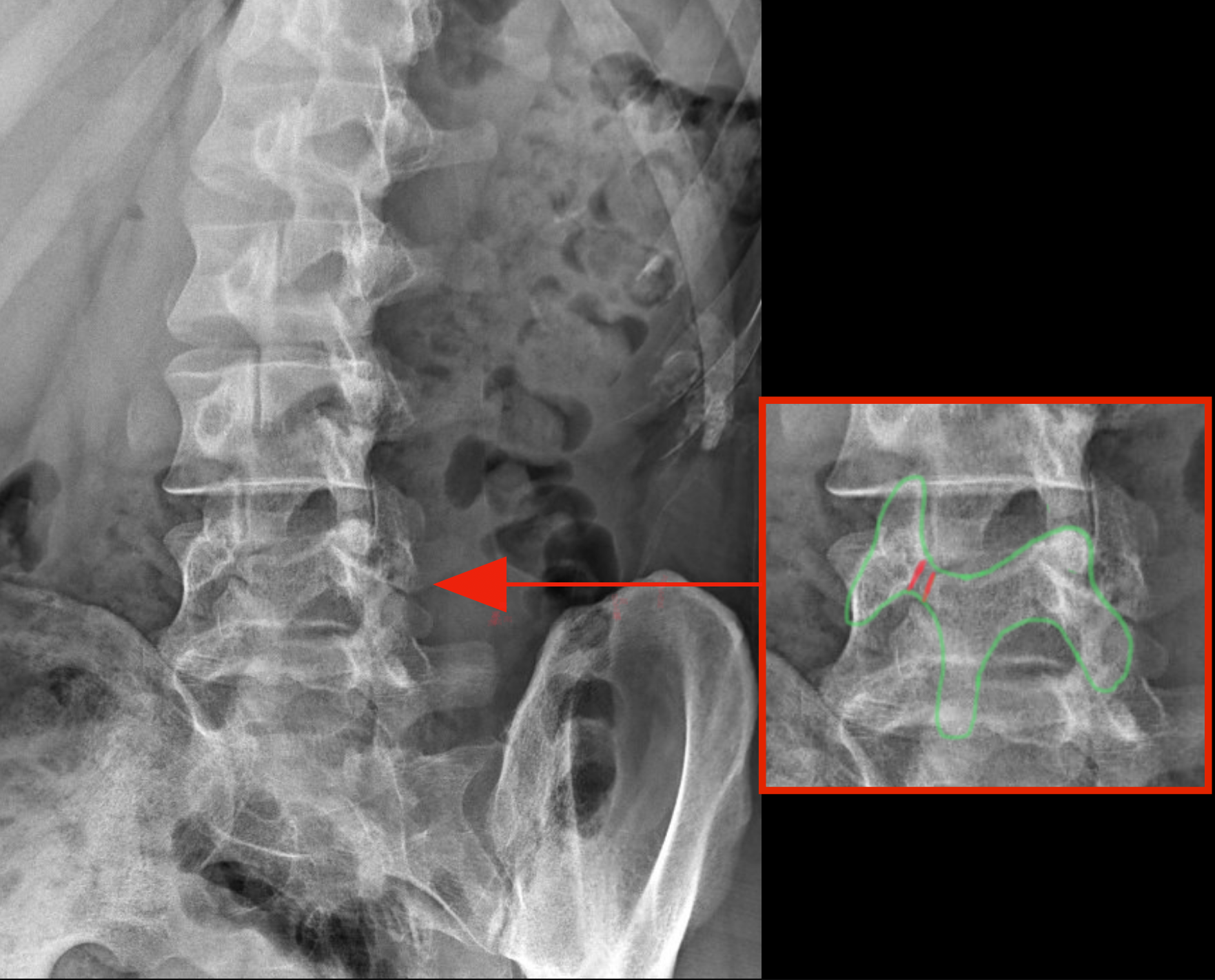
Oblique radiographs may reveal pars interarticularis defects, noted as the "scotty dog" sign, although their routine use is debated due to increased radiation exposure (see Image. "Scotty Dog" Finding). The "scotty dog collar" shows a defect where the collar would be on the cartoon dog, representing the pars interarticularis fracture. Full-length scoliosis films help characterize lumbosacral anatomy and global sagittal balance, particularly in high-grade spondylolisthesis.
Computed tomography (CT) is the gold standard for identifying pars defects and bony abnormalities, particularly with sagittal reconstructions. It is essential for preoperative planning, such as in evaluating the size of the pedicle for the selection of the pedicle screw size. Magnetic resonance imaging (MRI) is preferred for assessing neural elements, disc degeneration, and soft tissue pathology. MRI is particularly sensitive for detecting early stress reactions and bone marrow edema, though it may have limitations in identifying chronic pars defects. The degenerative spondylolisthesis is usually associated with central stenosis, while the isthmic spondylolisthesis typically is not. The reason is that the intact pars interarticularis in degenerative spondylolisthesis leads to a ventral slippage of the inferior articular processes and the lamina. In isthmic spondylolisthesis, the pars defects disconnect the inferior articular processes and the lamina, which are consequently not being displaced. Single photon emission CT can help detect early stress reactions or pars defects when findings on radiographs and CT are inconclusive.
The evaluation of spinopelvic parameters, including pelvic incidence, sacral slope, and pelvic tilt, is critical for understanding sagittal alignment and is especially important in high-grade slips to assess biomechanical strain and guide surgical strategies. Spinopelvic measurements are integral to planning corrective procedures, especially in cases with sagittal imbalance.[9][10] The Spinal Deformity Study Group has created a classification that guides treatment. This scale takes into account the spinopelvic parameters and the overall spinal alignment.[11]
- Type I: <50% slip, PI <45 degrees
- Type II: <50% slip, PI 45 to 60 degrees
- Type III: <50% slip, PI >60 degrees
- Type IV: >50% slip, balanced pelvis, balanced spine
- Type V: >50% slip, retroverted pelvis , balanced spine
- Type VI: >50% slip, retroverted pelvis, unbalanced spine
Several classifications exist for degenerative spondylolisthesis. A recent one is the University of California, San Francisco degenerative spondylolisthesis (UCSF DS) classification system, which assesses the segmental dynamic instability, the location of spinal stenosis, the sagittal alignment, and the primary clinical presentation (ie, leg and back pain).[12]
Treatment / Management
Nonsurgical Management
Nonoperative management is the first-line approach, focusing on symptom control and functional improvement. Conservative measures include NSAIDs, physical therapy, bracing, and activity modification. Physical therapy programs emphasize core stabilization, lumbar flexibility, and hamstring stretching, which aim to reduce mechanical strain and improve posture. Bracing may be employed for symptomatic instability, particularly in pediatric patients. Epidural steroid injections may provide temporary relief in cases of radicular pain. Medial branch blocks and subsequent radiofrequency ablation can be considered for low back pain associated with spondylolysis.
Surgical Management
Approximately 10% to 15% of younger patients with low-grade spondylolisthesis will fail conservative treatment and need surgical treatment. Patients with instability are more likely to require operative intervention. Surgical intervention is indicated for persistent pain unresponsive to conservative measures, neurological deficits, or progressive deformity. Dysplastic spondylolisthesis is treated more aggressively compared with isthmic spondylolisthesis due to a higher risk for neurological injury.[1]
In low-grade cases, decompression, usually combined with fusion, is the standard surgical approach. Specifically for adult patients with isthmic spondylolisthesis who undergo surgery, fusion is suggested for improved long-term clinical outcomes per North American Spine Society (NASS) guidelines.[9] NASS guidelines suggest decompression with fusion for the treatment of degenerative lumbar spondylolisthesis for improved clinical outcomes compared with decompression alone.[13] However, the same guidelines suggest that decompression alone with the preservation of midline structures provides similar outcomes compared with decompression-fusion in a subgroup of degenerative spondylolisthesis patients. This subgroup has symptomatic, low-grade, single-level degenerative spondylolisthesis without lateral foraminal stenosis. The UCSF DS classification system is a framework that has implications for selecting fusion vs decompression alone. Specifically, instability, foraminal stenosis, spine imbalance, and back pain might point toward fusion in this framework.[12] The degree of spondylolisthesis and the facet joint diastasis and effusion are 2 additional important factors that might point towards fusion in degenerative spondylolisthesis.[14]
The fusion option usually incorporates an interbody fusion component. There are several interbody fusion options, eg, posterior lumbar interbody fusion, transforaminal lumbar interbody fusion, or anterior lumbar interbody fusion (see Image. Transforaminal Lumbar Fusion). Minimally invasive variations of these procedures exist that might offer advantages in selected cases.[15] Interbody fusion can enhance fusion rates, facilitate reduction, and increase foraminal height, contributing to exiting nerve decompression.(A1)
The reduction of the spondylolisthesis is considered for achieving sagittal balance and decreasing the risk for further degenerative spinal changes in the future, particularly in high-grade spondylolisthesis, though a reduction is associated with higher technical demands and risks of neurological complications. The reduction goal is mainly restoring sagittal alignment versus reducing the anterior translation. In high-grade cases, aggressive reduction of anterior translation carries an increased risk for L5 radiculopathy due to traction injury. Fusion in situ (without changing the alignment of the spine) remains a viable option for stabilizing progression when minimizing surgical morbidity is a priority and when the spine alignment is satisfactory. Spinopelvic parameters, including pelvic incidence and sacral slope, are integral to surgical planning to ensure optimal alignment and load distribution. Reduction techniques might be considered for high-grade spondylolisthesis in patients with an unbalanced pelvis and should be strongly considered if a superimposed unbalanced spine is noted.[16][11] The reduction might require a vertebrectomy of L5 in challenging, high-grade spondylolisthesis cases.(B2)
Postoperative rehabilitation focuses on gradual reactivation with core strengthening and flexibility exercises. Regular postoperative imaging is critical for assessing fusion integrity and sagittal alignment.[17][18][13][19][20][9][21][22](A1)
Differential Diagnosis
The differential diagnosis of spondylolisthesis encompasses various conditions that present with similar symptoms, eg, low back pain and radiculopathy.
Degenerative lumbar disc disease is a common mimic of spondylolisthesis, encompassing entities, eg, discogenic pain syndrome, facet syndrome, and lumbosacral spondylosis. These conditions can present with mechanical low back pain or radiculopathy but typically lack evidence of vertebral displacement on imaging. Similarly, lumbar disc herniation frequently causes radicular symptoms and sensory deficits but is differentiated by the absence of vertebral slippage and by imaging findings of disc extrusion or protrusion.
Spinal stenosis, particularly when severe, can present with neurogenic claudication and nerve root compression, features that overlap with advanced spondylolisthesis. However, imaging in spinal stenosis reveals canal narrowing rather than translational displacement of vertebrae. Sacroiliac joint dysfunction, often presenting as localized back pain and gait disturbances, can mimic mechanical instability pain but is distinguished by a lack of neurological deficits and characteristic physical examination findings.
Developmental abnormalities, including spina bifida occulta and congenital scoliosis, are significant in the pediatric population. These disorders may present with deformities resembling spondylolisthesis but lack vertebral displacement. Hip pathology, eg, labral tears or advanced arthritis, should also be considered in cases of radiating pain, as these conditions may simulate the symptoms of lumbar instability or radiculopathy.
Infections, including discitis and osteomyelitis, should be suspected in the presence of systemic symptoms, eg, fever and elevated inflammatory markers. Imaging in these cases demonstrates inflammatory changes without evidence of vertebral slippage. Similarly, spinal tumors may present with pain and neurological deficits, necessitating imaging to identify mass lesions or vertebral destruction.
Imaging plays a critical role in differentiating these conditions. While plain radiographs are often adequate for detecting significant vertebral displacement, advanced imaging such as MRI or CT is necessary to identify subtle pars defects, stress reactions, or other pathologies. For instance, MRI may be preferred in early-stage stress reactions due to its sensitivity to edema, whereas CT is superior in visualizing bony abnormalities. Failure to recognize mimicking conditions risks mismanagement and potential progression of underlying pathology. Finally, the possibility of 2 or more concurrent pathologies is not rare. For example, the concurrent presence of spondylolisthesis in a level and stenosis in an adjacent level.
Prognosis
The natural history of low-grade spondylolisthesis is typically favorable, with many patients remaining asymptomatic or experiencing minimal progression over time. In pediatric patients, spondylolysis and low-grade slips often remain stable, and in some cases, pars defects may undergo spontaneous healing. Healing rates are highest in unilateral pars defects or lesions with early radiographic features of healing, such as bony edema. Adults with low-grade slips rarely experience significant progression; however, age-related degenerative changes, such as superimposed disk degeneration, can lead to clinical deterioration.
High-grade spondylolisthesis carries a more guarded prognosis due to the higher likelihood of progression and complications. In pediatric patients, growth spurts and immature vertebral anatomy increase the risk of slip progression, particularly in those with high pelvic incidence or sagittal imbalance. In adults, high-grade slips often present with significant mechanical instability, sagittal malalignment, and neurological symptoms. Surgical intervention is frequently required in such cases and generally results in favorable outcomes when sagittal alignment and neural decompression are achieved. However, the potential for complications, including adjacent segment degeneration and pseudarthrosis, necessitates careful surgical planning and follow-up. Continued advances in surgical techniques and rehabilitation strategies further enhance prognosis across all age groups and severities.
Complications
Spondylolisthesis is associated with several complications, which, if not appropriately addressed, can significantly affect functional capacity, quality of life, and long-term outcomes. Regular follow-up and tailored management strategies, including conservative and surgical approaches, are essential to optimizing outcomes and minimizing the risk of adverse events.
Neurological Complications
Neurological complications in the form of radiculopathy are among the most concerning and include pain, motor weakness, and sensory deficits due to nerve root compression. Chronic compression may lead to irreversible neurological deficits even after decompression surgery. Severe cases may culminate in cauda equina syndrome, characterized by bowel or bladder dysfunction and saddle anesthesia, requiring immediate surgical intervention.
Vertebral Alignment Complications
Spine alignment complications are frequently observed, particularly in high-grade slips. Progressive vertebral displacement increases the risk of sagittal imbalance, resulting in compensatory postural changes and impaired gait. Such imbalances may further exacerbate pain and instability, particularly in patients with high pelvic incidence. Untreated high-grade spondylolisthesis in pediatric patients can lead to long-term kyphotic deformities, which compromise spinal alignment and functional capacity.
Long-Term Complications
Adjacent segment degeneration, resulting from altered spinal biomechanics, is a notable long-term complication, especially in patients who undergo spinal fusion. Pain-related complications are significant and may persist despite management. Mechanical back pain often arises from disk degeneration, facet joint arthritis, or pars pseudarthrosis. In both untreated and surgically managed cases, chronic pain may become refractory, requiring interprofessional care.
Surgical Complications
Surgical complications, although relatively infrequent with modern techniques, can include pseudarthrosis, adjacent segment disease, and infections. Pseudarthrosis, or nonunion of the fusion site, is more common in patients with risk factors such as smoking, osteoporosis, or suboptimal surgical conditions. The typical imaging findings include fracture of hardware, bone lucencies around the screws, and absence of a continuous bone bridging the adjacent vertebrae in the operated level. Adjacent segment disease, which can develop due to altered biomechanics after fusion, may necessitate further surgical intervention. Postoperative infections can also occur, particularly in older or immunocompromised patients.
Growth-Related Complications
Pediatric and adolescent patients are at unique risk for growth-related complications. During growth spurts, untreated spondylolisthesis may result in significant sagittal deformity and kyphosis, which can have long-term impacts on mobility and functional independence. Early identification and management are essential to prevent these outcomes.
Psychological Complications
Psychological complications should also be considered. Chronic pain, functional impairment, and progressive deformity may lead to depression, anxiety, and reduced quality of life. These issues underscore the importance of a comprehensive, interprofessional approach to care.
Postoperative and Rehabilitation Care
Postoperative and rehabilitation care for spondylolisthesis is critical for optimizing surgical outcomes, minimizing complications, and restoring functional independence. Immediate postoperative care focuses on early mobilization to prevent complications, eg, deep vein thrombosis and pulmonary embolism. Patients are typically encouraged to ambulate by the first postoperative day. When indicated, a bilateral lower extremity Doppler ultrasound may be performed to screen for deep vein thrombosis. Pain management strategies include patient-controlled analgesia initially, followed by oral analgesics. Muscle relaxants (eg, diazepam) are often prescribed for postoperative muscle spasms. Epidural catheters may be used in select cases for enhanced pain control during the early recovery phase.
Radiological evaluation in the immediate postoperative period is obtained to confirm the absence of hardware complications and to function as a baseline study for future comparisons. Standing radiographs or CT scans are commonly performed at discharge and then x-rays during follow-up visits. The exact time intervals for imaging depend on surgeon preferences and the level of concern for potential complications. Bracing, eg, lumbar-sacral orthoses, may be used selectively based on the surgeon’s preference and the patient’s stability needs.
Rehabilitation programs are essential for restoring function and preventing future complications. The initial phase focuses on protecting the surgical site through activity modifications and gentle stretching exercises. Core stabilization exercises are introduced to strengthen paraspinal and abdominal musculature, reducing mechanical strain on the spine. Low-impact aerobic activities like walking, swimming, or cycling are typically initiated postoperatively to enhance cardiovascular fitness without placing excessive stress on the spine.
As recovery progresses, patients transition to intensive strengthening exercises to improve spinal stability and posture. Manual laborers may require work-conditioning programs that simulate physical demands before resuming full occupational duties. Depending on functional capacity evaluations and the complexity of the procedure, return to heavy physical work is typically delayed until 3 to 6 months postoperatively.
Patients are generally discharged by postoperative day 5, with driving restrictions for usually 4 weeks. Those in sedentary occupations may return to work within 4 to 6 weeks, while those in physically demanding roles require more extended recovery periods. Long-term follow-up is vital to monitor spinal alignment, fusion progress, and hardware integrity. Patients should undergo routine imaging and be evaluated for complications such as adjacent segment degeneration or pseudarthrosis. Maintenance of a healthy lifestyle, adherence to rehabilitation exercises, and avoidance of high-impact activities are emphasized to preserve surgical outcomes. A structured, patient-centered approach to postoperative and rehabilitation care ensures optimal recovery, functional independence, and prevention of long-term complications. Collaboration between surgical teams, physical therapists, and patients is essential for achieving these goals.
Pearls and Other Issues
Meyerding’s classification of spondylolisthesis is the most commonly used grading method. Its basis is on the percentage of anterior translation relative to the adjacent level. Grade I spondylolisthesis is 1% to 25% slippage, grade II is up to 50% slippage, grade III is up to 75% slippage, and grade IV is 76% to 100% slippage. More than 100% slippage is known as spondyloptosis or grade V spondylolisthesis.
Subclasses of isthmic spondylolisthesis are subtype A (stress fractures of the pars), subtype B (elongation of the pars without overt fracture), and subtype C (acute fracture of the pars). Subclasses of pathologic spondylolisthesis are subtype A (systemic causes) and subtype B (focal processes).
Enhancing Healthcare Team Outcomes
Effective management of spondylolisthesis relies on an interprofessional team approach to deliver patient-centered care, enhance outcomes, and ensure safety. A collaborative team comprising physicians, advanced practitioners, nurses, physical therapists, chiropractors, and pharmacists is crucial. Physicians or advanced practitioners lead the diagnostic and management process, coordinating surgical or nonoperative treatments. Nurses play essential roles in preoperative, intraoperative, and postoperative care, ensuring patient safety and continuity of care. Physical therapists focus on rehabilitation, informing the team of progress and providing crucial input for treatment adjustments. Chiropractors may identify the condition with imaging, facilitating early intervention. Pharmacists support pain management and optimize medication safety.
Regular interprofessional communication ensures smooth transitions between care phases, whether surgical or conservative. Strategies like encouraging weight loss in high BMI cases and aligning rehabilitation goals promote recovery. This coordinated effort enhances patient outcomes, minimizes complications, and strengthens team performance.
Media
(Click Image to Enlarge)
(Click Image to Enlarge)
(Click Image to Enlarge)
(Click Image to Enlarge)

Transforaminal Lumbar Fusion. L5-S1 isthmic spondylolisthesis: preoperative (left) and postoperative image (right). A transforaminal lumbar interbody fusion was performed using an expandable cage and pedicle screws-rod instrumentation. Note the reduction of the anterior translation and the improvement of the segmental sagittal alignment at L5-S1.
Contributed by K Margetis, MD
(Click Image to Enlarge)
(Click Image to Enlarge)
(Click Image to Enlarge)
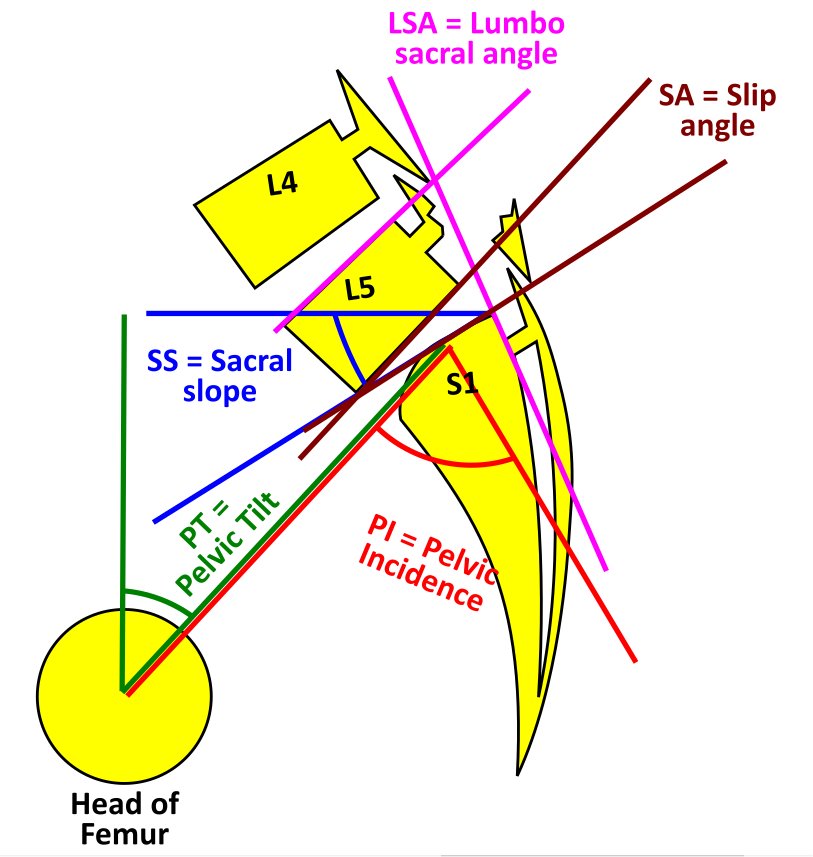
Spondylolisthesis Classification. Spondylolysis with spondylolisthesis, showing spinopelvic parameters, slip, dysplasia of upper endplate of S1, Slip angle (SA = angle between inferior endplate of L5 and a line perpendicular to the S1 posterior wall), and lumbosacral angle (LSA = angle between the superior endplate of L5 and posterior wall of the S1).
Contributed by G Ampat, MBBS, MS, FRCS
(Click Image to Enlarge)
(Click Image to Enlarge)
(Click Image to Enlarge)
References
Randall RM, Silverstein M, Goodwin R. Review of Pediatric Spondylolysis and Spondylolisthesis. Sports medicine and arthroscopy review. 2016 Dec:24(4):184-187 [PubMed PMID: 27811518]
Sassack B, Carrier JD. Anatomy, Back, Lumbar Spine. StatPearls. 2025 Jan:(): [PubMed PMID: 32491548]
Wiltse LL, Newman PH, Macnab I. Classification of spondylolisis and spondylolisthesis. Clinical orthopaedics and related research. 1976 Jun:(117):23-9 [PubMed PMID: 1277669]
Mohile NV, Kuczmarski AS, Lee D, Warburton C, Rakoczy K, Butler AJ. Spondylolysis and Isthmic Spondylolisthesis: A Guide to Diagnosis and Management. Journal of the American Board of Family Medicine : JABFM. 2022 Dec 23:35(6):1204-1216. doi: 10.3122/jabfm.2022.220130R1. Epub 2022 Dec 16 [PubMed PMID: 36526328]
Jacobsen S, Sonne-Holm S, Rovsing H, Monrad H, Gebuhr P. Degenerative lumbar spondylolisthesis: an epidemiological perspective: the Copenhagen Osteoarthritis Study. Spine. 2007 Jan 1:32(1):120-5 [PubMed PMID: 17202902]
Level 2 (mid-level) evidenceThakar S, Sivaraju L, Aryan S, Mohan D, Sai Kiran NA, Hegde AS. Lumbar paraspinal muscle morphometry and its correlations with demographic and radiological factors in adult isthmic spondylolisthesis: a retrospective review of 120 surgically managed cases. Journal of neurosurgery. Spine. 2016 May:24(5):679-85. doi: 10.3171/2015.9.SPINE15705. Epub 2016 Jan 15 [PubMed PMID: 26771373]
Level 2 (mid-level) evidenceTebet MA. Current concepts on the sagittal balance and classification of spondylolysis and spondylolisthesis. Revista brasileira de ortopedia. 2014 Jan-Feb:49(1):3-12. doi: 10.1016/j.rboe.2014.02.003. Epub 2014 Feb 18 [PubMed PMID: 26229765]
Hanson DS, Bridwell KH, Rhee JM, Lenke LG. Correlation of pelvic incidence with low- and high-grade isthmic spondylolisthesis. Spine. 2002 Sep 15:27(18):2026-9 [PubMed PMID: 12634563]
Level 1 (high-level) evidenceKreiner DS, Baisden J, Mazanec DJ, Patel RD, Bess RS, Burton D, Chutkan NB, Cohen BA, Crawford CH 3rd, Ghiselli G, Hanna AS, Hwang SW, Kilincer C, Myers ME, Park P, Rosolowski KA, Sharma AK, Taleghani CK, Trammell TR, Vo AN, Williams KD. Guideline summary review: an evidence-based clinical guideline for the diagnosis and treatment of adult isthmic spondylolisthesis. The spine journal : official journal of the North American Spine Society. 2016 Dec:16(12):1478-1485. doi: 10.1016/j.spinee.2016.08.034. Epub 2016 Sep 1 [PubMed PMID: 27592807]
Alqarni AM, Schneiders AG, Cook CE, Hendrick PA. Clinical tests to diagnose lumbar spondylolysis and spondylolisthesis: A systematic review. Physical therapy in sport : official journal of the Association of Chartered Physiotherapists in Sports Medicine. 2015 Aug:16(3):268-75. doi: 10.1016/j.ptsp.2014.12.005. Epub 2015 Jan 8 [PubMed PMID: 25797410]
Level 1 (high-level) evidenceLabelle H, Mac-Thiong JM, Roussouly P. Spino-pelvic sagittal balance of spondylolisthesis: a review and classification. European spine journal : official publication of the European Spine Society, the European Spinal Deformity Society, and the European Section of the Cervical Spine Research Society. 2011 Sep:20 Suppl 5(Suppl 5):641-6. doi: 10.1007/s00586-011-1932-1. Epub 2011 Aug 2 [PubMed PMID: 21809015]
Rangwalla K, Filley A, El Naga A, Gendelberg D, Baldwin A, Maziad A, Arora A, Wague A, O'Donnell J, Chryssikos T, Kasir R, Shah J, Theologis A, Tan L, Mummaneni P, Alamin T, Berven SH. Degenerative lumbar spondylolisthesis: review of current classifications and proposal of a novel classification system. European spine journal : official publication of the European Spine Society, the European Spinal Deformity Society, and the European Section of the Cervical Spine Research Society. 2024 May:33(5):1762-1772. doi: 10.1007/s00586-023-07818-x. Epub 2023 Aug 6 [PubMed PMID: 37543967]
Matz PG, Meagher RJ, Lamer T, Tontz WL Jr, Annaswamy TM, Cassidy RC, Cho CH, Dougherty P, Easa JE, Enix DE, Gunnoe BA, Jallo J, Julien TD, Maserati MB, Nucci RC, O'Toole JE, Rosolowski K, Sembrano JN, Villavicencio AT, Witt JP. Guideline summary review: An evidence-based clinical guideline for the diagnosis and treatment of degenerative lumbar spondylolisthesis. The spine journal : official journal of the North American Spine Society. 2016 Mar:16(3):439-48. doi: 10.1016/j.spinee.2015.11.055. Epub 2015 Dec 8 [PubMed PMID: 26681351]
Schönnagel L, Caffard T, Zhu J, Tani S, Camino-Willhuber G, Amini DA, Haffer H, Muellner M, Guven AE, Chiapparelli E, Arzani A, Amoroso K, Shue J, Duculan R, Zippelius T, Sama AA, Cammisa FP, Girardi FP, Mancuso CA, Hughes AP. Decision-making Algorithm for the Surgical Treatment of Degenerative Lumbar Spondylolisthesis of L4/L5. Spine. 2024 Feb 15:49(4):261-268. doi: 10.1097/BRS.0000000000004748. Epub 2023 Jun 15 [PubMed PMID: 37318098]
Jagtiani P, Karabacak M, Margetis K. Comparative Effectiveness of Open Versus Minimally Invasive Transforaminal Lumbar Interbody Fusion: An Umbrella Review of Meta-Analyses. Clinical spine surgery. 2024 Jul 1:37(6):E225-E238. doi: 10.1097/BSD.0000000000001561. Epub 2024 Jan 8 [PubMed PMID: 38245811]
Level 1 (high-level) evidenceHresko MT, Labelle H, Roussouly P, Berthonnaud E. Classification of high-grade spondylolistheses based on pelvic version and spine balance: possible rationale for reduction. Spine. 2007 Sep 15:32(20):2208-13 [PubMed PMID: 17873812]
Level 2 (mid-level) evidenceBouras T, Korovessis P. Management of spondylolysis and low-grade spondylolisthesis in fine athletes. A comprehensive review. European journal of orthopaedic surgery & traumatology : orthopedie traumatologie. 2015 Jul:25 Suppl 1():S167-75. doi: 10.1007/s00590-014-1560-7. Epub 2014 Nov 14 [PubMed PMID: 25394940]
Baker JF, Errico TJ, Kim Y, Razi A. Degenerative spondylolisthesis: contemporary review of the role of interbody fusion. European journal of orthopaedic surgery & traumatology : orthopedie traumatologie. 2017 Feb:27(2):169-180. doi: 10.1007/s00590-016-1885-5. Epub 2016 Nov 25 [PubMed PMID: 27888353]
Schöller K, Alimi M, Cong GT, Christos P, Härtl R. Lumbar Spinal Stenosis Associated With Degenerative Lumbar Spondylolisthesis: A Systematic Review and Meta-analysis of Secondary Fusion Rates Following Open vs Minimally Invasive Decompression. Neurosurgery. 2017 Mar 1:80(3):355-367. doi: 10.1093/neuros/nyw091. Epub [PubMed PMID: 28362963]
Level 1 (high-level) evidenceKoreckij TD, Fischgrund JS. Degenerative Spondylolisthesis. Journal of spinal disorders & techniques. 2015 Aug:28(7):236-41. doi: 10.1097/BSD.0000000000000298. Epub [PubMed PMID: 26172828]
Schulte TL, Ringel F, Quante M, Eicker SO, Muche-Borowski C, Kothe R. Surgery for adult spondylolisthesis: a systematic review of the evidence. European spine journal : official publication of the European Spine Society, the European Spinal Deformity Society, and the European Section of the Cervical Spine Research Society. 2016 Aug:25(8):2359-67. doi: 10.1007/s00586-015-4177-6. Epub 2015 Sep 12 [PubMed PMID: 26363561]
Level 1 (high-level) evidenceAlfieri A, Gazzeri R, Prell J, Röllinghoff M. The current management of lumbar spondylolisthesis. Journal of neurosurgical sciences. 2013 Jun:57(2):103-13 [PubMed PMID: 23676859]