Introduction
Accessing the central skull base has challenged surgeons for decades, given its location. Pituitary surgery or hypophysectomy has evolved over the last century from open surgery requiring craniotomy to a fully endoscopic endonasal procedure through the sphenoid sinus. In 1907, Hermann Schloffer (1868–1937), an Austrian surgeon from Vienna, performed the first transsphenoidal resection of a pituitary adenoma at the University of Innsbruck using a 3-stage operation that involved a lateral rhinotomy approach with resection of the sphenoid and ethmoid sinuses. Although the procedure marked a pioneering attempt, the patient developed cerebrospinal fluid rhinorrhea postoperatively and succumbed to intracranial hypertension 2 months later, with postmortem findings showing residual tumor obstructing the foramen of Monro.[1][2]
This transsphenoidal approach was described and popularized in 1910 by Harvey Cushing and Oskar Hirsch, utilizing sublabial and transnasal routes, respectively. However, the popularity of the transsphenoidal approach diminished when Cushing abandoned it for transcranial approaches. Norman Dott and Gerard Guiot maintained an interest in the transsphenoidal approach. In 1965, Canadian neurosurgeon Jules Hardy, following a 1-year fellowship with Guiot in Paris, revolutionized pituitary surgery by introducing the operating microscope and incorporating fluoroscopy, laying the foundation for modern techniques and establishing himself as a pioneer in microadenoma surgery.
In 1994, Egyptian surgeons Ahmed Gamea, Ahmed El-Guindy, and Salah Fathi pioneered endoscopy-assisted transsphenoidal surgery for pituitary adenomas, which evolved into a purely endoscopic approach for tumor removal. A major advancement came from Hae Dong Jho and Ricardo Carrau at the University of Pittsburgh, who popularized purely endoscopic endonasal surgery without a transsphenoidal retractor or microscope. Around the same time, Naples' Paolo Cappabianca and Enrico de Divitiis developed a mononostril endoscopic transsphenoidal approach, offering better visualization, reduced trauma, and improved patient tolerance, which became a cornerstone of European advancements in endonasal endoscopy.
Approaches to the pituitary gland can be broadly classified as transcranial or extracranial. Transcranial approaches—the anterior (subfrontal) and pterional approaches—are currently used when transsphenoidal approaches are contraindicated. The frontotemporal pterional approach involves removing part of the sphenoid wing, requires minimal brain retraction, provides the shortest trajectory to the parasellar region, and offers excellent visualization of the pituitary gland.
A subfrontal approach allows direct visualization of the pituitary tumor between the optic nerves. Still, it is less popular than the pterional approach due to the potential for injury to the olfactory nerves and entry into the frontal sinus. Extracranial approaches to the pituitary gland include the transsphenoidal microscopic (transnasal or sublabial) and the transnasal endoscopic approaches. Surgical modifications involve expanding the endoscopic endonasal approach and combining the transsphenoidal and transmaxillary approaches.
Microscopic approaches use sublabial or septal incisions, a wide dissection of the nasal mucosa on the septum and floor of the nose, with partial resection of the vomer, the perpendicular plate of the ethmoid, and the sphenoid rostrum. A self-retaining speculum holds the nasal contents laterally, allowing for bimanual instrumentation using the operative microscope. This technique may result in significant facial pain and swelling and requires nasal packing or septal splints. Potential sinonasal complications include sinusitis, numbness of the upper alveolus, nasal synechiae, and septal perforation.
Endoscopic pituitary surgery uses the medial nasal corridor to assess the sphenoid sinus.[3] This approach has gained popularity due to a shortened hospital stay and enhanced lateral surgical visualization with angled viewing capability.[4][5] Endoscopic transsphenoidal hypophysectomy represents a minimally invasive approach to skull base surgery with the potential for anterior fossa extension.[6]
Anatomy and Physiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Anatomy and Physiology
Surgical Anatomy of the Pituitary Gland
The pituitary gland resides within the sella turcica (pituitary fossa), a midline saddle-shaped depression in the sphenoid bone. The sella turcica has the following key features:
- Floor: Formed by the posterior roof of the sphenoid sinus
- Anterior wall: Begins at the posterior aspect of the tuberculum sella
- Lateral walls: Made of a dural reflection that extends between the anterior and posterior clinoid processes, beyond which lie the cavernous sinuses
- The cavernous sinuses contain cranial nerves III, IV, V1, and VI and the cavernous carotid arteries in the inferomedial aspect.
- Posterior wall: The vertical dorsum sellae, with the posterior clinoid processes on each side
- Superior wall: Comprised of a dural fold known as the diaphragma sella
- Intercommunicating sinuses beneath this structure connect the cavernous sinuses.
The following structures limit the suprasellar compartment:
- Inferiorly: Diaphragma sella
- Superiorly: Optic chiasm (situated in the chiasmatic sulcus) and floor of the 3rd ventricle
- Anteriorly: Anterior arachnoid membrane of the chiasmatic cistern
- Posteriorly: Diencephalic portion of Liliequist membrane
The chiasmatic sulcus leads laterally to the optic canals. Anterior to the chiasmatic groove is the planum sphenoidale, forming the roof of the sphenoid sinus.
Endoscopic Anatomy
The endoscope provides a minimally invasive approach to the sphenoid sinus and pituitary gland. Surgeons use a medial corridor between the nasal septum and middle turbinate to access the sphenoid ostia. These ostia are typically located in the following regions:
- Medial to the superior turbinate
- 1.5 cm above the choana
- 7 cm from the nasal sill at a 30° angle
- 11 mm from the skull base [7]
Sphenoid Sinus Variations
The sphenoid sinus exhibits variations in pneumatization, which include the following:
- Type I or conchal: The sphenoid sinus is either absent or minimally pneumatized, separated from the sella turcica by a wall approximately 10 mm thick. Although the bony barrier to the sella presents a clear challenge for the surgical approach, it does not serve as a contraindication for a transsphenoidal procedure.
- Type II or presellar: The posterior wall of the sphenoid sinus lies anterior to the anterior wall of the sella.
- Type III or sellar: The posterior wall of the sinus is located between the anterior and posterior walls of the sella. This type is the most common and ideal anatomical configuration for a transsphenoidal approach to the sella turcica, with the sella floor bulging into the operative field and providing direct visibility and access during the procedure.[8][9][10]
- Type IV or postsellar (occipitosphenoid): The posterior wall of the sinus extends beyond the dorsum sella, even across the occipital synchondrosis.[11] This type of pneumatization, characterized by spacious sinuses, a long intercarotid distance, and a thin anterior wall and floor of the sella turcica, provides favorable anatomical conditions for performing transsphenoidal hypophysectomy.[12]
Pneumatization of the sphenoid sinus can extend into various adjacent structures, including the following:
- The greater wing of the sphenoid, extending beyond a vertical line passing through the foramen rotundum
- The pterygoid plates, extending below a horizontal plane crossing the Vidian canal
- The anterior or posterior clinoid processes
Lateral pneumatization into a lateral recess can expose the maxillary (V2) and vidian nerves.
Key structures within the sphenoid sinus
The main structures encountered in a well-pneumatized sphenoid sinus include the optic nerve, internal carotid artery (ICA), and the lateral opticocarotid recess (OCR). The optic nerve is located at the junction of the lateral wall of the cavernous sinus. The ICA extends superiorly from the cavernous sinus, with its anterior genu within the sinus. The lateral OCR is a triangular bony depression representing the ventral surface of the optic strut. This structure marks the intersection of the paraclinoid carotid artery and optic canal.
Anatomical Challenges in Endonasal Surgery
Several anatomical variations can pose challenges during endonasal transsphenoidal surgery. For example, a deviated nasal septum or a concha bullosa can narrow the nasal passage, hindering instrument access and potentially affecting harvesting of the nasoseptal flap for reconstruction. Sinonasal disease or nasal polyps may necessitate additional sinus surgery to clear the nasal passages, further complicating the procedure.
Variations in sphenoid sinus anatomy, including pneumatization, septal attachments, and bony dehiscence (which may expose the optic nerve or carotid artery), require careful preoperative evaluation using computed tomography scans. Onodi cells—posterior ethmoid air cells that extend laterally and superiorly into the sphenoid sinus—can become pneumatized and increase the risk of optic nerve injury during surgery.
Important Considerations
Optic nerve dehiscence and carotid artery injury are some potential challenges during procedures involving the pituitary region. The optic nerve canal is dehiscent (lacking bony covering) in 4% to 8% of cases, increasing the risk of injury.[13] Meanwhile, the risk of carotid artery injury is approximately 5% in pituitary surgery and higher in parasellar surgery.[14] Surgeons must be aware of these risks and take precautions to avoid complications.
Indications
Pituitary adenomas represent the most common indication for transsphenoidal hypophysectomy.[15] Surgery is indicated for nonsecreting adenomas that are symptomatic and cause visual abnormalities, such as bilateral peripheral visual field loss, or demonstrate progressive enlargement on sequential magnetic resonance imaging (MRI). Patients with contraindications to MRI, such as an MRI-incompatible cardiac pacemaker, may be followed with computed tomography scans.
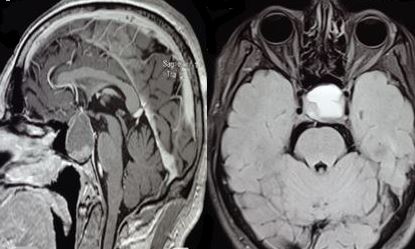
Surgery may be necessary in cases of pituitary apoplexy with acute-onset headaches and visual changes, such as diplopia or visual loss (see Images. Pituitary Apoplexy, Sagittal and Axial Views and Pituitary Apoplexy, Sagittal View). Surgery is recommended for secreting adenomas that cause Cushing disease, acromegaly, or amenorrhea, causing infertility. Other parasellar lesions may be approached surgically if symptomatic, including craniopharyngiomas, meningiomas, chordomas, and metastatic lesions. Rathke cleft cysts usually do not require surgery because they are asymptomatic and do not progressively enlarge. Hypopituitarism without visual abnormalities in a nonsecreting adenoma is generally monitored with sequential imaging.
The primary objectives of surgical treatment for patients with adenomas, guided by neurological and endocrinological symptoms, are as follows:
- Achieving maximal safe tumor removal to alleviate mass effects or symptoms of hormonal hypersecretion.
- Restoring or preserving normal neurological functions, focusing on visual function.
- Decompressing the pituitary gland to enhance or maintain its residual hormonal function.
Contraindications
The transsphenoidal approach is contraindicated in sphenoid sinusitis. Intrasellar vascular anomalies, ectatic midline carotid arteries, or significant lateral suprasellar tumor extension into the temporal fossa are relative contraindications depending on the expertise available. Suprasellar extension of a large pituitary adenoma above the sella turcica that is technically inaccessible with conventional instrumentation may be more appropriately approached by a craniotomy. Other relative contraindications include a poorly pneumatized sphenoid sinus and extension above a constrictive diaphragma sella.[16] Lateral extension to the cavernous sinuses and encasement of the cavernous carotids are not contraindications for a transsphenoidal approach.
Equipment
Endoscopic transsphenoidal hypophysectomy requires the full set of instruments for sinus surgery, along with a high-speed drill and hemostatic agents like cellulose or gelatin with thrombin. The microscopic approach requires a maxillofacial instrument set with a Hardy self-retaining bivalve speculum. An image-guiding navigation system is preferable in revision procedures, parasellar extensions, and cases with distorted anatomy.[17]
A micro-Doppler probe can be valuable for mapping the course of the ICA, particularly in cases involving parasellar tumor extension. This probe is especially recommended for pediatric patients with a significantly narrower intercarotid corridor than adults. However, study results indicate that this difference becomes less pronounced from age 9 onward. Intraoperative magnetic resonance imaging is significantly beneficial in pituitary surgery, as it can provide updated images showing the change in tumor volume, the dura, and the normal pituitary.
Personnel
Endoscopic surgery of pituitary tumors may require a 4-handed technique performed by 2 surgeons. The first surgeon holds the endoscope to provide an optimal view, while the second surgeon performs bimanual dissection. An ideal team would include a skilled endoscopic sinus surgeon who can provide fast and safe access to the sella and a neurosurgeon who understands the complexities of operating in this region. Although many neurosurgeons are capable of performing endonasal approaches independently, an otolaryngologist can significantly enhance the efficiency of the surgery and provide access when the area's architecture is compromised, as in cases involving nasal septum deviation, nasal polyp growth, and lack of sellar pneumatization.
In general, the otolaryngologist exposes the dura of the anterior aspect of the sella, which the neurosurgeon then opens before removing the tumor. The assistance of a skilled endoscopic sinus surgeon is often crucial for facilitating tumor removal in cases with extrasellar extension, such as when the tumor is adjacent to the cavernous sinus or has retrocarotid extension. Postoperative management of complications, including nasal synechiae, atrophic rhinitis, and cerebrospinal fluid leaks, often requires close collaboration between the neurosurgeon and the otolaryngologist.
Preparation
Clinical Workup
A comprehensive clinical history and a complete head and neck examination are mandatory before surgery. Ophthalmologic evaluation should be performed, including visual acuity testing, perimetry, and gaze restriction. Preoperative nasal endoscopy is crucial to exclude anatomical obstruction or sinonasal disease, which may need to be addressed before or during surgery. An endocrinologist's assessment of the preoperative pituitary function and hormone status is indispensable. After the evaluation, the interprofessional team should be aware of the following:
- The tumor's secretory status
- The patient's preoperative baseline pituitary function
- The patient's suitability for a medical alternative to surgery, such as cabergoline in prolactinoma
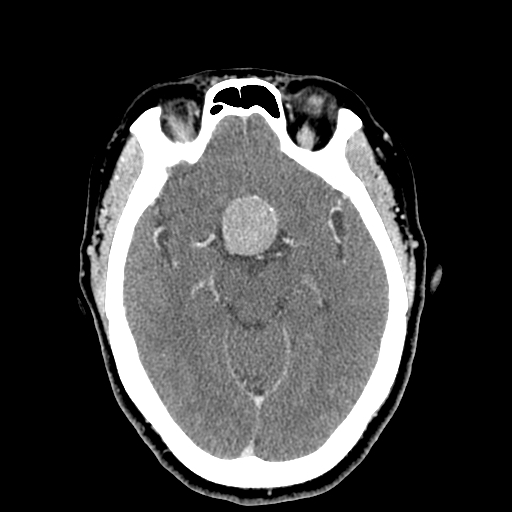
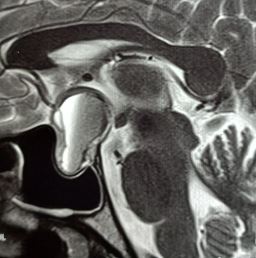
Levels of insulin-like growth factor 1, adrenocorticotropic hormone, prolactin, thyroid-stimulating hormone, follicle-stimulating hormone, luteinizing hormone, and testosterone are obtained.[18][19] Preoperative thyroid and cortisol levels are crucial, as hormonal replacement is essential for hypocortisolemia and hypothyroidism. CT or magnetic resonance imaging (MRI) is critical for any skull base surgery (see Image. Pituitary Macroadenoma on Head Computed Tomography). T1-weighted MRI with and without contrast is the best method for defining sellar pathology and planning the surgical approach in coronal and sagittal planes.[20] MRI is superior to CT due to its better soft-tissue discrimination and 3-dimensional ability to visualize the tumor, allowing for delineating vital structures such as the optic chiasm, intracavernous carotid arteries, optic nerves, and cavernous sinuses. MRI also demonstrates the degree of tumor extension, provides information on tumor texture, and helps differentiate solid tumor tissue from cystic fluid (see Image. Pituitary Macroadenoma Encasement of the Internal Carotid Artery). Additionally, MRI can provide information on dural involvement or invasion.
Microadenomas often present a diagnostic challenge. On T1-weighted images, these tumors are hypointense compared to the pituitary gland and enhance less than the normal gland. The tumor appearance on T2-weighted images varies. Microadenomas show early enhancement followed by early contrast washout, as opposed to the delayed enhancement of the pituitary gland. Macroadenomas are usually hypointense or isointense to normal brain tissue on T1-weighted images and variable on T2-weighted images. In postcontrast images, macroadenomas enhance well, making them easier to visualize.
Postcontrast spoiled gradient-recalled acquisition MRI combined with conventional T1-weighted spin-echo imaging has demonstrated superior diagnostic accuracy for evaluating corticotropinomas in pediatric and adult patients. Additionally, an 11C-methionine positron emission tomography scan can be a valuable tool for detecting recurrent Cushing disease in cases with inconclusive MRI findings.[21] Pituitary adenomas are isodense to brain parenchyma on CT without contrast. The tumor shows intense enhancement with contrast administration. CT scans may demonstrate the presence of bony erosion and expansion of the sella turcica. More importantly, CT scans are vital for planning the surgical approach. CT can identify septal deviations, sinonasal disease, nasal polyps, the extent of sinus pneumatization, ease of transnasal access to the sphenoid sinus, and anatomical variations such as Onodi cells, an aberrant carotid artery, or bony dehiscence of the skull base.
A history of a past nasal or septal surgery makes the preoperative planning and preparation more complex. Preoperative nasal endoscopy and radiological imaging are indicated if the previous surgery was related to a tumor or chronic disease, as in nasal polyposis. Evaluation of the septal mucosa with preoperative nasal endoscopy is necessary if the previous surgery involved septoplasty or septal flap harvesting. Scarred mucosa or septal perforation may prevent the harvest of a nasoseptal flap and warrant the use of alternate reconstruction methods after sellar surgery. Additionally, nasal endoscopy helps identify residual septal deviations or residual synechiae, which may be corrected before or concomitantly with the pituitary surgery.
Technique or Treatment
Operating Room Setup and Surgical Dynamics in Pituitary Surgery
An efficient operating room setup is crucial for the success of pituitary surgery. Endoscopic equipment is ergonomically arranged to ensure ease of access for the surgical team, while the anesthesiologist is typically positioned at the patient’s feet. Alternatively, the anesthesiologist may be stationed at the patient’s head based on specific procedural needs or operating room configuration. The surgical environment for endoscopic endonasal approaches is customized to the surgeon's preferences and experience, creating optimal conditions for safe and effective tumor resection.
Patient Positioning
In pituitary surgery, the patient is typically supine, with the trunk elevated by approximately 10° to facilitate venous drainage and optimize surgical access. The head positioning varies based on the surgical approach:
- Slightly turned toward the surgeon: Most common, used in approximately 45.5% of cases
- Neutral position: Standard for routine transsphenoidal approaches
- Extended by 10° to 20°: Provides better access to the anterior skull base
- Flexed by 10° to 20°: Facilitates targeting lower regions such as the clivus or craniovertebral junction
Head fixation
A head ring may support the head; a Mayfield-type fixation is optional even if the optical neuronavigation method is used.
Preoperative Considerations
Bladder catheterization is mandatory for accurate fluid balance management, especially due to the risk of diabetes insipidus, which can result from pituitary or hypothalamic disruption during surgery. The abdomen or thigh may be prepared as a potential graft donor site. In endoscopic cases, the navigation transmitter is attached to the patient’s forehead, and the patient is registered to the monitor.
Preoperative preparation
Preoperative preparation involves meticulous planning to reduce risks and ensure surgical precision. Routine nasal area disinfection is performed to minimize infection risks. For nasal decongestion and local anesthesia, cottonoids soaked in a decongestant and anesthetic solution are placed in the nasal cavity to reduce mucosal edema and provide local analgesia. A commonly used solution includes the following:
- 1 mg adrenaline: To minimize bleeding through vasoconstriction
- 5 ml of 20% diluted lidocaine: For local anesthetic effects
- 4 ml of saline: Acts as a diluent
The exact composition of the solution may vary depending on institutional protocols or surgeon preferences. This preparation enhances the surgeon’s view and reduces intraoperative bleeding, contributing to the procedure's safety and efficacy.
Endoscopic Equipment
Pituitary surgery is performed under direct endoscopic visualization, offering enhanced anatomical detail and surgical precision. The primary equipment includes a 0° and angled endoscopes. A 0° endoscope typically measures 18 cm long and 4 mm in diameter for standard cases. A smaller 2.7 mm diameter endoscope accommodates narrower nasal anatomy in pediatric patients. Angled endoscopes, 30° and 45°, help visualize lateral lesion aspects and concealed regions, particularly during tumor cavity inspection after the procedure. These tools provide superior access to areas beyond the direct line of sight of the 0° endoscope.
Instrumentation
Specialized surgical instruments are tailored for endoscopic pituitary surgery to optimize maneuverability. Straight instruments with slightly curved tips, including suction devices and bipolar cautery, are sometimes preferred over bayonet-shaped tools for their improved precision and ease of use within the confined surgical corridor. An external irrigation sheath on the endoscope facilitates continuous lens cleansing during surgery. This feature eliminates the need for repetitive removal and reinsertion of the endoscope through the nasal cavity, ensuring uninterrupted visualization and reducing procedure time.
Instrument positioning
For right-handed surgeons, the primary surgical instruments are introduced through the left nostril, while the endoscope is positioned in the right nostril. The reverse is true for left-handed surgeons. Initially, the endoscope is held in the nondominant hand, with the dominant hand operating key instruments such as dissectors, rongeurs, and debriders.
The widely adopted "4-hands/2-nostrils" technique enhances visualization and maneuverability from the sphenoid phase onward. The endoscope is placed in one nostril at the 12 o’clock position, while another instrument (eg, suction) is positioned in the same nostril at 6 o’clock. The surgeon and an assistant, often an otolaryngology colleague, neurosurgery resident, or another neurosurgeon, collaborate to operate the endoscope and instruments. Sometimes, the procedure may be performed independently by a single surgeon.
Alternatively, some surgeons utilize a fixed endoscope holder after the nasal phase. This option allows the primary surgeon to manage both the endoscope and surgical instruments independently or to continue with the assistance of a colleague. This setup can reduce physical strain and streamline instrument transitions.
The surgeon holding the endoscope must continuously adjust its position, angle, and rotation to maintain optimal visualization while accommodating instrument movements. However, proximity between the endoscope and instruments can lead to technical challenges. For example, "conflict of the sword" is a phenomenon where instruments clash, roll, or slip against each other due to limited space. Multiple tools occupying the same nasal corridor also compromise maneuverability and precision. Precise coordination and synchronization between surgical team members are essential to minimizing these conflicts. Regular practice and effective teamwork are pivotal in mastering these dynamics, ensuring smooth intraoperative flow, and reducing potential complications.
Nasal stage
The nasal stage uses the endoscopic endonasal, microscopic sublabial, or microscopic transnasal approaches. These parts are explained below. For the endoscopic endonasal transsphenoidal approach, bilateral nasal cavities are decongested, and anatomical obstructions, such as a deviated nasal septum or a concha bullosa, are corrected. A medial corridor between the nasal septum and middle turbinate is widened by lateralizing the latter. A pedicled septal flap, popularly known as the Hadad-Bassegastegay flap and supplied by the posterior nasoseptal artery, a branch of the sphenopalatine artery, may be raised if extended pituitary approaches are anticipated.[22][23] The sphenoid ostium is identified and widened to the lamina papyracea. The mucosa of the sphenoid sinus is elevated from medial to lateral, leaving 2 laterally based mucosal flaps for reconstituting the anterior wall of the pituitary fossa. Approximately 1 to 1.5 cm of the posterior edge of the septum is usually removed to facilitate instrumentation.
For the microscopic sublabial transsphenoidal approach, the gingival and nasal mucosa are infiltrated with a local anesthetic containing a vasoconstrictor. A sublabial incision is made from one canine fossa to the other, followed by subperiosteal dissection to expose the piriform aperture and the maxillary rostrum. The mucoperichondrium and mucoperiosteum from one side of the nasal septum and nasal floor are elevated, and the quadrangular cartilage is disarticulated from the vomer and perpendicular plate of the ethmoid. A handheld retractor is introduced, and the elevated mucosa is retracted laterally to expose the sphenoid rostrum. Once the sphenoid ostia are identified, a self-retaining Hardy bivalve speculum is introduced, allowing bimanual instrumentation. The operative microscope is then employed to improve magnification. The rostrum is resected with rongeurs to gain access to the sphenoid sinus.
The microscopic transnasal transsphenoidal approach is made through a single nostril to reach the sphenoid sinus. An incision is made at the posterior and inferior margins of the quadrangular cartilage. After submucosal dissection, the quadrangular cartilage is disarticulated at its posterior margin, and a bivalve speculum is introduced. Subsequent steps are similar to those of the sublabial approach. While this technique offers several advantages that include less soft tissue dissection, improved cosmesis, decreased postoperative morbidity, and the absence of upper jaw numbness, it can be difficult to perform in patients with a small nostril, requiring an alarotomy or external rhinoplasty incision, which may lead to scarring or nasal deformities.[24]
Sphenoid stage
The sphenoid sinus mucosa is stripped from the walls to prevent postoperative mucocele formation and reduce mucosal bleeding. Intersinus septations are carefully removed using a diamond drill or rongeur. The medial and lateral optic-carotid recesses, anterior genu of the carotid arteries, and optic nerve must be identified, as their location limits sella exposure. Using a diamond burr, the bone over the sella is thinned and then fractured. A Kerrison punch may remove the bone, exposing the underlying dura from one carotid artery canal to the opposite canal and from the planum sphenoidale to the clivus.
Sellar stage
The dura is opened in a U-shaped or cruciate fashion to expose the intrasellar contents. The normal pituitary gland appears yellow and firm, while the tumor may appear amorphous and white. An extracapsular resection of the tumor should be attempted. Superior cerebral spinal fluid (CSF) pulsations often facilitate tumor delivery through an intact arachnoid membrane. The angle between the arachnoid and the carotid artery should be determined. The diaphragma sella serves as the limit for superior and posterior dissection.
In cases of parasellar extension, 30° endoscopes are used to ensure maximal safe tumor resection. The pituitary fossa is filled with gel foam paste, and the dura is reapproximated to its preoperative location. The mucosal flaps are replaced over the dura and fixed in place with a surgical cellulose product and a layer of fibrin glue.
Indocyanine green fluorescence effectively distinguishes tumor tissue from normal tissue, with the former having a fourfold higher dye uptake. While 5-aminolevulinic acid can detect MRI-negative microadenomas, its results remain debated. OTL38 shows promise as a selective fluorescent agent for nonfunctioning adenomas.
Reconstruction Options
Over the years, numerous materials and techniques have been employed to address osteodural defects in pituitary surgery. These techniques involve intradural and extradural closure of the sella, with or without packing of the sphenoid sinus. Key objectives of reconstruction, as the final step of the endoscopic endonasal procedure, include the following:
- Obliteration of any dead space
- Isolation of the intradural compartment from the sinonasal tract
- Promotion of healing processes
- Prevention of intracranial pressure increases caused by postoperative strains such as coughing or bending
Preferred materials and techniques
Autologous graft materials (eg, fat, fascia, nasal mucosa) are often preferred over heterologous options (eg, dural substitutes) due to their availability, reduced risk of infection, and superior promotion of tissue regeneration. However, overpacking the sella cavity must be avoided to prevent compression of the optic apparatus. Packing of the nasal cavity is generally unnecessary except in cases of profuse mucosal bleeding, often observed in patients with uncontrolled hypertension or growth hormone/adrenocorticotropic hormone-secreting tumors, such as those in acromegaly or Cushing disease.
Tailored reconstruction approaches
Reconstruction techniques are customized based on the size and severity of the osteodural defect. In cases without CSF leakage, the sella is filled with an absorbable hemostatic sponge and fibrin glue. Packing of the sphenoid sinus is not mandatory. Despite the lack of evidence for intraoperative CSF leaks in cases of microadenomas, some surgeons prefer sellar reconstruction. Minor leaks may be sealed with bipolar coagulation of the arachnoid at the "de Tribolet point" on the sellar dura mater. Most surgeons perform reconstruction in microadenomas with small leaks, often involving sellar packing and closure with autologous materials.
Multilayer reconstruction combining intradural and extradural techniques is typically employed when reconstructing large osteodural defects, such as in cases following macroadenoma resection. A fat graft is widely adopted, molded to fit the surgical cavity and span the defect into the extradural space. Many surgeons use a nasoseptal flap to bolster the reconstruction. Some also employ a semisolid buttress for additional support, though synthetic materials are rarely used.
Best Practices and Considerations
Excessive packing must be avoided to prevent optic system compression. Thorough evaluation of sinonasal anatomy via preoperative imaging aids in identifying potential reconstruction challenges. Effective interprofessional collaboration ensures successful outcomes, particularly in complex osteodural defects or high-risk CSF leaks.
Multilayer closure and complete defect coverage are critical regardless of the reconstructive material. The edges of the defect must be denuded of mucosa to prevent mucocele formation and promote graft revascularization. Free tissue grafts are commonly used to reconstruct small- to medium-sized skull base defects.
Complications
Results from recent systematic reviews have shown conclusive evidence that endoscopic pituitary surgeries are safer than other microsurgical approaches, ie, transcranial or sublabial, in terms of established complication rates, though these approaches are equally efficacious.[25][26][27] The potential complications of transsphenoidal hypophysectomy are explained below.
Nonendocrine Complications
Nonendocrine complications are classified according to the stage at which they occur during surgery: intraoperative, immediate, and late postoperative. These complications can vary in severity but are generally infrequent, with many being preventable or manageable with proper surgical techniques and postoperative care.
Intraoperative complications
Complications can occur at every stage of the operation, though infrequently.[28] Adverse events that may arise during the nasal stage of the surgery include orbital injury, saddle nose deformity, anosmia, cribriform plate injury with CSF leak, and bleeding from injury to the sphenopalatine artery and its branches. Complications arising in the sphenoid sinus include sinusitis, carotid artery damage, and mucoceles. Problems that may be encountered during tumor removal include CSF leak, diabetes insipidus, hypopituitarism, meningitis, postoperative hematoma, carotid artery or optic nerve injury, vasospasm, ophthalmoplegia, subarachnoid hemorrhage, and tension pneumocephalus.
An ICA injury may be prevented intraoperatively, given adequate preoperative preparation.[29][30] First, risk factors must be identified to guide surgical planning. These risk factors include the following:
- Anatomical aberrations:
- Variations like a dehiscent ICA canal or thin bony coverings over the artery increase susceptibility.
- Lesions or tumors may dispense the ICA, especially in cases with sphenoidal septa attached to the blood vessel.
- Tumor characteristics:
- Larger, more invasive tumors (eg, chordomas and chondrosarcomas) elevate risks due to ICA displacement and the need for extensive dissection.
- Prior treatment:
- History of radiotherapy, previous surgeries, and use of drugs like bromocriptine lead to scarring, fibrosis, and increased vulnerability.
- Demographic factors:
Second, visualization tools must be optimized. Advanced intraoperative imaging, such as Doppler ultrasonography and navigation systems, should be used to locate the ICA before dural opening and guide during surgery. Adequate illumination and clear visualization must be maintained using a 2-nostril/4-hand approach with bimanual techniques.
Third, instruments that facilitate hemostasis must be prepared. Large-bore suction devices (12F or larger) should be ready for blood evacuation without obstructing the field. Hemostatic agents such as oxidized cellulose, thrombin-gelatin matrix, Avitene, and muscle patches should be readily available. Several measures can be taken to identify and control the ICA injury. First, upon recognizing an ICA injury, one surgeon should focus on directing suction to remove blood, while another works to achieve visualization for hemostasis. Excessive pressure on the vessel wall with suction tips must be avoided, as this step can worsen the injury.
Second, direct focal compression must be applied using cotton pledgets, iodoform gauze, or sponges to achieve temporary hemostasis. Newer hemostatic agents, such as Bioseal or Surgicel, may be employed for more robust bleeding control. Third, tamponade techniques may be used. Packing with crushed muscle patches or other tamponade materials may be necessary for arterial sealing. Complete exposure and packing of the nasal cavity must be ensured after initial bleeding control.
Hemostatic and supportive measures include adenosine administration, avoidance of coagulation on large vessels, and perfusion maintenance. Adenosine-induced transient hypotension may be considered to allow visualization during heavy bleeding. This technique, however, requires further validation regarding safety and dosage. Direct bipolar electrocoagulation should be avoided on major vessels but may be used for small arterial injuries caused by grinding or drilling. Normotension or slight hypertension must be maintained to ensure adequate collateral circulation to the brain.
Other supportive measures, including intravenous heparin administration, patient head elevation, and cervical carotid artery compression, may be considered. Heparin 5000 units may be administered intravenously to prevent thromboembolism resulting from intimal damage despite the bleeding. The patient’s head may be slightly elevated to reduce venous congestion and improve surgical field visualization. The cervical carotid artery may be digitally or surgically compressed to temporarily reduce bleeding, though this step is less effective due to retrograde flow.
A transition to endovascular management can be made. Intraoperative angiography must be immediately performed to assess for ongoing bleeding, pseudoaneurysms, carotid-cavernous fistulas, and arterial stenosis. Based on the site of injury and collateral circulation, endovascular repair may be performed by balloon occlusion or stent placement. Covered stents are preferred for pseudoaneurysm repair, while balloon occlusion may be used when vessel sacrifice is unavoidable. A surgical bypass may be needed if endovascular options fail or if preserving ICA patency is not possible.
Constant communication between the surgical and anesthesia teams must be maintained to manage hemodynamics and ensure resuscitation readiness. Specific tasks must be assigned to team members, such as controlling bleeding, managing suction, and preparing for endovascular transfer. Follow-up and postoperative considerations after addressing an intraoperative ICA injury include monitoring for complications and providing prophylactic care. Digital subtraction or CT angiography must be conducted postoperatively to detect delayed pseudoaneurysms. Patients must be monitored for neurological deficits like hemiparesis or aphasia, which may indicate intracranial complications. Antibiotics may be administered to prevent infections related to prolonged tamponade materials.
Immediate postoperative complications
The most common issues that may arise at this stage include CSF leaks, sinusitis, and meningitis.[33] CSF leaks occur in 6 out of 100 cases and are typically prevented by multilayer closure at the end of surgery. If a leak develops postoperatively, the patient is placed on bed rest, and a lumbar drain is inserted. Exploration and defect closure may be required if the leak persists beyond 24 hours.[34]
Worsening vision due to bleeding or manipulation and arterial hemorrhage are other immediate concerns. Thorough preoperative imaging is crucial to avoid injuries to the optic nerve and carotid artery. Anatomical variations like Onodi cells increase the risk of optic nerve damage. Suspected optic nerve injuries require tailored interventions, ranging from observation and high-dose intravenous steroids to optic nerve decompression, depending on the severity, timing, and progression of vision loss.
Late postoperative complications
Nasal congestion and mild nasal bleeding commonly occur within the first 1 to 2 weeks after surgery. Other complications that may develop in the late postoperative period include pain over the nasomaxillary region, nasal crusting, mucosal scarring, periorbital edema, and numbness of the upper incisors. Although transient, these issues are more frequent with the sublabial approach than with transnasal techniques due to extensive dissection and potential injury to the infraorbital nerve and its branches.
The use of nasal splints or packs often exacerbates postoperative morbidity. Mucosal damage may impair ciliary function, increasing the risk of sinusitis; some patients also report hyposmia. Long-term complications include nasal synechiae, which may be treated with radiofrequency ablation, nasal isolation, corticosteroids, and nasal irrigation, as well as septal perforation—which may require endoscopic repair using a mucoperiosteal pedicle flap of the nasal cavity floor if symptomatic. Atrophic rhinitis may need regular nasal endoscopic cleaning, soothing drops, and irrigation.[35]
Endocrine Complications
The most common endocrine complication in the early postoperative period involves abnormalities in antidiuretic hormone (ADH) secretion, including diabetes insipidus and syndrome of inappropriate antidiuretic hormone (SIADH) secretion. Diabetes insipidus is the most frequently reported endocrine complication following sellar surgery, with its postoperative incidence ranging from 5% to 35%.[36][37][38][39] Postoperative diabetes insipidus often follows a triphasic pattern: an initial phase of polyuria and polydipsia within the first 48 hours, lasting a few days, followed by a phase of antidiuresis with hyponatremia, typically occurring around 1 week postsurgery. This occurrence is succeeded by a polyuric phase, which may result in permanent diabetes insipidus.
In most cases, diabetes insipidus resolves spontaneously within 24 hours without specific therapy. Surgical treatment of lesions involving the pituitary stalk or areas above the median eminence is more likely to result in permanent diabetes insipidus. Mild diabetes insipidus, characterized by urine output of less than 4 to 6 liters per day and serum sodium below 150 mEq/L, is managed conservatively with oral fluid replacement. In unresolved cases, management escalates from fluid replacement with 5% dextrose to synthetic ADH analogs like desmopressin. Intranasal desmopressin is the treatment of choice for chronic diabetes insipidus.
SIADH is the most common cause of hyponatremia after pituitary surgery, occurring in 9% to 30% of patients undergoing a transsphenoidal approach.[40] Most patients with SIADH are asymptomatic, with the diagnosis identified through a serum electrolyte panel performed on postoperative day 7. Management of postoperative SIADH depends on the degree of hyponatremia, the presence of symptoms, and the patient's comorbid conditions.
Fluid restriction and, occasionally, salt administration form the primary treatment. Most patients are treated in outpatient settings, with serial sodium level measurements and fluid output monitoring. Sodium level correction may require up to 5 days. Symptomatic individuals are hospitalized and treated with intravenous 3% saline. Hyponatremia is the most frequent cause of unplanned readmission following pituitary surgery due to SIADH, typically presenting after postoperative day 5.[41]
The acute management of postoperative hypopituitarism involves appropriate glucocorticoid replenishment and fluid and electrolyte replacement. Glucocorticoid doses are adjusted during the perioperative period based on preoperative hormone and stress levels and gradually tapered to preoperative doses. The key principle is to ensure adequate glucocorticoid coverage before surgery and for 24 hours afterward.
Postoperative management
During the immediate postoperative period, patients are monitored in the intensive care unit for neurological deterioration, epistaxis, visual dysfunction, diabetes insipidus, and hypotension caused by acute hypocortisolism. Desmopressin or steroid replacement is continued postoperatively if initiated preoperatively, with strict electrolyte monitoring. For patients with normal preoperative pituitary function, serum cortisol and prolactin levels are measured in the morning following the procedure.
For secretory tumors, nonstimulatory hormone levels, such as adrenocorticotropic hormone, prolactin, or insulin-like growth factor 1, are obtained on postoperative days 1 and 2. Serum sodium and urine output are monitored serially for 48 hours. Steroid replacement therapy is started if cortisol levels are low. For suspected new-onset diabetes insipidus, oral fluid management with water may be sufficient if the patient is awake and stable. If desmopressin is initiated, close monitoring by an endocrinologist is necessary. Patients are typically discharged by postoperative day 2 or 3.
The need for nasal packs depends on the type of reconstructive technique performed and the surgeon's preference. Nasal packs are removed on postoperative day 1. Septal splints are typically required in traditional sublabial-transeptal-transsphenoidal approaches and removed on postoperative days 5 to 7. The first follow-up visit should occur 1 week after the procedure, during which postoperative day 7 serum sodium levels are reviewed to rule out occult hyponatremia. Serial nasal endoscopies are performed for debridement and assessment of healing. The extent of nasal crusting and maintenance of nasal hygiene with irrigation determines the frequency of follow-up visits. Routine early postoperative imaging is generally not performed for most patients. MRI is typically obtained 3 months after the procedure. However, it may be taken within 48 hours to rule out a remnant in a functioning pituitary adenoma.
Clinical Significance
Transsphenoidal hypophysectomy is an effective surgical technique for removing pituitary and similar intrasellar tumors with minimal morbidity and a short hospital stay. This procedure improves endocrine function and decreases the pressure on the pituitary gland and adjacent structures, such as the optic nerves.
Enhancing Healthcare Team Outcomes
Pituitary tumors are complex conditions that may potentially impact multiple endocrine systems and vision. Optimal management of these neoplasms warrants an interprofessional team approach involving endocrinologists, ophthalmologists, neurosurgeons, and otolaryngologists. The qualified nursing staff prepares patients for procedures, assists the surgeons during their surgery, and monitors them afterward.
Pituitary tumors are often incidentally detected on imaging studies by radiologists and referred to neurosurgeons for surgical management. Nonfunctioning tumors are monitored with serial imaging to identify an increase in the tumor size or complications due to mass effects. Functioning tumors, such as prolactinoma, require rigorous medical management by an endocrinologist; medically refractory neoplasms are referred to neurosurgeons. Further, ophthalmologists play a vital role in the early recognition of visual abnormalities in the preoperative and postoperative periods.
The surgical team should include a neurosurgeon, an otolaryngologist, neuro-anesthesiologists, and nurses specialized in handling such complex cases. The otolaryngologist provides access to the tumor via the nasal cavities, and the neurosurgeon resects the tumor. Postoperative management is crucial, and close coordination between critical care specialists, nurses, and endocrinologists is required. Hence, expert interprofessional team performance is vital to optimizing patient care.
Media
(Click Image to Enlarge)
(Click Image to Enlarge)
(Click Image to Enlarge)
(Click Image to Enlarge)
References
Cucu AI, Costea CF, Perciaccante A, Donell ST, Bianucci R. The Evolution of Pituitary Gland Surgery from the Ancients to the Millennials. World neurosurgery. 2023 Dec:180():52-65. doi: 10.1016/j.wneu.2023.09.004. Epub 2023 Sep 7 [PubMed PMID: 37683915]
Schmidt RF, Choudhry OJ, Takkellapati R, Eloy JA, Couldwell WT, Liu JK. Hermann Schloffer and the origin of transsphenoidal pituitary surgery. Neurosurgical focus. 2012 Aug:33(2):E5. doi: 10.3171/2012.5.FOCUS12129. Epub [PubMed PMID: 22853836]
Jankowski R, Auque J, Simon C, Marchal JC, Hepner H, Wayoff M. Endoscopic pituitary tumor surgery. The Laryngoscope. 1992 Feb:102(2):198-202 [PubMed PMID: 1738293]
Level 3 (low-level) evidenceHar-El G. Endoscopic transnasal transsphenoidal pituitary surgery--comparison with the traditional sublabial transseptal approach. Otolaryngologic clinics of North America. 2005 Aug:38(4):723-35 [PubMed PMID: 16005728]
Li K, Zhang J, Wang XS, Ye X, Zhao YL. A systematic review of effects and complications after transsphenoidal pituitary surgery: endoscopic versus microscopic approach. Minimally invasive therapy & allied technologies : MITAT : official journal of the Society for Minimally Invasive Therapy. 2020 Dec:29(6):317-325. doi: 10.1080/13645706.2019.1660369. Epub 2019 Sep 8 [PubMed PMID: 31495241]
Level 1 (high-level) evidencePrevedello DM, Doglietto F, Jane JA Jr, Jagannathan J, Han J, Laws ER Jr. History of endoscopic skull base surgery: its evolution and current reality. Journal of neurosurgery. 2007 Jul:107(1):206-13 [PubMed PMID: 17639897]
Harvey RJ, Shelton W, Timperley D, Debnath NI, Byrd K, Buchmann L, Gallagher RM, Orlandi RR, Sacks R, Schlosser RJ. Using fixed anatomical landmarks in endoscopic skull base surgery. American journal of rhinology & allergy. 2010 Jul-Aug:24(4):301-5. doi: 10.2500/ajra.2010.24.3473. Epub [PubMed PMID: 20819470]
Anusha B, Baharudin A, Philip R, Harvinder S, Shaffie BM. Anatomical variations of the sphenoid sinus and its adjacent structures: a review of existing literature. Surgical and radiologic anatomy : SRA. 2014 Jul:36(5):419-27. doi: 10.1007/s00276-013-1214-1. Epub 2013 Oct 22 [PubMed PMID: 24146215]
Štoković N, Trkulja V, Dumić-Čule I, Čuković-Bagić I, Lauc T, Vukičević S, Grgurević L. Sphenoid sinus types, dimensions and relationship with surrounding structures. Annals of anatomy = Anatomischer Anzeiger : official organ of the Anatomische Gesellschaft. 2016 Jan:203():69-76. doi: 10.1016/j.aanat.2015.02.013. Epub 2015 Mar 20 [PubMed PMID: 25843780]
Ouaknine GE, Hardy J. Microsurgical anatomy of the pituitary gland and the sellar region. 2. The bony structures. The American surgeon. 1987 May:53(5):291-7 [PubMed PMID: 3579041]
Banna M, Olutola PS. Patterns of pneumatization and septation of the sphenoidal sinus. Journal of the Canadian Association of Radiologists. 1983 Dec:34(4):291-3 [PubMed PMID: 6668290]
Elwany S, Yacout YM, Talaat M, El-Nahass M, Gunied A, Talaat M. Surgical anatomy of the sphenoid sinus. The Journal of laryngology and otology. 1983 Mar:97(3):227-41 [PubMed PMID: 6833847]
Moeller CW, Welch KC. Prevention and management of complications in sphenoidotomy. Otolaryngologic clinics of North America. 2010 Aug:43(4):839-54. doi: 10.1016/j.otc.2010.04.009. Epub 2010 May 26 [PubMed PMID: 20599088]
Valentine R, Padhye V, Wormald PJ. Management of arterial injury during endoscopic sinus and skull base surgery. Current opinion in otolaryngology & head and neck surgery. 2016 Apr:24(2):170-4. doi: 10.1097/MOO.0000000000000239. Epub [PubMed PMID: 26959844]
Level 3 (low-level) evidenceChavez-Herrera VR, Desai R, Gel G, Nilchian P, Schwartz TH. Endonasal endoscopic surgery for pituitary adenomas. Clinical neurology and neurosurgery. 2024 Feb:237():108172. doi: 10.1016/j.clineuro.2024.108172. Epub 2024 Feb 10 [PubMed PMID: 38359520]
Bove I, Solari D, Bruneau M, Berhouma M, Jouanneau E, Cornelius JF, Messerer M, Daniel RT, Froelich S, Mazzatenta D, Meling T, Paraskevopoulos D, Roche PH, Schoeder HWS, Zazpe I, Visocchi M, Kasper E, Tatagiba M, Cavallo LM. Endoscopic endonasal pituitary surgery: How we do it. Consensus statement on behalf of the EANS skull base section. Brain & spine. 2023:3():102687. doi: 10.1016/j.bas.2023.102687. Epub 2023 Oct 8 [PubMed PMID: 38021006]
Level 3 (low-level) evidenceJaver AR, Marglani O, Lee A, Matishak M, Genoway KA. Image-guided endoscopic transsphenoidal removal of pituitary tumours. Journal of otolaryngology - head & neck surgery = Le Journal d'oto-rhino-laryngologie et de chirurgie cervico-faciale. 2008 Aug:37(4):474-80 [PubMed PMID: 19128579]
Level 2 (mid-level) evidenceBaussart B, Gaillard S. Pituitary surgery for Cushing's disease. Acta neurochirurgica. 2021 Nov:163(11):3155-3159. doi: 10.1007/s00701-021-04995-w. Epub 2021 Sep 14 [PubMed PMID: 34519880]
Papaioannou C, Druce M. Preoperative medical treatments and surgical approaches for acromegaly: A systematic review. Clinical endocrinology. 2023 Jan:98(1):14-31. doi: 10.1111/cen.14790. Epub 2022 Jun 30 [PubMed PMID: 35726150]
Level 1 (high-level) evidenceBaban MIA, Omer SN, AlZuhairy AKA, Mahmoud AA. The role of preoperative MRI in endoscopic transnasal transsphenoidal hypophysectomy of pituitary adenoma. European archives of oto-rhino-laryngology : official journal of the European Federation of Oto-Rhino-Laryngological Societies (EUFOS) : affiliated with the German Society for Oto-Rhino-Laryngology - Head and Neck Surgery. 2024 Apr:281(4):1961-1969. doi: 10.1007/s00405-024-08472-9. Epub 2024 Feb 6 [PubMed PMID: 38319368]
Ishida A, Kaneko K, Minamimoto R, Hotta M, Inoshita N, Takano K, Yamada S. Clinical decision-making based on 11C-methionine PET in recurrent Cushing's disease with equivocal MRI findings. Journal of neurosurgery. 2023 Dec 1:139(6):1671-1680. doi: 10.3171/2023.5.JNS23179. Epub 2023 Jun 30 [PubMed PMID: 37410630]
Hoerter JE, Kshirsagar RS. Nasoseptal Flap. StatPearls. 2025 Jan:(): [PubMed PMID: 35015408]
Hadad G, Bassagasteguy L, Carrau RL, Mataza JC, Kassam A, Snyderman CH, Mintz A. A novel reconstructive technique after endoscopic expanded endonasal approaches: vascular pedicle nasoseptal flap. The Laryngoscope. 2006 Oct:116(10):1882-6 [PubMed PMID: 17003708]
Level 2 (mid-level) evidenceYamada S. Sublabial transsphenoidal pituitary surgery: a new modified technique for a less-invasive sublabial approach. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie. 2002:56 Suppl 1():165s-170s [PubMed PMID: 12487275]
Tabaee A, Anand VK, Barrón Y, Hiltzik DH, Brown SM, Kacker A, Mazumdar M, Schwartz TH. Endoscopic pituitary surgery: a systematic review and meta-analysis. Journal of neurosurgery. 2009 Sep:111(3):545-54. doi: 10.3171/2007.12.17635. Epub [PubMed PMID: 19199461]
Level 1 (high-level) evidenceBerker M, Hazer DB, Yücel T, Gürlek A, Cila A, Aldur M, Onerci M. Complications of endoscopic surgery of the pituitary adenomas: analysis of 570 patients and review of the literature. Pituitary. 2012 Sep:15(3):288-300. doi: 10.1007/s11102-011-0368-2. Epub [PubMed PMID: 22161543]
Level 2 (mid-level) evidenceDai W, Zhuang Z, Ling H, Yang Y, Hang C. Systematic review and network meta-analysis assess the comparative efficacy and safety of transsphenoidal surgery for pituitary tumor. Neurosurgical review. 2021 Feb:44(1):515-527. doi: 10.1007/s10143-020-01240-3. Epub 2020 Feb 8 [PubMed PMID: 32036504]
Level 2 (mid-level) evidenceVignolles-Jeong J, Kreatsoulas D, Godil S, Otto B, Carrau R, Prevedello D. Complications in Endoscopic Pituitary Surgery. Otolaryngologic clinics of North America. 2022 Apr:55(2):431-448. doi: 10.1016/j.otc.2021.12.011. Epub [PubMed PMID: 35365316]
Ma X, Zhang Y, Yang ZJ, Bo W, Wang XC, Zhang C, Liu PN, Bi ZY. Internal carotid artery injury during endoscopic transsphenoidal pituitary surgery: risk factors, management. Neuro-Chirurgie. 2024 Jan:70(1):101515. doi: 10.1016/j.neuchi.2023.101515. Epub 2023 Dec 4 [PubMed PMID: 38052154]
AlQahtani A, Castelnuovo P, Nicolai P, Prevedello DM, Locatelli D, Carrau RL. Injury of the Internal Carotid Artery During Endoscopic Skull Base Surgery: Prevention and Management Protocol. Otolaryngologic clinics of North America. 2016 Feb:49(1):237-52. doi: 10.1016/j.otc.2015.09.009. Epub [PubMed PMID: 26614841]
Sharma RK, Irace AL, Overdevest JB, Gudis DA. Carotid artery injury in endoscopic endonasal surgery: Risk factors, prevention, and management. World journal of otorhinolaryngology - head and neck surgery. 2022 Mar:8(1):54-60. doi: 10.1002/wjo2.7. Epub 2022 Mar 22 [PubMed PMID: 35619937]
Schaberg MR, Anand VK, Schwartz TH. 10 pearls for safe endoscopic skull base surgery. Otolaryngologic clinics of North America. 2010 Aug:43(4):945-54. doi: 10.1016/j.otc.2010.04.022. Epub [PubMed PMID: 20599097]
Riesgo P, Mariño P, Platero A, Tarazona FJ, Fajardo C, Llácer JL, Rovira V, Rodríguez R, Flor-Goikoetxea A, Piquer J. Postoperative CSF leakages after transsphenoidal surgery for pituitary adenomas: Analysis of a series of 302 surgical procedures. Neurocirugia (English Edition). 2019 Sep-Oct:30(5):215-221. doi: 10.1016/j.neucir.2019.03.003. Epub 2019 May 23 [PubMed PMID: 31130305]
do Amaral LC, Reis BL, Ribeiro-Oliveira A Jr, da Silva Santos TM, Giannetti AV. Comparative study of complications after primary and revision transsphenoidal endoscopic surgeries. Neurosurgical review. 2021 Jun:44(3):1687-1702. doi: 10.1007/s10143-020-01360-w. Epub 2020 Aug 11 [PubMed PMID: 32783077]
Level 2 (mid-level) evidenceCheng Y, Xue F, Wang TY, Ji JF, Chen W, Wang ZY, Xu L, Hang CH, Liu XF. Analyses and treatments of postoperative nasal complications after endonasal transsphenoidal resection of pituitary neoplasms. Medicine. 2017 Apr:96(15):e6614. doi: 10.1097/MD.0000000000006614. Epub [PubMed PMID: 28403108]
Wang S, Li D, Ni M, Jia W, Zhang Q, He J, Jia G. Clinical Predictors of Diabetes Insipidus After Transcranial Surgery for Pituitary Adenoma. World neurosurgery. 2017 May:101():1-10. doi: 10.1016/j.wneu.2017.01.075. Epub 2017 Jan 31 [PubMed PMID: 28153615]
Saldarriaga C, Lyssikatos C, Belyavskaya E, Keil M, Chittiboina P, Sinaii N, Stratakis CA, Lodish M. Postoperative Diabetes Insipidus and Hyponatremia in Children after Transsphenoidal Surgery for Adrenocorticotropin Hormone and Growth Hormone Secreting Adenomas. The Journal of pediatrics. 2018 Apr:195():169-174.e1. doi: 10.1016/j.jpeds.2017.11.042. Epub 2018 Feb 1 [PubMed PMID: 29395172]
Faltado AL, Macalalad-Josue AA, Li RJS, Quisumbing JPM, Yu MGY, Jimeno CA. Factors Associated with Postoperative Diabetes Insipidus after Pituitary Surgery. Endocrinology and metabolism (Seoul, Korea). 2017 Dec:32(4):426-433. doi: 10.3803/EnM.2017.32.4.426. Epub 2017 Nov 21 [PubMed PMID: 29199401]
Seltzer J, Wedemeyer MA, Bonney PA, Carmichael JD, Weiss M, Zada G. Outcomes following transsphenoidal surgical management of incidental pituitary adenomas: a series of 52 patients over a 17-year period. Journal of neurosurgery. 2019 May 1:130(5):1584-1592. doi: 10.3171/2017.11.JNS171485. Epub 2018 Jun 1 [PubMed PMID: 29999467]
Winograd D, Staggers KA, Sebastian S, Takashima M, Yoshor D, Samson SL. An Effective and Practical Fluid Restriction Protocol to Decrease the Risk of Hyponatremia and Readmissions After Transsphenoidal Surgery. Neurosurgery. 2020 Sep 15:87(4):761-769. doi: 10.1093/neuros/nyz555. Epub [PubMed PMID: 31993647]
Bohl MA, Ahmad S, Jahnke H, Shepherd D, Knecht L, White WL, Little AS. Delayed Hyponatremia Is the Most Common Cause of 30-Day Unplanned Readmission After Transsphenoidal Surgery for Pituitary Tumors. Neurosurgery. 2016 Jan:78(1):84-90. doi: 10.1227/NEU.0000000000001003. Epub [PubMed PMID: 26348011]