Introduction
Neurotrophic keratitis is a degenerative corneal disease caused by impaired sensory innervation. The condition is characterized by decreased or absent corneal sensation, leading to epithelial breakdown, delayed healing, and, ultimately, corneal ulceration, melting, and perforation.[1] Initially described as neuroparalytic keratitis, this corneal pathology was first demonstrated experimentally by Magendie in 1824.
Neurotrophic keratitis is a rare but vision-threatening disease that progresses due to corneal nerve dysfunction.[2] The corneal nerves play a crucial role in maintaining ocular surface health by regulating epithelial cell proliferation, tear film stability, and immune responses. When these nerves are compromised by systemic or local conditions, the cornea loses its protective mechanisms, making it vulnerable to ulceration, infection, and mechanical trauma.
The prevalence of neurotrophic keratitis is relatively low, affecting approximately 5 per 10,000 individuals, but it is often underdiagnosed due to its subtle clinical presentation. The disease follows the 3-stage Mackie classification. Stage 1 is characterized by corneal epithelial irregularities and punctate keratopathy. Stage 2 presents with a persistent epithelial defect (PED) and stromal involvement. Stage 3 encompasses corneal ulceration with stromal melting and possible perforation.
Progression is often insidious, with patients rarely reporting pain due to diminished corneal sensation. However, some individuals may experience blurred vision, mild redness, or recurrent ocular surface inflammation. Early recognition and prompt intervention are essential to prevent irreversible damage and vision loss.[3]
Neurotrophic keratitis results from damage to the sensory (afferent) fibers of the trigeminal nerve, the 5th cranial nerve. These fibers maintain corneal sensation and epithelial integrity. Several systemic and ocular conditions can lead to corneal nerve dysfunction, including herpetic infection from herpes simplex (HSV) or varicella-zoster (VZV) virus, diabetes mellitus, postsurgical nerve damage from procedures such as refractive surgery, corneal transplantation, or trigeminal neuralgia surgery, as well as chemical burns, chronic topical drug use, contact lens wear, and autoimmune diseases such as Sjögren syndrome.[4]
Central nervous system disorders affecting the trigeminal ganglion or brainstem can also disrupt corneal innervation, contributing to neurotrophic keratitis. The corneal epithelium relies on trophic factors from corneal nerves, including substance P, nerve growth factor (NGF), and insulin-like growth factor 1 (IGF-1), to maintain ocular surface health and prevent epithelial breakdown. Loss of nerve function slows epithelial turnover, disrupts tear film dynamics, and impairs corneal healing, producing PEDs, ulceration, and corneal thinning. The absence of sensory feedback further reduces reflex tearing and blinking, exacerbating ocular surface desiccation and inflammation.[5]
One of the most challenging aspects of diagnosing neurotrophic keratitis is the lack of significant symptoms despite ongoing corneal damage. Unlike other corneal diseases, where pain and discomfort prompt early medical attention, neurotrophic keratitis often remains undetected until severe complications arise. Common clinical signs include punctate epithelial erosions, PEDs, corneal ulceration, and thinning. In advanced cases, stromal necrosis and perforation may develop, necessitating urgent surgical intervention.
A comprehensive clinical evaluation should include corneal sensation testing with a cotton wisp, a Cochet-Bonnet esthesiometer, or dental floss. A slit-lamp examination can assess epithelial irregularities, tear meniscus height reduction, punctate keratopathy, and stromal opacities. Fluorescein and Rose Bengal staining help detect epithelial damage and conjunctival involvement, while tear film analysis using the Schirmer test, tear breakup time (TBUT), and ocular surface inflammation markers further support diagnosis. Corneal imaging with anterior segment optical coherence tomography (AS-OCT) or in vivo confocal microscopy (IVCM) provides detailed visualization of nerve density, epithelial integrity, and subbasal nerve plexus abnormalities.[6]
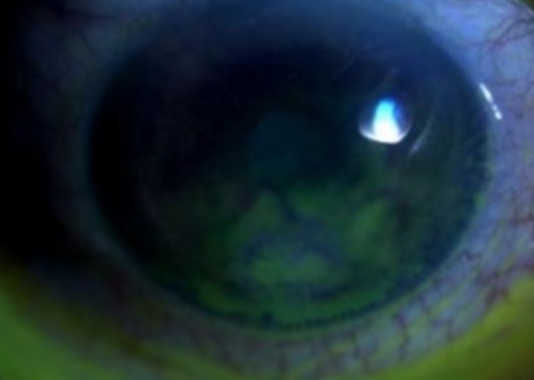
The Mackie classification divides neurotrophic keratitis into 3 progressive stages. Stage 1 presents with early epithelial irregularities, corneal haze, reduced tear film stability, and punctate epithelial erosions. Stage 2 involves a PED with underlying stromal involvement, often leading to stromal thinning and edema (see Image. Stage 2 Neurotrophic Keratitis). Stage 3 represents the most severe form, characterized by corneal ulceration, stromal melting, and potential perforation, which significantly increases the risk of infectious keratitis and permanent vision loss. Early recognition and classification are crucial for guiding treatment and preventing disease progression.[7]
Management of neurotrophic keratitis depends on disease severity and focuses on promoting corneal healing, restoring nerve function, and preventing complications. Treatment is multimodal, incorporating topical, surgical, and regenerative approaches. Conservative medical therapy serves as the first-line intervention, including lubrication with nonpreserved artificial tears, hydrogel inserts, and autologous serum tears to maintain ocular surface hydration. Discontinuing toxic medications, such as preserved eye drops, glaucoma drugs, and long-term corticosteroids, is essential to prevent further epithelial toxicity. Therapeutic contact lenses, including bandage, scleral, and amniotic membrane lenses, can support corneal healing in patients with PEDs.[8]
For more advanced cases, biological and pharmacologic treatments have transformed neurotrophic keratitis management. Recombinant NGF (cenegermin-bkbj, Oxervate™) stimulates corneal nerve regeneration and enhances epithelial healing. Platelet-rich plasma (PRP) and autologous serum drops, which contain epithelial growth factors and anti-inflammatory cytokines, support ocular surface repair and reduce inflammation.
Surgical and interventional approaches are reserved for severe or refractory cases.[9] Tarsorrhaphy, a partial or complete closure of the eyelids, reduces corneal exposure and promotes healing. Amniotic membrane transplantation (AMT) provides growth factors and anti-inflammatory properties to facilitate epithelial recovery. Corneal neurotization, a novel technique involving the transplantation of sensory nerves, such as the supraorbital or infraorbital nerve, has shown promise in restoring corneal sensation in severe cases. For patients with corneal perforation, conjunctival flaps and keratoplasty procedures, including lamellar or penetrating keratoplasty (PKP), may be necessary.[10]
Managing neurotrophic keratitis requires an interprofessional approach involving ophthalmologists, cornea specialists, neurologists, and optometrists. Early referral to an ophthalmologist is essential, as delayed diagnosis can lead to irreversible corneal damage.
The future of neurotrophic keratitis treatment lies in regenerative medicine, neuroprotection, and gene therapy. Emerging therapies, including mesenchymal stem cell therapy, gene-based neurotrophic factor delivery, and bioengineered corneal substitutes, offer hope for improved outcomes. Advances in biologic treatments, regenerative medicine, and corneal neurotization are transforming the treatment landscape, enhancing both visual prognosis and quality of life. Increased awareness, research, and accessibility of novel therapies will be crucial in optimizing long-term patient outcomes.[11]
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
Damage or dysfunction at any level of the trigeminal nerve, from the trigeminal nucleus to the corneal nerve endings, can lead to neurotrophic keratitis. The most common causes include herpetic keratitis, chemical burns, prolonged contact lens wear, corneal surgery, ablative procedures for trigeminal neuralgia, and surgical interventions for jaw fracture reduction. Less frequently, space-occupying intracranial lesions such as schwannomas, meningiomas, and aneurysms may compress the trigeminal nerve, impairing corneal sensitivity.
Systemic conditions that compromise trigeminal function, including diabetes, multiple sclerosis, and leprosy, also contribute to the development of neurotrophic keratitis. In rare cases, children may present with neurotrophic keratitis due to congenital syndromes, such as Riley-Day, Goldenhar-Gorlin, and Möbius syndromes, as well as familial corneal hypesthesia and congenital insensitivity to pain with anhidrosis.[12]
Neurotrophic keratitis results from impaired corneal innervation, disrupting corneal sensation, epithelial integrity, and ocular surface homeostasis. The corneal epithelium depends on trophic support from the trigeminal nerve, and loss of this innervation compromises wound healing and increases the risk of epithelial breakdown, ulceration, and stromal damage. The underlying etiology varies widely, encompassing infectious, inflammatory, traumatic, iatrogenic, metabolic, and neurological conditions. Recognizing these causes is essential for timely diagnosis and intervention to prevent disease progression.[13]
Infection
Several infections can directly damage the corneal nerves, leading to the development of neurotrophic keratitis. Among these conditions, herpetic keratitis caused by HSV or VZV is one of the most common etiologies (see Image. Ocular and Perioral Involvement in Herpes Simplex Virus Infection). HSV invades the trigeminal ganglion, causing permanent nerve damage and reducing corneal sensation. Similarly, VZV, the causative agent of herpes zoster ophthalmicus (HZO), affects the trigeminal nerve, leading to corneal anesthesia and epithelial dysfunction. Recurrent episodes of herpetic keratitis, even with antiviral treatment, result in progressive corneal sensory loss, increasing the risk of epithelial breakdown and ulceration.[14]
Leprosy, caused by Mycobacterium leprae, is another significant infectious cause of corneal anesthesia, particularly in endemic regions. The bacterium infiltrates the trigeminal nerve, leading to chronic inflammation, nerve atrophy, and eventual loss of corneal sensation. Patients with leprosy face a heightened risk of developing chronic corneal ulcers due to unnoticed trauma and impaired corneal healing.[15]
Traumatic and Iatrogenic Injury
Various surgical and traumatic events can directly or indirectly injure the corneal nerves, resulting in neurotrophic keratitis. Corneal surgery is a common cause, as procedures such as laser-assisted in situ keratomileusis (LASIK), photorefractive keratectomy (PRK), and small incision lenticule extraction (SMILE) disrupt corneal nerve integrity through flap creation or epithelial ablation, potentially leading to temporary or permanent neurotrophic changes. PKP (corneal transplantation) severs corneal nerve fibers during grafting, reducing sensitivity in the transplanted cornea. Patients undergoing keratoprosthesis (KPro), such as the Boston KPro, frequently develop corneal anesthesia, increasing the risk of recurrent epithelial defects and ulceration.[16]
Ocular trauma can also contribute to neurotrophic keratitis, particularly chemical burns from acid or alkali exposure, which can destroy corneal nerves and lead to irreversible neurotrophic damage. Thermal and mechanical injuries affecting the corneal epithelium and stroma may disrupt nerve function, impairing corneal healing. Additionally, penetrating injuries involving the trigeminal nerve can damage the ophthalmic division (cranial nerve V1), significantly reducing corneal sensation.[17]
Neurosurgical procedures involving the trigeminal nerve pose another risk. Surgeries for trigeminal neuralgia, including microvascular decompression and γ knife surgery, can impair corneal sensation by damaging the trigeminal nerve. Resection of cranial nerve tumors, such as acoustic neuromas, skull base tumors, or meningiomas, may inadvertently injure the trigeminal nerve, resulting in neurotrophic keratitis. Orbital decompression surgeries performed for thyroid eye disease can also lead to unintended corneal nerve injury, further predisposing patients to neurotrophic keratitis.[18]
Neurological Pathology
Conditions affecting the central and peripheral nervous systems can impair corneal innervation, contributing to neurotrophic keratitis. Stroke and other brainstem lesions can disrupt the trigeminal nerve nucleus, leading to corneal hypoesthesia or anesthesia. Ischemic or hemorrhagic strokes affecting the pons may impair corneal sensation, while brainstem tumors and demyelinating diseases, such as multiple sclerosis, can interfere with trigeminal nerve pathways, further compromising corneal innervation and sensation.[19]
Diabetic neuropathy is a significant risk factor for neurotrophic keratitis due to progressive trigeminal nerve dysfunction. Chronic diabetes mellitus leads to decreased corneal sensation, reduced tear secretion, and impaired corneal healing, increasing the risk of PEDs and ulceration.[20]
Congenital and genetic disorders that affect sensory nerve development can also lead to early-onset neurotrophic keratitis. Riley-Day syndrome (familial dysautonomia) and congenital insensitivity to pain (CIP) impair corneal nerve function, resulting in corneal anesthesia. Moebius syndrome, a congenital disorder involving multiple cranial nerves, including the trigeminal nerve, can similarly cause severe corneal hypoesthesia.[21]
Chronic Topical Medication and Ocular Surface Disease
Prolonged use of certain topical ophthalmic medications and extended contact lens wear can contribute to epithelial toxicity and corneal nerve damage, increasing the risk of neurotrophic keratitis.[22] Chronic use of preserved glaucoma medications, especially those containing benzalkonium chloride (BAK), is known to cause corneal epithelial and nerve toxicity. Long-term exposure to topical anesthetics, such as in proparacaine abuse cases, can result in severe neurotrophic corneal damage.[23]
Extended contact lens wear reduces corneal oxygenation, leading to progressive nerve degeneration and diminished corneal sensation. Patients who use extended-wear contact lenses face an increased risk of neurotrophic keratitis due to chronic hypoxia-induced nerve dysfunction.[24]
Autoimmune and Inflammatory Disorders
Certain immune-mediated diseases can lead to chronic ocular surface inflammation, resulting in progressive corneal nerve damage.[25] Sjögren syndrome, a chronic autoimmune disease affecting the lacrimal glands and ocular surface, causes severe dry eye disease (DED) and progressive corneal hypoesthesia. Persistent inflammation damages corneal nerves, contributing to neurotrophic keratitis.[26]
Severe mucocutaneous reactions in Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN) lead to extensive corneal scarring and limbal stem cell deficiency, resulting in trigeminal nerve damage. Patients who survive these conditions often develop chronic ocular complications, including neurotrophic keratitis.[27] Granulomatous inflammation in diseases such as sarcoidosis, granulomatosis with polyangiitis (GPA), and systemic lupus erythematosus (SLE) can infiltrate the trigeminal nerve, disrupting corneal sensation and predisposing patients to neurotrophic keratitis.[28]
Neurotrophic keratitis arises from a complex interplay of infectious, traumatic, neurological, iatrogenic, autoimmune, and metabolic factors. The trigeminal nerve plays a critical role in preserving corneal health, and any disruption to its function can lead to corneal anesthesia, PEDs, and ulceration. Identifying the underlying cause is essential for effective management, as targeted treatment can help prevent disease progression and support corneal healing.
Comprehensive diagnosis and treatment often require an interprofessional approach involving ophthalmologists, neurologists, rheumatologists, and infectious disease specialists. Advances in neurodegenerative therapies, corneal neurotization techniques, and NGF treatments hold promise for improving long-term outcomes in patients with neurotrophic keratitis.[29]
Epidemiology
Neurotrophic keratitis is a rare disease with an estimated prevalence of less than 5 per 10,000. The condition affects approximately 6% of herpetic keratitis cases, 12.8% of herpes zoster keratitis cases, and 2.8% of patients who have undergone surgical procedures for trigeminal neuralgia. Neurotrophic keratitis is rare but often underdiagnosed due to its insidious onset and lack of significant symptoms in the early stages. This potentially sight-threatening condition results from trigeminal nerve damage, leading to reduced or absent corneal sensation and impaired corneal healing. Epidemiologic patterns vary based on underlying etiology, geographic distribution, and access to diagnostic resources. A thorough understanding of these factors is essential for improving early detection, risk stratification, and therapeutic outcomes.
Prevalence and Incidence
Neurotrophic keratitis is a rare disease with an estimated prevalence of 5 per 10,000 individuals in the general population. However, this figure may underestimate the actual burden, as the condition is often undiagnosed or misdiagnosed, particularly in patients with chronic ocular surface disease or multiple comorbidities. The incidence of neurotrophic keratitis is closely linked to the prevalence of its primary causes, including herpetic keratitis, diabetes, and ocular surgeries.
Epidemiologic studies have provided insights into the condition's impact across different populations. A European multicenter study estimated the prevalence of moderate-to-severe neurotrophic keratitis at 0.02% of the general population. In the U.S., an epidemiological survey found that neurotrophic keratitis accounts for approximately 5% of corneal ulcers requiring ophthalmologic intervention. In tertiary eye care centers, the condition is identified in 3% to 4% of nonhealing corneal ulcer cases, underscoring its significance in corneal pathology. With the rising prevalence of diabetes, HZO, and iatrogenic corneal nerve damage from refractive and ocular surgeries, the burden of neurotrophic keratitis is expected to rise in the coming years.[30]
Distribution According to Age and Sex
Neurotrophic keratitis affects individuals across all age groups, but its incidence is higher in older adults, particularly those with underlying neurological disorders, diabetes, or a history of HZO. Adults older than 60 face an increased risk due to age-related decline in corneal nerve density and a higher prevalence of systemic diseases that impair trigeminal nerve function.
Pediatric cases of neurotrophic keratitis are rare but can occur in congenital trigeminal neuropathies, genetic dysautonomias, or birth-related cranial nerve injuries. Sex distribution varies based on the underlying cause. While neurotrophic keratitis associated with diabetes and herpetic infections is slightly more common in male patients, autoimmune conditions such as Sjögren syndrome and SLE-related neurotrophic keratitis are more frequently observed in female individuals.[31]
Geographic and Regional Variation
The prevalence of neurotrophic keratitis varies across regions due to differences in risk factors, healthcare access, and genetic predisposition. Herpetic keratitis remains the leading cause worldwide, with a higher prevalence in developed nations, where HSV and VZV are frequently encountered in ophthalmic practice.
Leprosy-related neurotrophic keratitis is a major public health concern in endemic regions such as South Asia, Africa, and parts of Latin America, where Mycobacterium leprae remains a leading cause of corneal nerve damage. Diabetes-related neurotrophic keratitis is more common in industrialized nations, where rising diabetes prevalence has increased the burden of trigeminal neuropathy and corneal complications.
Chemical burn-related neurotrophic keratitis occurs more frequently in regions with greater occupational exposure to industrial chemicals, particularly in developing countries with limited protective measures. Global disparities in healthcare infrastructure further influence diagnosis and management. In low-resource settings, a lack of early diagnostic tools and advanced treatments contributes to higher rates of corneal ulceration and vision loss due to undiagnosed neurotrophic keratitis.[32]
Etiology-Specific Epidemiology
The incidence of neurotrophic keratitis is closely tied to the prevalence of its underlying conditions. Herpetic infections, diabetes, ocular surgeries, and trauma account for most cases.[33]
HSV keratitis affects approximately 1.5 million individuals globally each year, with 10% to 20% of cases progressing to neurotrophic keratitis due to trigeminal nerve damage. HZO is a leading cause of postviral neurotrophic keratitis, affecting 20% to 25% of patients with ophthalmic shingles.[34]
Diabetes affects over 500 million people worldwide, and 20% to 30% of patients with diabetes exhibit corneal nerve abnormalities. Diabetes-related corneal neuropathy is a major risk factor for neurotrophic keratitis, particularly in individuals with diabetic retinopathy and autonomic neuropathy.[35]
Corneal surgeries, including LASIK, PRK, and corneal transplants, directly damage the corneal nerve plexus and contribute to up to 10% of neurotrophic keratitis cases. Trigeminal nerve injury following neurosurgical procedures, such as microvascular decompression for trigeminal neuralgia, has been implicated in the development of neurotrophic keratitis in approximately 5% to 8% of cases.[36]
Autoimmune diseases such as Sjögren syndrome, SLE, and sarcoidosis contribute to neurotrophic keratitis by causing chronic ocular surface inflammation and progressive nerve damage. Among patients with severe DED, neurotrophic keratitis occurs in approximately 3% to 5% of those with Sjögren syndrome.
Congenital disorders, including familial dysautonomia and Moebius syndrome, lead to early-onset neurotrophic keratitis, often detected in infancy. Stroke-related neurotrophic keratitis affects approximately 2% to 4% of patients with brainstem infarcts involving the trigeminal nerve pathways.[37]
Prevalence Across Disease Stages
The progression of neurotrophic keratitis follows a defined course, with an increasing risk of corneal ulceration, infection, and vision loss if left untreated. Stage 1 (mild disease) affects approximately 50% of diagnosed patients, with many cases remaining stable through lubricating therapy and regular monitoring. Stage 2 (moderate disease) accounts for 30% to 40% of cases, characterized by PEDs that require biological therapies such as autologous serum or NGF therapy. Stage 3 (severe disease) represents 10% to 20% of diagnosed cases. Due to stromal melting and perforation, management often requires surgical intervention, including tarsorrhaphy, AMT, or corneal neurotization.[9]
The prognosis of neurotrophic keratitis depends on early diagnosis and effective management. Patients diagnosed in Stage 1 or 2 typically achieve favorable visual outcomes, while those progressing to Stage 3 face an increased risk of blindness, secondary infections, and permanent corneal scarring.
Neurotrophic keratitis is rare but underdiagnosed, with significant clinical implications. The global burden of the disease is rising, driven by increasing cases of diabetes, herpetic infections, and postsurgical corneal nerve damage. While its overall prevalence remains low compared to other corneal diseases, delayed recognition and limited awareness contribute to severe complications and vision loss.
The epidemiology of neurotrophic keratitis varies by region and etiology, with herpetic keratitis, diabetes, and ocular trauma representing the most common causes. Understanding risk factors, geographic distribution, and disease progression is essential for early intervention and improved patient outcomes. Advances in emerging therapies, including cenegermin, corneal neurotization, and regenerative medicine, offer the potential to improve prognosis and long-term visual preservation in affected individuals.[38]
Pathophysiology
Corneal nerves are important in maintaining epithelial integrity, promoting proliferation, and facilitating wound healing. Corneal sensory nerve damage is believed to cause significant alterations in neuromodulator levels, leading to impaired epithelial cell vitality and metabolism. Disruptions in these processes can hinder epithelial cell mitosis, ultimately resulting in epithelial breakdown. Sensory nerve involvement also reduces the lacrimation reflex, further exacerbating corneal damage.[39]
Corneal epithelial thickness decreases in neurotrophic keratitis, with affected epithelial cells exhibiting intracellular swelling, microvilli loss, and abnormal basal lamina production. These morphological and metabolic disturbances contribute to persistent or recurrent epithelial defects, which may progress to corneal ulceration, stromal melting, and perforation. Various chemical mediators are implicated in the pathogenesis of neurotrophic keratitis, including NGF, substance P, neuropeptide Y, calcitonin gene-related peptide, galanin, and acetylcholine.[40]
Histopathology
Histopathological changes in neurotrophic keratitis involve progressive alterations in the corneal epithelium, stroma, and nerves, with variations based on disease severity and duration. Examining the microscopic and ultrastructural changes in neurotrophic keratitis provides valuable insight into its pathophysiology, progression, and response to treatment.
Corneal Epithelium Changes
Neurotrophic keratitis initially presents with pathological changes in the corneal epithelium, which progressively worsen as the disease advances. One of the earliest histological findings is epithelial thinning, where the normal 5 to 7 cell layers of the corneal epithelium are reduced to just 1 to 2 layers. Loss of basal epithelial cell adhesion to the basement membrane leads to epithelial instability and delayed wound healing. Epithelial degeneration is characterized by cytoplasmic vacuolization, loss of cellular polarity, and nuclear pyknosis.[41]
Moderate-to-severe neurotrophic keratitis presents with epithelial erosions, denuded regions, and PEDs that fail to heal due to inadequate trophic support from corneal nerves. A hallmark histopathological feature of neurotrophic keratitis is the absence of mitotic activity in basal epithelial cells. Apoptotic cell death frequently affects both the superficial and basal epithelial layers, further compromising epithelial stability.[42]
Neurotrophic keratitis leads to an irregular, fragmented, and thickened corneal basement membrane. Defective hemidesmosome formation weakens epithelial adhesion, increasing the risk of recurrent epithelial breakdown and ulceration.[43]
Stromal Findings
The corneal stroma undergoes progressive degenerative changes, particularly in advanced stages.[44] A reduction in collagen fibril density leads to stromal thinning, while electron microscopy studies reveal fragmented and irregularly arranged collagen fibrils, contributing to corneal haze and opacification. Keratocyte density is significantly reduced, impairing stromal repair.[45]
Apoptosis and necrosis of keratocytes are prominent in moderate-to-severe neurotrophic keratitis. The absence of keratocyte proliferation results in poor stromal healing, progressive tissue loss, and corneal melting. In advanced cases, stromal liquefaction and necrosis may lead to corneal perforation. Chronic neurotrophic keratitis often leads to vascularization of the normally avascular corneal stroma, particularly in longstanding cases with chronic inflammation. Fibrotic tissue deposition, driven by increased fibroblast activity and disorganized extracellular matrix remodeling, contributes to corneal scarring.
Corneal Nerve Changes
Neurotrophic keratitis is fundamentally a disorder of nerve dysfunction, leading to profound morphological alterations in corneal nerves. One of the most striking histopathological findings is the loss of subbasal corneal nerves, with confocal microscopy studies demonstrating a 60% to 80% reduction in corneal nerve fiber density compared to healthy controls. Degeneration of the corneal nerve plexus results in a loss of trophic factors, further exacerbating epithelial and stromal damage.[46]
The remaining nerve fibers appear fragmented, swollen, and irregular, while electron microscopy reveals axonal degeneration, Schwann cell loss, and myelin breakdown. In viral infections such as HSV and VZV, inflammatory infiltrates may surround the remaining nerve fibers.[47] A key characteristic of neurotrophic keratitis is the decreased expression of crucial neurotrophic factors, including NGF, brain-derived neurotrophic factor, and substance P. The absence of neurotrophic signaling impairs epithelial proliferation, disrupts wound healing, and leads to corneal anesthesia.[48]
Tear Film and Ocular Surface Changes
The tear film composition and conjunctival epithelium are also affected in neurotrophic keratitis, contributing to ocular surface instability.[49] Histological analysis of the conjunctiva reveals a reduction in goblet cell density, leading to decreased mucin production. Altered mucin composition disrupts tear film stability, exacerbating corneal desiccation and epithelial damage.[50] Inflammatory changes in the tear film further contribute to disease progression, with increased levels of pro-inflammatory cytokines such as interleukin 1 (IL-1), tumor necrosis factor α (TNF-α), and matrix metalloproteinase 9 (MMP-9) promoting corneal nerve damage and delayed epithelial healing.[51] Dysfunction of the meibomian glands, often secondary to chronic ocular surface inflammation, leads to lipid layer abnormalities in the tear film, increasing evaporative stress on the cornea.[52]
Advanced Histopathological Findings in Severe Neurotrophic Keratitis
In late-stage neurotrophic keratitis, several histopathological alterations become prominent. Corneal melting and perforation occur due to the complete loss of epithelial and stromal layers, leaving only the Descemet membrane intact. Inflammatory cell infiltrates, including macrophages, lymphocytes, and neutrophils, are frequently observed in response to chronic corneal ulceration. Corneal neovascularization develops in cases with longstanding inflammation or trauma, further compromising corneal transparency. In some instances, secondary bacterial or fungal infections lead to corneal abscess formation.[53]
Diagnostic Correlation Using Confocal Microscopy
IVCM is a noninvasive imaging tool that visualizes histopathological changes in real time. In early-stage disease, IVCM reveals reduced nerve density, irregular nerve beading, and decreased epithelial cell reflectivity. Moderate-stage findings include significant nerve loss, keratocyte dropout, and increased stromal haze. In severe cases, IVCM detects complete loss of corneal nerves, stromal necrosis, inflammatory cell infiltration, and vascularization. These observations closely align with histopathological findings and assist in disease staging and management.
The histopathology of neurotrophic keratitis offers critical insights into disease progression, from early epithelial dysfunction to advanced corneal melting and perforation. Hallmark features include epithelial thinning, basement membrane disruption, keratocyte loss, stromal fibrosis, and profound corneal nerve degeneration. Recognizing these microscopic changes facilitates accurate diagnosis, staging, and targeted therapeutic strategies. Advances in neurotrophic therapies, corneal neurotization, and regenerative approaches hold the potential for reversing some of these alterations, improving long-term outcomes for affected individuals.[54]
Toxicokinetics
Toxicokinetics encompasses the absorption, distribution, metabolism, and excretion of toxic substances and their effects on biological tissues. In neurotrophic keratitis, toxicokinetic factors mainly involve topical ophthalmic medications, systemic drugs, environmental toxins, and metabolic disorders that contribute to corneal nerve damage, ocular surface toxicity, and impaired healing. Prolonged exposure to ocular irritants, preservatives, neurotoxic agents, and metabolic imbalances disrupts corneal homeostasis, increasing the risk of progressive corneal degeneration and epithelial breakdown.[55]
Absorption and Local Toxic Effects in Neurotrophic Keratitis
The cornea functions as a semipermeable barrier that absorbs topically applied medications and environmental toxins, which can directly contribute to corneal toxicity and neurotrophic damage. Ophthalmic drugs containing preservatives and some active ingredients can damage corneal epithelial cells, impair nerve function, and delay wound healing. BAK, a common preservative in eye drops, induces dose-dependent corneal epithelial cytotoxicity, mitochondrial dysfunction, and oxidative stress.
Chronic use of preserved glaucoma medications, such as prostaglandin analogs and β-blockers, can promote neurotrophic changes by destabilizing the tear film, increasing ocular surface inflammation, and triggering corneal nerve apoptosis. Topical anesthetic abuse (eg, proparacaine, tetracaine) leads to progressive neuroepithelial toxicity, corneal ulceration, and stromal necrosis.[56]
Systemic medications with toxicokinetic properties can also affect the corneal epithelium and nerves. Chemotherapeutic agents such as 5-fluorouracil, paclitaxel, and docetaxel contribute to corneal epithelial toxicity and reduced nerve density, increasing the risk of neurotrophic keratitis. Additionally, antidepressants, antipsychotics, and anticonvulsants that block sodium or calcium channels may impair corneal nerve function, leading to corneal anesthesia and delayed epithelial healing.[57]
Environmental and chemical exposures further contribute to corneal nerve damage. Prolonged exposure to industrial chemicals, air pollution, and UV radiation induces oxidative damage to corneal nerves. Chemical burns from acids or alkalis cause irreversible nerve injury, resulting in severe neurotrophic changes and corneal melting.[58]
Distribution of Toxic Agents in the Cornea and Tear Film
The distribution of toxic substances in the ocular microenvironment significantly influences the pathophysiology of neurotrophic keratitis. Lipophilic drugs and preservatives penetrate deeper into the corneal stroma, where they accumulate and induce chronic corneal nerve inflammation and toxicity. In contrast, hydrophilic agents, such as β-blockers and prostaglandin analogs, persist in the tear film, leading to prolonged ocular surface irritation and epithelial compromise.[59]
Chronic exposure to toxic ophthalmic agents disrupts tear film stability, altering tear osmolarity and increasing inflammatory cytokines (IL-1, TNF-α), which contribute to oxidative stress. Neuroinflammatory pathways involving reactive oxygen species (ROS) and matrix metalloproteinases (MMPs) further exacerbate corneal nerve apoptosis and delay healing.[60]
Metabolism and Biotransformation of Toxic Agents in Neurotrophic Keratitis
The corneal epithelium and stroma have limited metabolic capacity, making them susceptible to prolonged exposure to toxic substances. Unlike the liver, the cornea lacks robust enzymatic pathways for metabolizing toxic compounds, leading to prolonged retention and bioaccumulation of harmful agents. Chronic exposure to oxidative stressors overwhelms glutathione-dependent detoxification mechanisms in corneal cells, exacerbating damage to corneal nerves and epithelial cells.[61]
Systemic conditions further influence ocular metabolism. Diabetes mellitus increases oxidative stress, promotes the accumulation of glycation end products, and impairs nerve function, heightening the risk of neurotrophic keratitis. Vitamin A deficiency compromises corneal metabolism by reducing mucin production and epithelial integrity, further predisposing to corneal surface damage and neurotrophic changes.[62]
Excretion and Clearance of Toxic Agents
Tear turnover, blinking, and lacrimal drainage regulate the clearance of toxic agents from the ocular surface. Patients with neurotrophic keratitis exhibit reduced blink reflexes and impaired tear film dynamics, resulting in prolonged retention of toxic agents. Tear film stagnation extends corneal exposure to inflammatory mediators and oxidative stress, worsening corneal toxicity.[63]
Ocular surface diseases further delay drug clearance. Chronic use of ophthalmic medications in patients with DED increases drug retention, heightening the risk of toxicity. Lacrimal drainage impairment in conditions such as facial nerve palsy or Sjögren syndrome prolongs the contact time of toxic agents on the corneal surface, exacerbating corneal damage.[64]
Role of Toxicokinetics in Neurotrophic Keratitis Progression
The toxicokinetics of ophthalmic and systemic agents contribute to neurotrophic keratitis by directly damaging corneal nerves, impairing neurotrophic signaling, and inducing oxidative stress and inflammatory pathways. These mechanisms delay epithelial healing, promote stromal degradation, and disrupt tear film homeostasis, exacerbating corneal desiccation and ulcer formation. Bioaccumulation of toxic agents further leads to progressive neurodegeneration and corneal thinning.
The absorption, distribution, metabolism, and clearance of ophthalmic preservatives, systemic neurotoxic agents, metabolic disorders, and environmental toxins significantly impact corneal nerve function, epithelial healing, and ocular surface integrity. Chronic exposure to ophthalmic medications, such as BAK, anesthetics, and glaucoma drops, along with systemic neurotoxic agents and oxidative stressors, contributes to corneal neurodegeneration and impaired wound healing.
Addressing toxicokinetic factors through pharmacologic modifications, preservative-free formulations, antioxidant therapy, and neuroprotective strategies is essential for managing and preventing disease progression. Future research into ocular pharmacokinetics, neurotrophic factor modulation, and regenerative therapies may offer novel approaches to mitigating toxic effects on corneal nerves and improving outcomes in neurotrophic keratitis.
History and Physical
Neurotrophic keratitis is a rare but serious degenerative corneal disease characterized by corneal anesthesia, impaired healing, and progressive epithelial breakdown. Although clinical symptoms may be subtle or absent due to corneal sensory loss, a thorough patient history, comprehensive ocular examination, and targeted corneal sensitivity testing are essential for early detection and management.
History
The hallmark feature of neurotrophic keratitis is reduced or absent corneal sensation. A detailed patient history helps identify risk factors and underlying conditions contributing to trigeminal nerve dysfunction and corneal anesthesia.
Patients with neurotrophic keratitis rarely report symptoms and often present late in the disease course, likely due to diminished corneal sensation. However, some may initially develop redness and blurred vision. Visual impairment can result from PEDs, corneal edema, or scarring. A history of recurrent redness, eye pain, or cutaneous blistering and scarring may suggest prior herpetic infections. Additional risk factors include corneal trauma, surgery, chemical burns, prolonged use of topical medications, neurosurgical procedures, and diabetes.
Disease severity is classified into 3 stages based on the Mackie classification, with progression from stage 1 to stage 3 reflecting increasing corneal damage. Stage 1 presents with corneal epithelial changes, including a dry and cloudy epithelium, superficial punctate keratopathy, and corneal edema. Stage 2 is marked by persistent or recurrent epithelial defects, typically oval or circular in shape and most often localized to the superior half of the cornea. Stage 3 involves corneal ulceration with stromal involvement, which may lead to stromal melting and, in severe cases, corneal perforation.
The most common complaints include the following:
- Blurred vision due to PEDs, corneal edema, or scarring
- Mild redness, as inflammation is blunted by sensory loss
- Foreign body sensation, which is rare unless some sensation remains
- Increased tear film instability or DED symptoms
Severe cases may present with corneal ulceration or stromal thinning without preceding pain. Nonhealing PEDs or secondary infections may result from epithelial compromise.
Neurotrophic keratitis is often secondary to systemic or ocular conditions affecting the trigeminal nerve. The following must be carefully assessed:
- History of herpetic eye disease (HSV, HZO)
- Diabetes mellitus (peripheral neuropathy affecting corneal nerves)
- Ocular surgeries (LASIK, PRK, corneal transplantation, trigeminal nerve decompression)
- Neurological disorders (stroke, brainstem tumors, multiple sclerosis)
- Chemical or thermal burns (exposure to acids, alkalis, and temperature extremes)
- Chronic topical medication use (glaucoma drugs, anesthetic abuse, preservatives like BAK)
- Autoimmune diseases (Sjögren syndrome, sarcoidosis)
- Congenital conditions (Riley-Day syndrome, Moebius syndrome, familial dysautonomia)
Chronic use of preserved glaucoma medications, especially prostaglandin analogs, β-blockers, and carbonic anhydrase inhibitors, can lead to corneal toxicity and neurotrophic changes. Long-term corticosteroid use may suppress corneal healing. Abuse of topical anesthetics (proparacaine, tetracaine) can cause severe corneal damage and should always be ruled out.[65]
Physical Examination and Ocular Assessment
A detailed ocular examination is necessary to assess corneal structure, function, and nerve integrity. Visual acuity testing provides insight into disease severity. In early neurotrophic keratitis, visual acuity may remain relatively unaffected. As the disease progresses to a moderate stage, PEDs and mild corneal haze begin to impair vision. In severe cases, corneal ulceration, stromal scarring, and secondary infections can result in significant visual loss.[66]
Slit-lamp biomicroscopy is essential for staging the disease and identifying hallmark corneal changes. In stage 1, findings include punctate epithelial erosions, an irregular tear film, reduced corneal luster, and epithelial thinning without ulceration. Stage 2 is characterized by a PED with rolled edges, stromal involvement with mild haze or edema, and minimal inflammation despite evident epithelial damage. Stage 3 involves corneal ulceration, stromal melting, and thinning. In extreme cases, the Descemet membrane may bulge outward, forming a descemetocele. End-stage disease can progress to corneal perforation.[67]
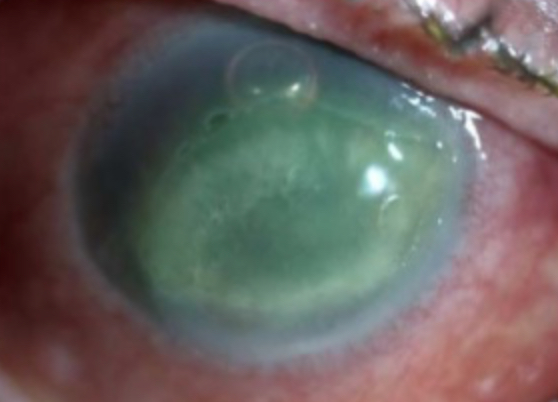
Fluorescein dye application helps detect epithelial defects, ulcerations, and tear film abnormalities (see Image. Stage 1 Neurotrophic Keratitis). A delayed or absent corneal staining response suggests neurotrophic dysfunction. Additional staining techniques, such as Rose Bengal or Lissamine Green, can highlight conjunctival and epithelial irregularities.[68]
Loss or reduction of corneal sensation is the hallmark feature of neurotrophic keratitis that distinguishes it from other corneal diseases. The Cochet-Bonnet esthesiometer measures mechanical corneal sensitivity using a thin filament. This method is considered the gold standard for assessing corneal sensation. Normal corneal sensitivity corresponds to a response at a filament length of 5 to 6 cm. In early neurotrophic keratitis, sensitivity is reduced to 2 to 4 cm, while severe cases exhibit no response at 0 cm.
Alternative bedside methods include the cotton wisp test, which involves lightly touching the cornea to assess the blink response, and the dental floss test, a more standardized technique for evaluating corneal sensation. Contact lens wearers require separate corneal sensitivity assessments, as long-term lens use can diminish corneal nerve function.[69]
The Schirmer test evaluates tear production and often reveals reduced tear secretion in patients with neurotrophic keratitis. TBUT is typically shortened, reflecting tear film instability. Abnormalities in the mucin and lipid layers further contribute to corneal desiccation and impaired epithelial healing.[70]
Ancillary Diagnostic Tests
Several advanced imaging and diagnostic techniques help confirm neurotrophic keratitis and evaluate disease severity.[71] IVCM is a noninvasive imaging tool that provides real-time visualization of corneal nerves. In neurotrophic keratitis, IVCM findings include reduced subbasal nerve plexus density, nerve beading and fragmentation, and increased keratocyte apoptosis in severe cases.[72]
AS-OCT helps quantify corneal thinning and ulcer depth while also assessing stromal changes and tear film dynamics.[73] Impression cytology and biomarker analysis evaluate ocular surface inflammation and epithelial dysfunction. Patients with neurotrophic keratitis exhibit increased levels of pro-inflammatory cytokines, such as IL-1 and TNF-α, and decreased neurotrophic factors, including NGF and brain-derived neurotrophic factor.[74]
Corneal nerve imaging and analysis provide a comprehensive assessment of nerve integrity and regenerative potential. Combining corneal esthesiometry with confocal microscopy offers valuable insight into disease progression and response to treatment.[75]
Differential Diagnosis
While corneal anesthesia is pathognomonic for neurotrophic keratitis, similar clinical presentations occur in the following conditions:
- Exposure keratopathy (eg, lagophthalmos in facial nerve palsy)
- Severe DED (eg, Sjögren syndrome)
- Persistent corneal infections (eg, fungal keratitis, bacterial ulcers)
- Neurotrophic-related postsurgical complications (eg, post-LASIK dry eye vs. true neurotrophic keratitis)
A detailed history and physical examination are essential in the early detection and management of neurotrophic keratitis. Patients often lack pain perception, making clinical suspicion and objective tests, such as corneal sensitivity assessment, slit-lamp examination, and confocal microscopy, vital for accurate diagnosis. Identifying underlying risk factors, including diabetes, herpetic eye disease, prior ocular surgeries, and neurovascular conditions, enables targeted therapeutic interventions. Corneal sensitivity testing remains the cornerstone of diagnosing neurotrophic keratitis, while advanced imaging tools assist in disease staging and treatment monitoring. Early detection and an interprofessional approach are vital for preventing corneal ulceration, stromal melting, and vision-threatening complications.[76]
Evaluation
Neurotrophic keratitis is often suspected based on patient history, particularly in individuals with trigeminal impairment, PEDs or corneal ulcers, and decreased corneal sensitivity. Evaluating systemic diseases such as diabetes mellitus, medication use (eg, neuroleptics), and corneal risk factors (eg, contact lens misuse and chemical burns) is essential. A clinical assessment of cranial nerve function can help localize the site of the lesion. The presence of 7th or 8th nerve palsy may suggest an acoustic neuroma or surgical resection affecting the trigeminal nerve, while 3rd, 4th, and 6th cranial nerve involvement may indicate cavernous sinus pathology. Quantitative corneal sensitivity testing using a Cochet-Bonnet or no-contact gas esthesiometer provides an objective measure of sensory impairment.
A slit-lamp examination is crucial for identifying characteristic corneal lesions and sectoral iris atrophy, which is often associated with herpetic infections. If an ulcer is present, a microbiological evaluation is necessary to rule out an infection. A dilated fundus examination may reveal a pale or swollen optic disc in cases where intracranial tumors compress the trigeminal nerve. Assessing tear film function is also important, as reduced corneal sensitivity can alter the tear film and contribute to a cycle of worsening ocular surface health. Eyelid evaluation is necessary for both diagnostic and prognostic purposes, as conditions such as lagophthalmos can exacerbate neurotrophic keratitis.
The differential diagnosis of neurotrophic keratitis includes several other ocular conditions that may present with similar clinical features, such as the following:
- Bacterial keratitis
- Corneal mucus plaques
- DED
- HSV or VZV infection
- Keratoconjunctivitis
- Postoperative corneal melt
- Sjogren syndrome
A systematic clinical assessment, including history-taking, corneal sensitivity testing, slit-lamp examination, and advanced imaging, is essential for evaluating neurotrophic keratitis. Patients often lack significant symptoms due to corneal anesthesia. Therefore, early detection relies on a high index of suspicion and objective diagnostic testing to stage the disease and guide management.
Clinical Examination and Ocular Assessment
The cornerstone of neurotrophic keratitis evaluation is a detailed ocular examination focusing on corneal integrity, nerve function, and disease staging. Visual acuity testing helps assess disease severity. In stage 1, vision may remain normal or slightly reduced due to minor epithelial irregularities. Stage 2 presents with decreased vision caused by PEDs, mild corneal haze, or an irregular tear film. In stage 3, vision is markedly impaired due to corneal ulceration, stromal melting, scarring, or secondary infection.
Corneal sensation testing is an essential step for diagnosing neurotrophic keratitis, as loss or significant reduction in corneal sensation differentiates the condition from other causes of corneal ulceration. The Cochet-Bonnet esthesiometer measures corneal mechanical sensitivity using a nylon filament. A normal response occurs at 5 to 6 cm, while early neurotrophic keratitis shows reduced sensitivity (2–4 cm), and severe disease results in no response at 0 cm. Alternative bedside methods include the cotton wisp test, which assesses the blink reflex by lightly touching the cornea, and the dental floss test, which provides a standardized sensory evaluation for corneal anesthesia.
Slit-lamp biomicroscopy helps stage neurotrophic keratitis and identify structural damage. In stage 1, punctate epithelial erosions, tear film instability, and corneal haze are observed. Stage 2 is characterized by a PED with rolled edges, stromal involvement, and corneal thinning. Stage 3 presents with corneal ulceration, stromal melting, descemetocele formation, or perforation.[77]
Fluorescein staining and tear film analysis aid in detecting epithelial defects, delayed wound healing, and ulceration. A reduced or absent staining response suggests corneal anesthesia and neurotrophic dysfunction. Rose Bengal or Lissamine Green staining highlights conjunctival involvement.[78]
The Schirmer test and TBUT further assess tear function. The Schirmer test often reveals reduced tear production in neurotrophic keratitis patients, while a shortened TBUT indicates tear film instability, which can exacerbate ocular surface stress.[79]
Advanced Imaging and Diagnostic Modalities
Several imaging techniques aid in evaluating neurotrophic keratitis severity, nerve integrity, and disease progression.[80] IVCM provides real-time visualization of corneal nerve damage. Findings in neurotrophic keratitis include reduced subbasal nerve plexus density, fragmented and beaded nerve fibers, and keratocyte apoptosis in moderate-to-severe disease.[81] AS-OCT offers high-resolution corneal imaging to assess epithelial integrity, stromal thinning, and ulcer depth. This technique is valuable for quantifying disease progression and monitoring treatment response.
Impression cytology and biomarker analysis help detect ocular surface inflammation and epithelial abnormalities. Elevated levels of IL-1 and TNF-α, along with reduced NGF, suggest chronic neurotrophic disease.[82]
A systematic evaluation incorporating history, corneal sensitivity testing, slit-lamp examination, and advanced imaging is crucial for early detection, disease staging, and appropriate management. Neurotrophic keratitis should always be suspected in patients with nonhealing corneal defects and reduced corneal sensation. Prompt diagnosis and intervention can prevent severe complications such as corneal perforation and vision loss.
Treatment / Management
Overview
Early diagnosis, treatment, and careful monitoring of patients with neurotrophic keratitis are mandatory to achieve epithelial healing and prevent the progression of corneal damage. Preservative-free artificial tears may help improve the corneal surface at all stages of disease severity. In cases of stromal melting, topical collagenase inhibitors such as N-acetylcysteine, along with systemic administration of tetracycline or medroxyprogesterone, may be considered. The use of topical antibiotic eye drops is recommended in stages 2 and 3 to prevent infection. Topical NGF and autologous serum eye drops have emerged as promising treatments for neurotrophic keratopathy.[83]
Surgical treatments are reserved for refractory cases. Options include partial or total tarsorrhaphy, AMT, conjunctival flap procedures, and botulinum A toxin injection into the eyelid elevator muscle. Management strategies vary by disease stage. In stage 1, preservative-free artificial tears remain the primary treatment approach. Stage 2, characterized by PEDs, may require conjunctival flap procedures and partial tarsorrhaphy.
Stage 3, which involves corneal ulceration and risk of perforation, is managed with therapeutic contact lenses and AMT. Corneal perforations may necessitate cyanoacrylate glue application, conjunctival flap procedures, or lamellar or penetrating keratoplasty. Neurotization surgery, involving direct transfer of the supratrochlear or supraorbital nerves to the subconjunctival space, has shown promise in restoring corneal innervation.[84][85][86](B2)
The management of neurotrophic keratitis focuses on protecting the ocular surface, promoting corneal healing, preventing complications, and, when possible, restoring corneal nerve function. Treatment is stage-dependent, with mild cases managed conservatively and moderate-to-severe cases requiring advanced therapies and surgical interventions. To achieve optimal outcomes, an interprofessional approach involving ophthalmologists, neurologists, and corneal specialists is often necessary.[87]
The general principles of treatment for neurotrophic keratitis focus on reducing epithelial stress, optimizing corneal hydration, and protecting the ocular surface from trauma, desiccation, and infection. Depending on the severity of the disease, strategies to enhance epithelial healing involve both pharmacologic and surgical interventions. In select cases, efforts to restore corneal nerve function may be considered to improve long-term outcomes.[88]
Treatment for Mild Neurotrophic Keratitis
Treatment for mild neurotrophic keratitis focuses on improving ocular surface hydration, stabilizing the tear film, and promoting epithelial healing. Lubrication therapy is the cornerstone of treatment, with frequent use of preservative-free artificial tears, typically administered hourly. Gel-based or lipid-based tear substitutes may offer better retention and protection. Autologous serum eye drops (ASED) or PRP, rich in epithelial growth factors, cytokines, and vitamins, are often used 4 to 6 times daily to enhance corneal healing.[89](B3)
Tear film protection is crucial for management. Punctal occlusion, either temporary or permanent, helps improve tear retention and ocular surface hydration. Depending on individual needs, silicone punctal plugs or thermal cautery may be used. Moisture chamber goggles provide an additional protective measure by reducing tear evaporation and minimizing ocular surface stress.[90]
Toxic medications must be discontinued to prevent further corneal damage. Preserved eye drops, such as glaucoma medications containing BAK, should be avoided. Chronic topical anesthetics must be discontinued due to their highly toxic effects on corneal nerves. Long-term corticosteroids should be reduced or eliminated, as they impair epithelial healing.
NGF therapy with cenegermin, an FDA-approved recombinant human NGF, offers a targeted approach to stimulate corneal nerve regeneration. This therapy is particularly beneficial for stage 1 and stage 2 neurotrophic keratitis, helping prevent disease progression. Cenegermin is administered 6 times daily for 8 weeks to enhance corneal healing and nerve function recovery.[91]
Treatment for Moderate Neurotrophic Keratitis
Treatment for moderate neurotrophic keratitis focuses on promoting epithelial healing, preventing corneal ulceration, and protecting the ocular surface. Biological therapy is key to management, with AMT offering anti-inflammatory, anti-scarring, and epithelial growth-promoting effects. This treatment can be applied as a self-retaining cryopreserved amniotic membrane, such as ProKera, or as sutured or glued grafts for larger defects.[92] Cenegermin has proven effective in managing PEDs by stimulating nerve regeneration and epithelial healing. Serum eye drops, including autologous serum or PRP, provide additional support for nonhealing corneal epithelial defects.[93]
Protective strategies help maintain corneal integrity and reduce further damage. Soft therapeutic contact lenses, such as bandage or scleral lenses, shield the cornea from mechanical trauma while ensuring continuous hydration. Tarsorrhaphy, either temporary or permanent, serves as another protective measure by partially or completely closing the eyelids to minimize corneal exposure and desiccation. This procedure may be performed surgically or induced with botulinum toxin to achieve protective ptosis.
Treatment for Severe Neurotrophic Keratitis
Treatment for severe neurotrophic keratitis aims to prevent corneal perforation, restore structural integrity, and promote healing. Surgical interventions are often necessary for advanced disease. Tarsorrhaphy is essential in cases with severe epithelial defects and stromal thinning, providing protection and reducing desiccation. Amniotic membrane grafts serve as a critical measure in managing deep ulcers or corneal perforation risk. For larger defects, scleral or conjunctival flap surgery promotes healing by covering the damaged area.
Corneal neurotization, an emerging therapy, restores nerve function by rerouting healthy nerves such as the supraorbital, infraorbital, or great auricular nerve. This technique has demonstrated promising results in corneal reinnervation. Keratoplasty, either lamellar or penetrating, is considered in cases of deep corneal scarring or perforation, though graft survival remains poor due to persistent neurotrophic dysfunction. For end-stage neurotrophic keratitis, the Boston KPro provides an option when other treatments fail. However, this intervention requires lifelong monitoring due to the risk of complications, including infection and corneal melt.
Adjunctive treatments help stabilize corneal integrity and prevent further deterioration. Topical and systemic MMP inhibitors, such as doxycycline (50 mg BID) and medroxyprogesterone 1% eye drops, help reduce corneal melting and stromal degradation. Systemic immunosuppressive therapy, including cyclosporine, tacrolimus, and biologics, is considered in neurotrophic keratitis associated with autoimmune diseases such as Sjögren syndrome and SLE.[94](B3)
Emerging and future therapies hold promise for improving outcomes in severe neurotrophic keratitis. Gene therapy research focuses on NGF gene delivery to restore corneal innervation. Corneal stem cell therapy, including mesenchymal stem cells and limbal epithelial transplantation, is under investigation for its potential to enhance corneal regeneration. Bioengineered corneal replacements, such as artificial corneas incorporating neurotrophic factors, represent another avenue for advancing corneal healing and restoring visual function.[95]
An interprofessional approach is essential for managing neurotrophic keratitis, requiring collaboration among specialists. Ophthalmologists, including cornea specialists and refractive surgeons, lead the management by assessing corneal integrity and guiding treatment. Neurologists play a crucial role in evaluating trigeminal nerve pathology, which underlies the disease. Endocrinologists contribute by addressing diabetic-related neurotrophic keratitis, while rheumatologists assist in managing cases associated with autoimmune disorders.
Overall, effective treatment hinges on stage-dependent therapy. Early intervention with NGF therapy, lubricants, and punctal occlusion helps prevent disease progression. In more advanced cases, biological and surgical options, such as AMT, tarsorrhaphy, and corneal neurotization, become necessary. Corneal neurotization offers a promising approach to restoring nerve function and improving long-term outcomes. Timely diagnosis and aggressive management of neurotrophic keratitis can significantly reduce the risk of corneal perforation and vision loss.[96]
Differential Diagnosis
Neurotrophic keratitis presents with persistent corneal epithelial defects, ulceration, and impaired healing, often without pain due to corneal sensory loss. However, several ocular surface diseases and corneal pathologies mimic this condition, leading to potential misdiagnosis and inappropriate treatment. A systematic approach to differentiating neurotrophic keratitis from similar conditions is essential for accurate diagnosis and management.
Exposure Keratopathy
Neurotrophic keratitis and exposure keratopathy share key clinical features, including epithelial defects, corneal ulceration, and ocular surface instability. Both conditions may arise in the context of facial nerve palsy, as seen in Bell palsy or acoustic neuroma, as well as eyelid malposition, such as lagophthalmos or ectropion. A reduced blink rate, commonly observed in conditions like Parkinson disease or stroke, can also contribute to their development. Key differences are summarized in Table 1 (see Table 1. Key Differences Between Neurotrophic Keratitis and Exposure Keratopathy).
Table 1. Key Differences Between Neurotrophic Keratitis and Exposure Keratopathy
| Condition | Neurotrophic Keratitis | Exposure Keratopathy |
| Corneal Sensation | Severely diminished or completely absent | Normal or slightly reduced |
| Blink Reflex | Functionally normal, but corneal anesthesia prevents reflexive protection | Reduced blink frequency or incomplete eyelid closure |
| Fluorescein Staining | Epithelial defects primarily affecting the central cornea | Staining concentrated in the inferior or interpalpebral region |
| Management | Treatments aimed at nerve regeneration and corneal healing, including NGF therapy, tarsorrhaphy, and autologous serum eye drops | Interventions focused on improving eyelid function and ocular surface protection, such as eyelid surgery, lubricating eye drops, and moisture goggles |
Severe Dry Eye Disease
Severe DED or aqueous-deficient dry eye is often associated with Sjögren syndrome. Chronic ocular surface inflammation leads to punctate epithelial erosions and epithelial instability in both severe DED and neurotrophic keratitis. Patients with Sjögren syndrome or other autoimmune diseases may also develop PEDs. Key differences are summarized in Table 2 (see Table 2. Key Differences Between Neurotrophic Keratitis and Severe Dry Eye Disease).
Table 2. Key Differences Between Neurotrophic Keratitis and Severe Dry Eye Disease
| Condition | Neurotrophic Keratitis | Severe Dry Eye Disease |
| Corneal Sensation | Absent or significantly reduced | Normal or slightly reduced |
| Tear Production | Usually remains normal unless DED occurs concurrently | Significantly reduced in cases related to Sjögren syndrome |
| Inflammatory Markers | Minimal inflammation due to an impaired neurogenic response | Increased levels of inflammatory cytokines such as IL-1 and TNF-α, along with conjunctival redness |
| Management | Treatment includes NGF therapy, AMT, and corneal neurotization | Treatment involves the use of lifitegrast, cyclosporine, and punctal occlusion |
Infectious Keratitis
Infectious keratitis, whether bacterial, viral, or fungal, shares key clinical features with neurotrophic keratitis. Both conditions can present with corneal ulceration and stromal involvement. In severe cases, patients may experience corneal thinning, melting, and an increased risk of perforation.[97][98] Key differences are summarized in Table 3 (see Table 3. Key Differences Between Neurotrophic Keratitis and Infectious Keratitis).
Table 3. Key Differences Between Neurotrophic Keratitis and Infectious Keratitis
| Condition | Neurotrophic Keratitis | Infectious Keratitis |
| Pain | Absent or minimal | Significant pain with photophobia |
| Corneal Sensation | Reduced or absent | Normal or mildly reduced |
| Ulcer Characteristics | Indolent, nonhealing with rolled edges | Suppurative and infiltrative with active inflammation |
| Fluorescein Staining | Epithelial defects that do not retain stain | Dense ulceration with prominent staining |
| Presence of Hypopyon (Anterior Chamber Reaction) | Minimal inflammation due to lack of neurogenic response | Common in bacterial and fungal infections |
| Microbiological Evaluation | Negative culture results | Positive culture results, indicating bacterial or fungal infection |
| Management | Lubrication, NGF therapy, AMT | Antimicrobial therapy and corneal debridement |
Herpetic Keratitis
Both neurotrophic keratitis and herpetic keratitis involve trigeminal nerve impairment, leading to corneal epithelial defects and recurrent ulcers. Viral neuropathy in herpetic keratitis can result in corneal anesthesia, similar to neurotrophic keratitis. Key differences are summarized in Table 4 (see Table 4. Key Differences Between Neurotrophic Keratitis and Herpetic Keratitis).
Table 4. Key Differences Between Neurotrophic Keratitis and Herpetic Keratitis
| Condition | Neurotrophic Keratitis | Herpetic Keratitis |
| Pain | Absent or minimal | Pain or photophobia often present, especially in active HSV or VZV infection |
| Corneal Sensation | Absent or reduced | Reduced, but may be patchy |
| Ulcer Shape | Indolent, smooth-edged, PED | Dendritic (HSV) or geographic (VZV) ulcers with terminal bulbs |
| Recurrence | Progressive worsening without active inflammation | Recurrent episodes with acute inflammation |
| Polymerase chain reaction/Serology | Negative result | Positive HSV or VZV test result |
| Management | NGF therapy, AMT, tarsorrhaphy | Antivirals (acyclovir, valacyclovir), topical steroids for stromal disease |
Postsurgical Persistent Epithelial Defects
Persistent corneal epithelial defects following LASIK, PRK, and corneal transplantation may mimic those of neurotrophic keratitis. Each case involves impaired corneal healing and epithelial instability. Key differences are summarized in Table 5 (see Table 5. Key Differences Between Neurotrophic Keratitis and Postsurgical Persistent Epithelial Defects).
Table 5. Key Differences Between Neurotrophic Keratitis and Postsurgical Persistent Epithelial Defects
| Condition | Neurotrophic Keratitis | Herpetic Keratitis |
| Corneal Sensation | Reduced or absent | Usually normal unless significant nerve disruption occurs |
| History | Trigeminal neuropathy or systemic disease | Recent ocular surgery (LASIK, PKP, radial keratotomy, PRK, Descemet stripping endothelial keratoplasty, deep anterior lamellar keratoplasty) |
| Management | Neurotrophic support, NGF, tarsorrhaphy | Tear supplements, contact lenses, epithelial debridement |
Neuropathic Pain Syndromes
Both neurotrophic keratitis and corneal neuropathy result from corneal nerve dysfunction. In both conditions, patients may report ocular discomfort despite minimal or absent clinical findings.[99] Key differences are summarized in Table 6 (see Table 6. Key Differences Between Neurotrophic Keratitis and Corneal Neuropathy).
Table 6. Key Differences Between Neurotrophic Keratitis and Corneal Neuropathy
| Condition | Neurotrophic Keratitis | Corneal Neuropathy (Neuropathic Ocular Pain) |
| Pain | Absent or minimal | Disproportionate pain, ie, burning, stabbing, or hypersensitivity, despite normal exam |
| Corneal Sensation | Reduced or absent | Hyperalgesia or paradoxical hypersensitivity often present |
| Confocal Microscopy | Absent nerve fibers | Hyperactive nerve endings with abnormal morphology |
| Management | NGF therapy, surgical intervention | Neuromodulators, eg, gabapentin, amitriptyline, pregabalin, and nerve blocks |
Diagnosing neurotrophic keratitis requires a comprehensive patient history, corneal sensation testing, slit-lamp examination, and targeted imaging. To prevent misdiagnosis and inappropriate management, conditions such as exposure keratopathy, severe DED, herpetic keratitis, and microbial infections must be carefully excluded. Corneal sensitivity testing remains the primary differentiator in neurotrophic keratitis. Early intervention with neurotrophic support, AMT, or surgical options can prevent progression to corneal perforation.
Pertinent Studies and Ongoing Trials
Significant treatment advancements have been made for neurotrophic keratitis, particularly NGF therapy, AMT, and corneal neurotization. Several clinical trials and observational studies provide evidence supporting the efficacy of these novel interventions.
Nerve Growth Factor Therapy: The Cenegermin Trials
NGF therapy, specifically cenegermin (Oxervate™), has shown promising results in treating neurotrophic keratitis. The REPARO trial (Phase II/III, 2018) evaluated the efficacy of recombinant human NGF (cenegermin-bkbj). The results demonstrated that 72% of patients achieved complete corneal healing after 8 weeks of treatment. Additionally, long-term follow-up showed sustained corneal integrity in 80% of the responders. These findings support the conclusion that NGF therapy promotes corneal nerve regeneration and enhances epithelial healing.[100]
Meanwhile, the ongoing NGF-PATCH study is investigating the long-term benefits of NGF therapy and its role in preventing the recurrence of neurotrophic keratitis. Preliminary results indicate that NGF therapy improves corneal sensation and epithelial stability after treatment.[101]
Amniotic Membrane Transplantation Studies
The PROKERA® Clinical Trials evaluated the use of cryopreserved amniotic membrane (PROKERA) for treating moderate-to-severe neurotrophic keratitis. The results indicated that 65% to 80% of patients experienced complete healing of PEDs. In addition to promoting epithelial healing, PROKERA reduced inflammation and improved tear film stability. The study concluded that AMT may offer an effective alternative for epithelial healing in patients who do not respond to conventional therapies.[102]
The Sutureless AMT vs. Sutured AMT Trial (2022) compared the outcomes of self-retaining versus sutured amniotic membrane grafts. Findings revealed that sutureless AMT led to faster epithelial healing and improved patient comfort, highlighting its advantages over the traditional sutured method.[103]
Corneal Neurotization Trials
The Neurotrophic Keratopathy Surgical Neurotization Trial (2021) assessed the outcomes of direct nerve transfer techniques in neurotrophic keratitis. The trial demonstrated that 70% to 90% of patients experienced restoration of corneal sensation. Additionally, the procedure was shown to have significantly improved epithelial healing and reduced recurrent corneal ulcers. The study concluded that corneal neurotization is a promising approach for restoring functional corneal nerves and improving the management of neurotrophic keratitis.
Studies Involving Topical and Systemic Anti-Inflammatory Agents
The Doxycycline & Medroxyprogesterone for Corneal Melting Study (2020) explored the effects of systemic doxycycline (50 mg twice daily) and topical medroxyprogesterone 1% on stromal melting in severe neurotrophic keratitis cases. The study found that this combination significantly reduced stromal melting and recommended it for cases at high risk of corneal perforation.[104]
Future and Ongoing Research
Ongoing research into gene therapy aims to regenerate corneal nerves, with early trials conducted in animal models. Additionally, biological corneal replacements that incorporate neurotrophic factors show promise. Artificial intelligence-based monitoring systems are also being developed to detect early stages of neurotrophic keratitis using ocular surface imaging.
The evidence from clinical trials and ongoing studies underscores the effectiveness of NGF therapy, AMT, and surgical neurotization in managing neurotrophic keratitis. Future advancements in gene therapy and regenerative medicine bring optimism for restoring corneal innervation and halting the progression of this condition.
Treatment Planning
Treatment goals include the following:
- To prevent corneal complications, ie, ulceration, melting, and perforation
- To promote epithelial healing and nerve regeneration
- To restore corneal integrity and visual function
Stepwise Approach to Neurotrophic Keratitis Treatment
Patient with suspected neurotrophic keratitis
↓
Corneal sensation testing + Slit lamp examination
↓
STAGE 1: Mild neurotrophic keratitis (punctate epithelial defects)
| → Lubrication (preservative-free tears, ointments)
| → Autologous serum/PRP drops
| → Punctal occlusion
| → Discontinue toxic medications (preservatives, anesthetics)
↓
STAGE 2: Moderate neurotrophic keratitis (PEDs)
| → NGF therapy
| → AMT (PROKERA®)
| → Scleral/bandage contact lens
| → Tarsorrhaphy (if severe)
↓
STAGE 3: Severe neurotrophic keratitis (Corneal ulceration, stromal melting)
| → Urgent amniotic membrane graft
| → Corneal neurotization surgery (if applicable)
| → Boston KPro (end-stage) [105]
Table 7. Treatment Planning Based on Severity
|
Stage |
Key Clinical Features |
Treatment Options |
|
Stage 1 (Mild) |
Punctate epithelial erosions, tear film instability |
- Preservative-free lubricants - Autologous serum drops - Punctal occlusion - Moisture goggles |
|
Stage 2 (Moderate) |
PEDs, mild stromal involvement |
- Cenegermin - AMT - Scleral or bandage contact lenses - Tarsorrhaphy (partial) |
|
Stage 3 (Severe) |
Corneal ulceration, stromal melting, risk of perforation |
- Urgent amniotic membrane graft - Corneal neurotization surgery - Conjunctival flap surgery - Keratoplasty (if with perforation) |
Surgical Intervention Decision Pathway
Presence of PEDs?
↓
NO → Continue conservative therapy (NGF, lubrication)
↓
YES → Consider AMT or scleral lens
↓
Ulceration or stromal melting?
↓
NO → Monitor with NGF therapy
↓
YES → Consider corneal neurotization or keratoplasty
↓
End-stage corneal disease?
↓
YES → Consider Boston KPro [106]
Key Takeaways
Early intervention is critical in preventing the progression of neurotrophic keratitis to corneal perforation. NGF therapy and AMT serve as the foundation for treating moderate cases of neurotrophic keratitis. In severe cases, surgical interventions such as tarsorrhaphy, corneal neurotization, and keratoplasty should be considered. An interprofessional approach involving ophthalmologists, neurologists, and other specialists ensures improved patient outcomes by addressing all aspects of the condition. Structured treatment planning is essential for managing neurotrophic keratitis effectively and preventing vision-threatening complications.
Toxicity and Adverse Effect Management
Neurotrophic keratitis management involves various pharmacological and surgical interventions, some of which have associated toxicities and adverse effects. Recognizing and mitigating these effects are crucial to optimizing treatment outcomes.
Table 8. Adverse Effects of Pharmacologic Therapies
|
Therapy |
Common Adverse Effects |
Management Strategies |
|
Cenegermin (Oxervate™) |
Eye pain, conjunctival hyperemia, increased tearing |
Dose adjustment, topical lubricants for comfort |
|
Autologous serum or PRP drops |
Contamination risk, microbial keratitis |
Proper storage at -20 °C, aseptic handling |
|
Long-term lubricant use, gel or ointment-based |
Blurry vision, preservative toxicity if not preservative-free |
Use preservative-free formulations, adjust viscosity |
|
Prolonged corticosteroid use |
Delayed wound healing, increased intraocular pressure (IOP), secondary infections |
Use the lowest effective dose, monitor IOP |
|
Tetracyclines (doxycycline for corneal melting prevention) |
Gastrointestinal upset, photosensitivity |
Take with food, avoid excessive sun exposure |
|
Topical nonsteroidal anti-inflammatory drugs |
Corneal thinning, delayed epithelial healing |
Limit use, prefer lubricants or neurotrophic support |
Table 9. Toxicities of Surgical and Biological Interventions
|
Intervention |
Potential Complications |
Management Approach |
|
Amniotic Membrane Transplantation |
Dislodgement, infection |
Frequent lubrication, careful placement |
|
Scleral or bandage contact lenses |
Hypoxia-related keratitis, microbial infection |
Proper hygiene, frequent follow-up |
|
Tarsorrhaphy (eyelid closure) |
Eyelid malposition, cosmetic concerns |
Partial tarsorrhaphy in selected cases |
|
Corneal neurotization surgery |
Donor site morbidity, nerve dysfunction |
Careful nerve selection, postoperative monitoring |
|
Boston KPro |
Graft failure, chronic inflammation |
Lifelong antibiotic prophylaxis, close monitoring |
Strategies for Managing Toxicities and Adverse Effects
Monitoring for early signs of adverse effects is crucial during neurotrophic keratitis treatment. Regular corneal examinations and IOP monitoring help identify steroid-related complications. Additionally, frequent corneal sensitivity testing is important for patients undergoing long-term neurotrophic keratitis therapy to detect any changes in corneal nerve function.[107]
To reduce medication-related toxicity, preservative-free lubricants should be preferred to minimize irritation. The chronic use of nonsteroidal anti-inflammatory drugs (NSAIDs) or anesthetics should be avoided, as these agents can exacerbate epithelial damage.[108]
Optimizing surgical interventions is also key. Scleral contact lenses should be closely monitored for the risk of corneal hypoxia and infection. If amniotic membrane grafts show signs of nonadherence or infection, they should be replaced promptly to prevent further complications.[109]
Patient education plays an essential role in treatment adherence. Patients should be encouraged to comply with aseptic techniques when using serum eye drops. Before initiating aggressive therapies, the risks and benefits should be thoroughly discussed with the patient.[110]
Key Takeaways
Adverse effects of neurotrophic keratitis treatments must be anticipated and managed proactively. Regular follow-up is critical to detect complications early. A personalized approach that balances treatment efficacy with potential toxicities ensures the best outcomes. Addressing toxicity risks early improves treatment adherence, leading to better visual outcomes for patients with neurotrophic keratitis.
Staging
Staging neurotrophic keratitis is essential for guiding treatment and predicting disease progression. The most widely used classification system is the Mackie Classification, which divides the disorder into 3 stages based on corneal involvement and severity (see Table 10. Mackie Classification of Neurotrophic Keratitis).
Table 10. Mackie Classification of Neurotrophic Keratitis
|
Stage |
Clinical Features |
Key Findings |
|
Stage 1 (Mild) |
Corneal epithelial irregularities, punctate keratopathy, tear film instability |
Corneal sensitivity reduced, early fluorescein staining |
|
Stage 2 (Moderate) |
PEDs, stromal thinning, rolled edges |
Epithelial defect without infiltrate, mild haze |
|
Stage 3 (Severe) |
Corneal ulceration, stromal melting, perforation risk |
Descemetocele formation, possible secondary infection |
Visual Representation of Neurotrophic Keratitis Staging
Mild neurotrophic keratitis is characterized by dry corneal epithelium, punctate erosions, and early loss of corneal sensitivity. Moderate neurotrophic keratitis demonstrates a PED accompanied by epithelial thinning and mild stromal involvement. Stage 3 represents severe neurotrophic keratitis, marked by ulceration, stromal melting, and a heightened risk of corneal perforation.
Table 11. Clinical and Management Implications Based on Staging
|
Stage |
Primary Management |
|
Stage 1 |
Lubrication, preservative-free artificial tears, autologous serum drops |
|
Stage 2 |
AMT, cenegermin, scleral lenses |
|
Stage 3 |
Urgent surgical intervention with tarsorrhaphy, corneal neurotization, or keratoplasty |
Key Takeaways
Early-stage neurotrophic keratitis should be managed with lubrication and neurotrophic support to maintain corneal integrity and prevent disease progression. In moderate cases, biological therapies, such as NGF and AMT, along with corneal protection strategies, become essential to promote healing. Severe neurotrophic keratitis requires urgent intervention to prevent corneal perforation and irreversible damage. Staging is vital in guiding personalized treatment, ensuring timely intervention, and reducing the risk of vision loss.
Prognosis
The prognosis of neurotrophic keratitis depends on early diagnosis, disease severity, and timely treatment. The condition progresses without intervention, resulting in epithelial breakdown, corneal ulceration, stromal melting, and eventual perforation, resulting in severe vision loss. Advances in treatment, including NGF therapy, AMT, and corneal neurotization, have significantly improved outcomes.
Table 12. Prognosis Based on Staging
|
Stage |
Prognosis |
Risk Factors for Poor Outcome |
|
Stage 1 (Mild) |
Good prognosis with timely lubrication, NGF therapy, and tear film support |
Delayed diagnosis, continued use of toxic eye drops |
|
Stage 2 (Moderate) |
Variable prognosis, with risk of progression, if PED is untreated |
Inadequate corneal protection, noncompliance with treatment |
|
Stage 3 (Severe) |
Poor prognosis, high risk of corneal perforation, and permanent vision loss |
Late-stage disease, corneal melting, secondary infections |
Factors Influencing Prognosis
Timely diagnosis and treatment enhance corneal healing rates, reducing the risk of disease progression. NGF therapy improves corneal sensation and lowers the likelihood of worsening pathology. Corneal neurotization surgery offers long-term nerve restoration, supporting sustained epithelial healing. Adherence to treatment plays a critical role in preventing complications, as poor compliance can hinder recovery. Systemic conditions, including diabetes, HZO, and autoimmune diseases, negatively impact prognosis by further compromising corneal health.
With optimal management, many patients retain functional vision. However, untreated cases face a high risk of irreversible vision loss due to corneal scarring, perforation, or end-stage keratopathy. Emerging therapies, such as gene therapy and regenerative medicine, offer the potential for nerve restoration and visual recovery. Early diagnosis and a proactive, multimodal treatment strategy remain essential for preserving vision and preventing complications in neurotrophic keratitis.
Complications
Neurotrophic keratitis is a progressive degenerative corneal disease that, if left untreated, can lead to severe complications such as vision loss and corneal perforation. These complications result from corneal anesthesia, impaired wound healing, and PEDs.
Table 13. Major Complications of Neurotrophic Keratitis
|
Complication |
Pathophysiology |
Clinical Impact |
|
Persistent Epithelial Defects |
Failure of corneal epithelium to regenerate due to impaired innervation |
Chronic inflammation and further corneal decompensation |
|
Corneal Ulceration |
Stromal degradation due to prolonged epithelial defects |
Increased risk of microbial infections |
|
Stromal Melting (Keratolysis) |
Uncontrolled MMP activity and inflammation |
Progressive stromal thinning and structural instability |
|
Corneal Perforation |
Advanced stromal melting leads to full-thickness corneal rupture |
Urgent surgical intervention, ie, keratoplasty, cyanoacrylate glue, or tarsorrhaphy, required |
|
Scarring and Opacification |
Chronic epithelial breakdown and secondary fibrosis |
Permanent visual impairment |
|
Infectious Keratitis |
Opportunistic bacterial or fungal infections in ulcerated corneas |
Can lead to endophthalmitis and globe loss if untreated |
|
Neovascularization |
Chronic hypoxia and inflammation, promoting abnormal blood vessel growth |
May cause visual distortion, increased corneal edema |
Risk Factors for Developing Severe Complications
Delayed diagnosis allows the disease to advance, leading to irreversible damage. Poor adherence to treatment increases the likelihood of persistent corneal defects and secondary infections. Diabetes and herpetic disease heighten the risk of progressive stromal melting and ulceration. Chronic use of topical anesthetics or preservative-containing eye drops further exacerbates corneal toxicity.
Preventing Complications in Neurotrophic Keratitis
Early detection and aggressive lubrication therapy help prevent disease progression. NGF therapy enhances corneal healing and sensation. AMT supports the resolution of PEDs. In severe cases, surgical interventions, including tarsorrhaphy, neurotization, and keratoplasty, may be necessary.[111] Timely intervention and comprehensive management are essential to preventing vision-threatening complications and preserving corneal integrity in neurotrophic keratitis.
Postoperative and Rehabilitation Care
Postoperative care in neurotrophic keratitis is crucial for optimizing corneal healing, preventing complications, and improving visual outcomes. Patients undergoing surgical or advanced medical interventions require long-term monitoring and rehabilitation strategies to maintain corneal integrity.
General Postoperative Guidelines
Frequent follow-up is essential for monitoring recovery and detecting complications. During the 1st week, daily assessments help evaluate epithelial healing and identify signs of infection or inflammation. Weekly follow-ups throughout the 1st month allow for monitoring graft integration, neurotization success, or keratoplasty healing. Long-term follow-up, ranging from monthly to quarterly visits, is necessary depending on disease severity.
Protecting the ocular surface supports healing and prevents further damage. Preservative-free lubricants, including artificial tears and ointments, help maintain hydration, while moisture goggles reduce tear evaporation. Punctal occlusion enhances tear film retention, improving overall ocular surface stability.
Preventing infection and inflammation is critical in postoperative management. Topical antibiotics may be required if epithelial defects persist. However, prolonged use of corticosteroids should be avoided due to the risk of delayed healing.[112]
Table 14. Postoperative Care Based on Surgical Interventions
|
Procedure |
Postoperative Management |
Key Considerations |
|
Amniotic Membrane Transplant |
The membrane should be removed after 7–14 days, and infection should be monitored. |
Lubricants should be applied to support healing. |
|
Corneal Neurotization Surgery |
Nerve function and corneal sensitivity should be assessed over several months. |
Nerve recovery is expected within 3–6 months. |
|
Tarsorrhaphy |
Partial release should be performed if healing is complete, and prolonged closure should be avoided. |
Temporary botulinum toxin-induced ptosis should be considered if necessary. |
|
Boston KPro |
Lifelong topical antibiotics should be administered, and regular monitoring for corneal melt should be conducted. |
Retroprosthetic membrane formation should be monitored. |
Visual Rehabilitation Strategies
Optical rehabilitation is crucial in protecting the ocular surface and enhancing visual function. Scleral contact lenses improve corneal hydration while providing a protective barrier. Rigid gas-permeable lenses help manage irregular astigmatism that may develop after surgery. For patients with severe visual impairment, low-vision aids such as magnifiers and specialized filters can enhance daily functioning.
Monitoring and supporting nerve function are essential for long-term corneal health. Periodic corneal sensitivity testing through esthesiometry helps track nerve recovery. In some cases, nerve stimulation therapy may be considered to improve neurotization outcomes.
Long-Term Follow-Up and Patient Education
Adherence to topical therapy and lubrication remains a cornerstone of postoperative care. Patients should be educated on recognizing signs of recurrence, including foreign body sensation and reduced vision. Psychosocial support can help improve the quality of life of individuals experiencing severe visual disability. A structured rehabilitation program is essential for preserving corneal integrity and optimizing visual outcomes.
Consultations
Managing neurotrophic keratitis requires an interprofessional approach due to the complex interplay between ocular and systemic factors. Collaboration among specialists ensures comprehensive care that addresses underlying conditions, optimizes treatment, and prevents complications.
Ophthalmologists, particularly cornea specialists, play a central role in diagnosing and managing neurotrophic keratitis through medical therapies and surgical interventions. Neurologists contribute by identifying trigeminal nerve dysfunction and associated neurodegenerative diseases, particularly in cases involving cranial nerve deficits, stroke, or postneurosurgical complications. Endocrinologists help manage diabetic neuropathy-associated neurotrophic keratitis, particularly in patients with poor glycemic control. Rheumatologists assess for systemic autoimmune conditions such as Sjögren syndrome and SLE when autoimmune diseases contribute to corneal pathology.
Oculoplastic surgeons address eyelid malposition and exposure keratopathy, offering procedures such as tarsorrhaphy or gold weight implantation when mechanical protection of the cornea is necessary. Infectious disease specialists manage cases complicated by herpetic, bacterial, or fungal infections, particularly in patients with recurrent HSV or VZV infection or microbial keratitis. Low vision specialists provide rehabilitation strategies for individuals with permanent visual impairment.
When to Refer?
In early-stage neurotrophic keratitis, ophthalmologists oversee medical management, including lubricants, NGF therapy, and AMT. Progressive disease requires referral to a cornea specialist for advanced interventions such as scleral lenses, tarsorrhaphy, and corneal neurotization. When systemic conditions contribute to disease progression, appropriate management requires referral to neurology, endocrinology, or rheumatology. Severe or end-stage cases may require surgical intervention, including Boston KPro, which falls under the expertise of a corneal surgeon.[113]
Key Takeaways
Early interprofessional consultation improves patient outcomes by addressing both ocular and systemic factors. Long-term control of neurotrophic keratitis relies on the effective management of underlying systemic diseases. Close collaboration among cornea specialists, neurologists, and endocrinologists enhances therapeutic success, ensuring a comprehensive and patient-centered approach to care.[114]
Deterrence and Patient Education
Patient education is critical in preventing complications and ensuring effective management of neurotrophic keratitis. Patients may not recognize early symptoms due to reduced or absent corneal sensation, leading to delayed presentation and a worsened prognosis. Providing clear, structured education helps patients take an active role in their care.
Patients should understand that neurotrophic keratitis results from corneal nerve dysfunction, which impairs wound healing and increases the risk of corneal ulcers. Since pain perception is diminished, patients should monitor for signs such as redness, blurry vision, or increased discharge rather than relying on pain as an indicator of disease progression.
Regular ophthalmic follow-ups are essential, even in mild cases, as early intervention can prevent corneal damage and perforation. Lubrication therapy remains a cornerstone of management. Patients should use preservative-free artificial tears and avoid over-the-counter drops containing preservatives, which may exacerbate the condition.
People with neurotrophic keratitis have a reduced blink reflex and corneal sensation, making them more susceptible to ocular trauma. These individuals should avoid eye rubbing, excessive screen time, and exposure to dry environments. Strict adherence to prescribed treatments, such as NGF therapy, autologous serum drops, and AMT, is necessary to optimize healing and reduce disease progression. Moisture chamber glasses or wraparound goggles can help prevent ocular surface desiccation by shielding the eyes from environmental stressors.
Routine eye exams are crucial, as early-stage neurotrophic keratitis often presents without noticeable symptoms. Patients should strictly avoid topical anesthetics and preservatives, which can worsen corneal damage. Any signs of blurred vision, discharge, or a foreign body sensation require immediate medical attention. Lifestyle modifications such as maintaining proper hydration, using humidifiers, and limiting screen exposure support ocular surface health.[115]
Providing structured educational resources enhances patient engagement. Flowcharts outlining disease progression can help patients recognize early warning signs, while instructional videos on proper eye drop techniques improve treatment adherence. Handouts detailing neurotrophic keratitis-friendly lifestyle changes, including environmental modifications and dietary recommendations for nerve health, further support disease management. Empowering patients with knowledge promotes adherence, encourages early intervention, and leads to better long-term outcomes in neurotrophic keratitis management.
Pearls and Other Issues
The prognosis of neurotrophic keratitis depends on the cause and extent of trigeminal nerve damage and the presence of coexisting ocular surface disease. Greater corneal sensory impairment increases the risk of disease progression. Current treatments focus on halting further deterioration, but none effectively restore corneal sensation or visual acuity. However, topical NGF derivatives and the ergoline compound nicergoline show potential in improving corneal sensitivity, offering significant benefits for patients who do not respond to standard therapy.[116]
Key Clinical Pearls
Neurotrophic keratitis is a progressive degenerative corneal disease resulting from corneal nerve dysfunction, thus impairing wound healing. Loss of corneal sensation is a defining feature, making corneal sensitivity testing essential in suspected cases. Early intervention remains critical, as the disease progresses silently and often without pain, leading to corneal ulceration and perforation. Neurotrophic keratitis is frequently misdiagnosed as DED or exposure keratopathy, emphasizing the need to rule out systemic conditions, such as autoimmune disease, diabetes, and HSV or VZV infection. Advances in treatment, including cenegermin, AMT, and corneal neurotization, have significantly improved disease management.
Common Pitfalls in Neurotrophic Keratitis Management
Misdiagnosing neurotrophic keratitis as DED is a common pitfall. However, patients with corneal anesthesia do not experience the classic symptoms of burning or foreign body sensation. Corneal sensitivity testing should always be performed in the presence of overlapping symptoms.[117]
Overuse of preservative-containing eye drops poses another risk, as prolonged use of preserved artificial tears or medications, such as glaucoma drops, can worsen epithelial toxicity. Switching to preservative-free lubricants helps minimize ocular surface damage.
Delaying surgical interventions can lead to serious complications. Tarsorrhaphy, scleral lenses, and AMT should not be postponed when PEDs are found. Without timely intervention, neurotrophic keratitis can rapidly progress to corneal perforation.
Inappropriate use of topical steroids or NSAIDs further exacerbates the condition. Steroids delay epithelial healing and increase the risk of secondary infections, while NSAIDs contribute to corneal melting.[118]
Prevention Strategies
Early identification of high-risk patients, such as those with diabetes, a history of HSV or VZV infection, or prior neurosurgical procedures, is essential for preventing neurotrophic keratitis. Patient education is key to corneal protection, emphasizing strategies such as moisture goggles and punctal occlusion to maintain ocular surface health. Proactive use of neurotrophic therapies can help prevent disease progression, particularly in individuals with known risk factors. Regular monitoring of corneal healing is vital in patients with reduced corneal sensitivity to detect complications early and initiate timely interventions.[119]
Enhancing Healthcare Team Outcomes
Neurotrophic keratitis requires management by an interprofessional team that includes ophthalmologists, neurologists, endocrinologists, optometrists, rehabilitation specialists, and ophthalmology nurses. Clinicians should recognize that patients with neurotrophic keratitis rarely report symptoms, likely due to corneal anesthesia. However, some may present with redness and blurred vision, which can result from PEDs, corneal edema, or scarring.
A history of episodic redness and eye pain, as well as cutaneous blistering or scarring, may indicate prior herpetic infections. Other relevant risk factors include corneal trauma, surgery, chemical burns, long-term topical medication use, neurosurgical procedures, and diabetes. Once neurotrophic keratitis is suspected, prompt referral to an ophthalmologist is necessary, as early treatment helps prevent corneal injury from worsening.
Managing neurotrophic keratitis requires an interprofessional approach to optimize outcomes, prevent complications, and ensure effective long-term care. Ophthalmologists oversee primary diagnosis, staging, and treatment, including lubrication therapy, NGF administration, AMT, and surgical interventions. Neurologists evaluate neuropathic causes such as trigeminal nerve dysfunction or postneurosurgical complications.
Endocrinologists manage diabetes-related neurotrophic keratitis and optimize glycemic control. Optometrists provide vision rehabilitation and fit scleral lenses to protect the ocular surface. Rheumatologists assess systemic autoimmune conditions such as Sjögren and SLE, which can contribute to corneal pathology. Infectious disease specialists manage cases related to HSV, VZV, or microbial keratitis.
Low-vision specialists assist in rehabilitation for patients with severe visual impairment. Nurses and patient educators play crucial roles in ensuring medication adherence, proper lubrication therapy, and symptom monitoring.
Effective collaboration within the healthcare team is essential for improving outcomes in neurotrophic keratitis management. Early screening and diagnosis require standardized corneal sensitivity testing in high-risk populations, including individuals with diabetes, prior HSV or VZV infection, or a history of neurosurgical procedures. A coordinated interprofessional approach, facilitated through regular team meetings among ophthalmology, neurology, and endocrinology specialists, helps tailor patient-specific treatment plans. Integrating electronic health records allows seamless access to patient history across specialties.
Enhancing patient education and adherence to therapy is essential in preventing complications. Providing educational materials on self-care, including proper eye drop instillation and strategies to avoid ocular trauma, empowers patients to take an active role in managing their condition. Telemedicine follow-ups can help monitor treatment adherence and detect early signs of disease progression. Encouraging participation in clinical trials and emerging therapies, such as gene therapy and neurostimulation techniques, may further improve patient outcomes. A well-coordinated interprofessional healthcare team can significantly reduce complications, preserve visual function, and enhance the quality of life for individuals with neurotrophic keratitis.[120]
Media
(Click Image to Enlarge)
(Click Image to Enlarge)
(Click Image to Enlarge)
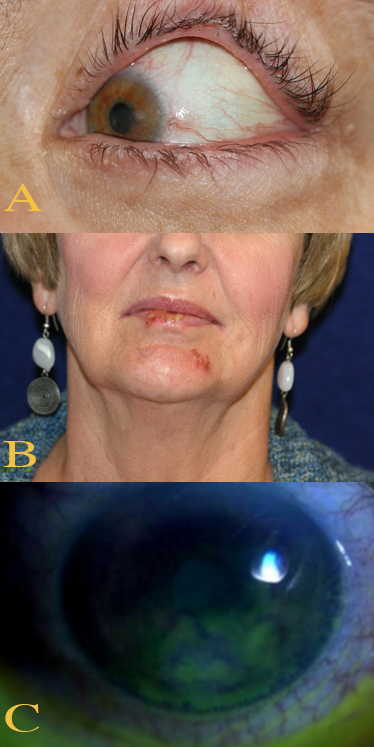
Ocular and Perioral Involvement in Herpes Simplex Virus Infection. Various clinical findings of herpetic keratitis are demonstrated. In image A (top), herpetic keratitis with neovascularization of the cornea is noted. In image B (middle), HSV type 1 lesions are seen on the patient's lips and chin. Image C (bottom) demonstrates findings of stage 1 neurotrophic keratitis, including fluorescein epithelial staining with Gaule spots, which are scattered areas of dried epithelium.
Contributed by BCK Patel, MD, FRCS
References
Semeraro F, Forbice E, Romano V, Angi M, Romano MR, Filippelli ME, Di Iorio R, Costagliola C. Neurotrophic keratitis. Ophthalmologica. Journal international d'ophtalmologie. International journal of ophthalmology. Zeitschrift fur Augenheilkunde. 2014:231(4):191-7. doi: 10.1159/000354380. Epub 2013 Oct 2 [PubMed PMID: 24107451]
Level 3 (low-level) evidenceVera-Duarte GR, Jimenez-Collado D, Kahuam-López N, Ramirez-Miranda A, Graue-Hernandez EO, Navas A, Rosenblatt MI. Neurotrophic keratopathy: General features and new therapies. Survey of ophthalmology. 2024 Sep-Oct:69(5):789-804. doi: 10.1016/j.survophthal.2024.04.004. Epub 2024 Apr 26 [PubMed PMID: 38679146]
Level 3 (low-level) evidenceShaheen BS, Bakir M, Jain S. Corneal nerves in health and disease. Survey of ophthalmology. 2014 May-Jun:59(3):263-85. doi: 10.1016/j.survophthal.2013.09.002. Epub 2014 Jan 23 [PubMed PMID: 24461367]
Level 3 (low-level) evidenceVersura P, Giannaccare G, Pellegrini M, Sebastiani S, Campos EC. Neurotrophic keratitis: current challenges and future prospects. Eye and brain. 2018:10():37-45. doi: 10.2147/EB.S117261. Epub 2018 Jun 28 [PubMed PMID: 29988739]
NaPier E, Camacho M, McDevitt TF, Sweeney AR. Neurotrophic keratopathy: current challenges and future prospects. Annals of medicine. 2022 Dec:54(1):666-673. doi: 10.1080/07853890.2022.2045035. Epub [PubMed PMID: 35243932]
Almeida J, Costa TR, Vivas M, Monteiro C, Vaz FT, Silva D, Vendrell C, Prieto I. Long-term Results of Topical Insulin Treatment for Persistent Corneal Epithelial Defects. Journal of ophthalmic & vision research. 2024 Dec:19(4):397-404. doi: 10.18502/jovr.v19i4.13977. Epub 2024 Dec 31 [PubMed PMID: 39917454]
Bu JB, Gericke A, Pfeiffer N, Wasielica-Poslednik J. Neurotrophic keratopathy: Clinical presentation and effects of cenegermin. American journal of ophthalmology case reports. 2022 Jun:26():101488. doi: 10.1016/j.ajoc.2022.101488. Epub 2022 Mar 16 [PubMed PMID: 35330588]
Level 3 (low-level) evidenceKoay SY, Larkin DFP. New Pharmacological Approaches for the Treatment of Neurotrophic Keratitis. Frontiers in pharmacology. 2022:13():796854. doi: 10.3389/fphar.2022.796854. Epub 2022 Mar 22 [PubMed PMID: 35392574]
Ahuja AS, Bowden FW 3rd, Robben JL. A Novel Treatment for Neurotrophic Corneal Ulcer Using Topical Cenegermin (OXERVATE™) Containing Recombinant Human Nerve Growth Factor. Cureus. 2020 Nov 27:12(11):e11724. doi: 10.7759/cureus.11724. Epub 2020 Nov 27 [PubMed PMID: 33391953]
Ozcan AA, Ulas B, Ciloglu E. Tarsorrhaphy with Sutureless Amnion Transplantation: Practical Management of Ocular Surface Pathologies. Beyoglu eye journal. 2022:7(4):313-319. doi: 10.14744/bej.2022.10327. Epub 2022 Nov 15 [PubMed PMID: 36628076]
Jabbour S, Ashton C, Balal S, Kaye A, Ahmad S. The management of neurotrophic keratitis. Current opinion in ophthalmology. 2021 Jul 1:32(4):362-368. doi: 10.1097/ICU.0000000000000766. Epub [PubMed PMID: 33966014]
Level 3 (low-level) evidenceBonini S, Rama P, Olzi D, Lambiase A. Neurotrophic keratitis. Eye (London, England). 2003 Nov:17(8):989-95 [PubMed PMID: 14631406]
Mantelli F, Nardella C, Tiberi E, Sacchetti M, Bruscolini A, Lambiase A. Congenital Corneal Anesthesia and Neurotrophic Keratitis: Diagnosis and Management. BioMed research international. 2015:2015():805876. doi: 10.1155/2015/805876. Epub 2015 Sep 16 [PubMed PMID: 26451380]
Ahmad B, Gurnani B, Patel BC. Herpes Simplex Keratitis. StatPearls. 2025 Jan:(): [PubMed PMID: 31424862]
Chen KH, Lin CY, Su SB, Chen KT. Leprosy: A Review of Epidemiology, Clinical Diagnosis, and Management. Journal of tropical medicine. 2022:2022():8652062. doi: 10.1155/2022/8652062. Epub 2022 Jul 4 [PubMed PMID: 35832335]
Liu CY, Arteaga AC, Fung SE, Cortina MS, Leyngold IM, Aakalu VK. Corneal neurotization for neurotrophic keratopathy: Review of surgical techniques and outcomes. The ocular surface. 2021 Apr:20():163-172. doi: 10.1016/j.jtos.2021.02.010. Epub 2021 Feb 26 [PubMed PMID: 33647470]
Dua HS, Ting DSJ, Al Saadi A, Said DG. Chemical eye injury: pathophysiology, assessment and management. Eye (London, England). 2020 Nov:34(11):2001-2019. doi: 10.1038/s41433-020-1026-6. Epub 2020 Jun 22 [PubMed PMID: 32572184]
Hannan C, Shoakazemi A, Quigley G. Microvascular Decompression for Trigeminal Neuralgia: A regional unit's experience. The Ulster medical journal. 2018 Jan:87(1):30-33 [PubMed PMID: 29588554]
Walker JL, McLaughlin J, Dickerson J, Pradhan SS, Newton FA. Unique Etiology of Trigeminal Neuralgia After Acute Ischemic Stroke. Kansas journal of medicine. 2023:16(2):185-186. doi: 10.17161/kjm.vol16.19500. Epub 2023 Jul 25 [PubMed PMID: 37539368]
Bikbova G, Oshitari T, Baba T, Bikbov M, Yamamoto S. Diabetic corneal neuropathy: clinical perspectives. Clinical ophthalmology (Auckland, N.Z.). 2018:12():981-987. doi: 10.2147/OPTH.S145266. Epub 2018 May 25 [PubMed PMID: 29872257]
Level 3 (low-level) evidenceDrissi I, Woods WA, Woods CG. Understanding the genetic basis of congenital insensitivity to pain. British medical bulletin. 2020 May 15:133(1):65-78. doi: 10.1093/bmb/ldaa003. Epub [PubMed PMID: 32219415]
Level 3 (low-level) evidenceZhang X, Vadoothker S, Munir WM, Saeedi O. Ocular Surface Disease and Glaucoma Medications: A Clinical Approach. Eye & contact lens. 2019 Jan:45(1):11-18. doi: 10.1097/ICL.0000000000000544. Epub [PubMed PMID: 30199425]
Datta S, Baudouin C, Brignole-Baudouin F, Denoyer A, Cortopassi GA. The Eye Drop Preservative Benzalkonium Chloride Potently Induces Mitochondrial Dysfunction and Preferentially Affects LHON Mutant Cells. Investigative ophthalmology & visual science. 2017 Apr 1:58(4):2406-2412. doi: 10.1167/iovs.16-20903. Epub [PubMed PMID: 28444329]
Stretton S, Jalbert I, Sweeney DF. Corneal hypoxia secondary to contact lenses: the effect of high-Dk lenses. Ophthalmology clinics of North America. 2003 Sep:16(3):327-40, v [PubMed PMID: 14564756]
Stern ME, Schaumburg CS, Dana R, Calonge M, Niederkorn JY, Pflugfelder SC. Autoimmunity at the ocular surface: pathogenesis and regulation. Mucosal immunology. 2010 Sep:3(5):425-42. doi: 10.1038/mi.2010.26. Epub 2010 May 19 [PubMed PMID: 20485329]
Negrini S, Emmi G, Greco M, Borro M, Sardanelli F, Murdaca G, Indiveri F, Puppo F. Sjögren's syndrome: a systemic autoimmune disease. Clinical and experimental medicine. 2022 Feb:22(1):9-25. doi: 10.1007/s10238-021-00728-6. Epub 2021 Jun 7 [PubMed PMID: 34100160]
Wakamatsu TH, Dos Santos MS, Barreiro TP, Sant'Anna AEBPP, Murta F, da Costa AX, Marculino LGC, de Alcântara RJA, de Farias CC, Gomes JÁP. Clinical Aspects of Stevens-Johnson Syndrome and Toxic Epidermal Necrolysis With Severe Ocular Complications in Brazil. Frontiers in medicine. 2021:8():649369. doi: 10.3389/fmed.2021.649369. Epub 2021 Jun 18 [PubMed PMID: 34222274]
Hsu T, Nguyen P, Petronic-Rosic V. A case of systemic lupus erythematosus with cutaneous granulomatous vasculitis. JAAD case reports. 2022 Mar:21():93-96. doi: 10.1016/j.jdcr.2022.01.006. Epub 2022 Jan 19 [PubMed PMID: 35198710]
Level 3 (low-level) evidenceSacchetti M, Lambiase A. Diagnosis and management of neurotrophic keratitis. Clinical ophthalmology (Auckland, N.Z.). 2014:8():571-9. doi: 10.2147/OPTH.S45921. Epub 2014 Mar 19 [PubMed PMID: 24672223]
Roth M, Dierse S, Alder J, Holtmann C, Geerling G. Incidence, prevalence, and outcome of moderate to severe neurotrophic keratopathy in a German tertiary referral center from 2013 to 2017. Graefe's archive for clinical and experimental ophthalmology = Albrecht von Graefes Archiv fur klinische und experimentelle Ophthalmologie. 2022 Jun:260(6):1961-1973. doi: 10.1007/s00417-021-05535-z. Epub 2022 Jan 6 [PubMed PMID: 34989864]
Kawai K, Yawn BP, Wollan P, Harpaz R. Increasing Incidence of Herpes Zoster Over a 60-year Period From a Population-based Study. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2016 Jul 15:63(2):221-6. doi: 10.1093/cid/ciw296. Epub 2016 May 8 [PubMed PMID: 27161774]
Ung L, Bispo PJM, Shanbhag SS, Gilmore MS, Chodosh J. The persistent dilemma of microbial keratitis: Global burden, diagnosis, and antimicrobial resistance. Survey of ophthalmology. 2019 May-Jun:64(3):255-271. doi: 10.1016/j.survophthal.2018.12.003. Epub 2018 Dec 24 [PubMed PMID: 30590103]
Level 3 (low-level) evidenceChaloulis SK, Mousteris G, Tsaousis KT. Incidence and Risk Factors of Bilateral Herpetic Keratitis: 2022 Update. Tropical medicine and infectious disease. 2022 Jun 7:7(6):. doi: 10.3390/tropicalmed7060092. Epub 2022 Jun 7 [PubMed PMID: 35736971]
McCormick I, James C, Welton NJ, Mayaud P, Turner KME, Gottlieb SL, Foster A, Looker KJ. INCIDENCE OF HERPES SIMPLEX VIRUS KERATITIS AND OTHER OCULAR DISEASE: GLOBAL REVIEW AND ESTIMATES. Ophthalmic epidemiology. 2022 Aug:29(4):353-362. doi: 10.1080/09286586.2021.1962919. Epub 2021 Oct 8 [PubMed PMID: 34622738]
Skarbez K, Priestley Y, Hoepf M, Koevary SB. Comprehensive Review of the Effects of Diabetes on Ocular Health. Expert review of ophthalmology. 2010 Aug 1:5(4):557-577 [PubMed PMID: 21760834]
Bandeira F, Yusoff NZ, Yam GH, Mehta JS. Corneal re-innervation following refractive surgery treatments. Neural regeneration research. 2019 Apr:14(4):557-565. doi: 10.4103/1673-5374.247421. Epub [PubMed PMID: 30632489]
Dietrich P, Dragatsis I. Familial Dysautonomia: Mechanisms and Models. Genetics and molecular biology. 2016 Oct-Dec:39(4):497-514. doi: 10.1590/1678-4685-GMB-2015-0335. Epub 2016 Aug 4 [PubMed PMID: 27561110]
Tu EY, Joslin CE, Sugar J, Shoff ME, Booton GC. Prognostic factors affecting visual outcome in Acanthamoeba keratitis. Ophthalmology. 2008 Nov:115(11):1998-2003. doi: 10.1016/j.ophtha.2008.04.038. Epub 2008 Jun 24 [PubMed PMID: 18571729]
Yu FX, Lee PSY, Yang L, Gao N, Zhang Y, Ljubimov AV, Yang E, Zhou Q, Xie L. The impact of sensory neuropathy and inflammation on epithelial wound healing in diabetic corneas. Progress in retinal and eye research. 2022 Jul:89():101039. doi: 10.1016/j.preteyeres.2021.101039. Epub 2022 Jan 4 [PubMed PMID: 34991965]
Ghafar NA, Jalil NAA, Kamarudin TA. Wound healing of the corneal epithelium: a review. Asian biomedicine : research, reviews and news. 2021 Oct:15(5):199-212. doi: 10.2478/abm-2021-0026. Epub 2021 Oct 29 [PubMed PMID: 37551323]
Levy A, Georgeon C, Knoeri J, Tourabaly M, Leveziel L, Bouheraoua N, Borderie VM. Corneal Epithelial Thickness Mapping in the Diagnosis of Ocular Surface Disorders Involving the Corneal Epithelium: A Comparative Study. Cornea. 2022 Nov 1:41(11):1353-1361. doi: 10.1097/ICO.0000000000003012. Epub 2022 Mar 30 [PubMed PMID: 35349542]
Level 2 (mid-level) evidenceVaidyanathan U, Hopping GC, Liu HY, Somani AN, Ronquillo YC, Hoopes PC, Moshirfar M. Persistent Corneal Epithelial Defects: A Review Article. Medical hypothesis, discovery & innovation ophthalmology journal. 2019 Fall:8(3):163-176 [PubMed PMID: 31598519]
Laibson PR. Recurrent corneal erosions and epithelial basement membrane dystrophy. Eye & contact lens. 2010 Sep:36(5):315-7. doi: 10.1097/ICL.0b013e3181f18ff7. Epub [PubMed PMID: 20724847]
Kamil S, Mohan RR. Corneal stromal wound healing: Major regulators and therapeutic targets. The ocular surface. 2021 Jan:19():290-306. doi: 10.1016/j.jtos.2020.10.006. Epub 2020 Oct 28 [PubMed PMID: 33127599]
Sun M, Chen S, Adams SM, Florer JB, Liu H, Kao WW, Wenstrup RJ, Birk DE. Collagen V is a dominant regulator of collagen fibrillogenesis: dysfunctional regulation of structure and function in a corneal-stroma-specific Col5a1-null mouse model. Journal of cell science. 2011 Dec 1:124(Pt 23):4096-105. doi: 10.1242/jcs.091363. Epub 2011 Dec 8 [PubMed PMID: 22159420]
Gurnani B, Kaur K, Agarwal S, Lalgudi VG, Shekhawat NS, Venugopal A, Tripathy K, Srinivasan B, Iyer G, Gubert J. Pythium insidiosum Keratitis: Past, Present, and Future. Ophthalmology and therapy. 2022 Oct:11(5):1629-1653. doi: 10.1007/s40123-022-00542-7. Epub 2022 Jul 5 [PubMed PMID: 35788551]
Shukla UV, Gurnani B, Kaufman EJ. Intraocular Hemorrhage. StatPearls. 2025 Jan:(): [PubMed PMID: 33620856]
Xiao N, Le QT. Neurotrophic Factors and Their Potential Applications in Tissue Regeneration. Archivum immunologiae et therapiae experimentalis. 2016 Apr:64(2):89-99. doi: 10.1007/s00005-015-0376-4. Epub 2015 Nov 26 [PubMed PMID: 26611762]
Kaur RP, Gurnani B, Kaur K. Intricate insights into immune response in dry eye disease. Indian journal of ophthalmology. 2023 Apr:71(4):1248-1255. doi: 10.4103/IJO.IJO_481_23. Epub [PubMed PMID: 37026255]
Alam J, de Paiva CS, Pflugfelder SC. Immune - Goblet cell interaction in the conjunctiva. The ocular surface. 2020 Apr:18(2):326-334. doi: 10.1016/j.jtos.2019.12.006. Epub 2020 Jan 14 [PubMed PMID: 31953222]
Burrow MK, Gurnani B, Patel BC. Keratoconjunctivitis. StatPearls. 2025 Jan:(): [PubMed PMID: 31194419]
Kaur K, Stokkermans TJ. Meibomian Gland Disease. StatPearls. 2025 Jan:(): [PubMed PMID: 35593799]
Liu Q,Smith CW,Zhang W,Burns AR,Li Z, NK cells modulate the inflammatory response to corneal epithelial abrasion and thereby support wound healing. The American journal of pathology. 2012 Aug; [PubMed PMID: 22728064]
Level 3 (low-level) evidenceJalbert I, Stapleton F, Papas E, Sweeney DF, Coroneo M. In vivo confocal microscopy of the human cornea. The British journal of ophthalmology. 2003 Feb:87(2):225-36 [PubMed PMID: 12543757]
Dixit R, Riviere J, Krishnan K, Andersen ME. Toxicokinetics and physiologically based toxicokinetics in toxicology and risk assessment. Journal of toxicology and environmental health. Part B, Critical reviews. 2003 Jan-Feb:6(1):1-40 [PubMed PMID: 12587252]
Fraunfelder FW. Corneal toxicity from topical ocular and systemic medications. Cornea. 2006 Dec:25(10):1133-8 [PubMed PMID: 17172885]
Hollander DA, Aldave AJ. Drug-induced corneal complications. Current opinion in ophthalmology. 2004 Dec:15(6):541-8 [PubMed PMID: 15523201]
Level 3 (low-level) evidenceRauchman SH, Locke B, Albert J, De Leon J, Peltier MR, Reiss AB. Toxic External Exposure Leading to Ocular Surface Injury. Vision (Basel, Switzerland). 2023 Apr 3:7(2):. doi: 10.3390/vision7020032. Epub 2023 Apr 3 [PubMed PMID: 37092465]
Leclercq B, Mejlachowicz D, Behar-Cohen F. Ocular Barriers and Their Influence on Gene Therapy Products Delivery. Pharmaceutics. 2022 May 6:14(5):. doi: 10.3390/pharmaceutics14050998. Epub 2022 May 6 [PubMed PMID: 35631584]
Wei Y, Asbell PA. The core mechanism of dry eye disease is inflammation. Eye & contact lens. 2014 Jul:40(4):248-56. doi: 10.1097/ICL.0000000000000042. Epub [PubMed PMID: 25390549]
Dart J. Corneal toxicity: the epithelium and stroma in iatrogenic and factitious disease. Eye (London, England). 2003 Nov:17(8):886-92 [PubMed PMID: 14631393]
Shih KC, Lam KS, Tong L. A systematic review on the impact of diabetes mellitus on the ocular surface. Nutrition & diabetes. 2017 Mar 20:7(3):e251. doi: 10.1038/nutd.2017.4. Epub 2017 Mar 20 [PubMed PMID: 28319106]
Level 1 (high-level) evidenceZhang X, M VJ, Qu Y, He X, Ou S, Bu J, Jia C, Wang J, Wu H, Liu Z, Li W. Dry Eye Management: Targeting the Ocular Surface Microenvironment. International journal of molecular sciences. 2017 Jun 29:18(7):. doi: 10.3390/ijms18071398. Epub 2017 Jun 29 [PubMed PMID: 28661456]
Mondal H, Kim HJ, Mohanto N, Jee JP. A Review on Dry Eye Disease Treatment: Recent Progress, Diagnostics, and Future Perspectives. Pharmaceutics. 2023 Mar 19:15(3):. doi: 10.3390/pharmaceutics15030990. Epub 2023 Mar 19 [PubMed PMID: 36986851]
Level 3 (low-level) evidenceStoner A, Harris A, Oddone F, Belamkar A, Verticchio Vercellin AC, Shin J, Januleviciene I, Siesky B. Topical carbonic anhydrase inhibitors and glaucoma in 2021: where do we stand? The British journal of ophthalmology. 2022 Oct:106(10):1332-1337. doi: 10.1136/bjophthalmol-2021-319530. Epub 2021 Aug 25 [PubMed PMID: 34433550]
Daiber HF, Gnugnoli DM. Visual Acuity. StatPearls. 2025 Jan:(): [PubMed PMID: 33085445]
Kaur K, Gurnani B. Slit-Lamp Biomicroscope. StatPearls. 2025 Jan:(): [PubMed PMID: 36512656]
Koh S, Rao SK, Srinivas SP, Tong L, Young AL. Evaluation of ocular surface and tear function - A review of current approaches for dry eye. Indian journal of ophthalmology. 2022 Jun:70(6):1883-1891. doi: 10.4103/ijo.IJO_1804_21. Epub [PubMed PMID: 35647953]
Crabtree JR, Tannir S, Tran K, Boente CS, Ali A, Borschel GH. Corneal Nerve Assessment by Aesthesiometry: History, Advancements, and Future Directions. Vision (Basel, Switzerland). 2024 May 12:8(2):. doi: 10.3390/vision8020034. Epub 2024 May 12 [PubMed PMID: 38804355]
Level 3 (low-level) evidenceBrott NR, Zeppieri M, Ronquillo Y. Schirmer Test. StatPearls. 2025 Jan:(): [PubMed PMID: 32644585]
Okada AA, Stanford M, Tabbara K. Ancillary testing, diagnostic/classification criteria and severity grading in Behçet disease. Ocular immunology and inflammation. 2012 Dec:20(6):387-93. doi: 10.3109/09273948.2012.723111. Epub 2012 Nov 19 [PubMed PMID: 23163622]
Gurnani B, Kaur K. Anti-infective therapies for Pythium insidiosum keratitis. Expert review of anti-infective therapy. 2024 Oct:22(10):805-817. doi: 10.1080/14787210.2024.2403146. Epub 2024 Sep 13 [PubMed PMID: 39268901]
Abdelghany AA, D'Oria F, Alio Del Barrio J, Alio JL. The Value of Anterior Segment Optical Coherence Tomography in Different Types of Corneal Infections: An Update. Journal of clinical medicine. 2021 Jun 27:10(13):. doi: 10.3390/jcm10132841. Epub 2021 Jun 27 [PubMed PMID: 34199039]
Tamhane M, Cabrera-Ghayouri S, Abelian G, Viswanath V. Review of Biomarkers in Ocular Matrices: Challenges and Opportunities. Pharmaceutical research. 2019 Jan 23:36(3):40. doi: 10.1007/s11095-019-2569-8. Epub 2019 Jan 23 [PubMed PMID: 30673862]
Cruzat A, Pavan-Langston D, Hamrah P. In vivo confocal microscopy of corneal nerves: analysis and clinical correlation. Seminars in ophthalmology. 2010 Sep-Nov:25(5-6):171-7. doi: 10.3109/08820538.2010.518133. Epub [PubMed PMID: 21090996]
Alhoutan K, Alarfaj K. Exposure Keratopathy: An Idiopathic Lagophthalmos Case Report. Cureus. 2021 Oct:13(10):e18945. doi: 10.7759/cureus.18945. Epub 2021 Oct 21 [PubMed PMID: 34812323]
Level 3 (low-level) evidenceLobo AM, Agelidis AM, Shukla D. Pathogenesis of herpes simplex keratitis: The host cell response and ocular surface sequelae to infection and inflammation. The ocular surface. 2019 Jan:17(1):40-49. doi: 10.1016/j.jtos.2018.10.002. Epub 2018 Oct 11 [PubMed PMID: 30317007]
Srinivas SP, Rao SK. Ocular surface staining: Current concepts and techniques. Indian journal of ophthalmology. 2023 Apr:71(4):1080-1089. doi: 10.4103/ijo.IJO_2137_22. Epub [PubMed PMID: 37026238]
Lopez Montes T, Gurnani B, Stokkermans TJ. Assessment of the Watery Eye. StatPearls. 2025 Jan:(): [PubMed PMID: 36508543]
Hussain S, Mubeen I, Ullah N, Shah SSUD, Khan BA, Zahoor M, Ullah R, Khan FA, Sultan MA. Modern Diagnostic Imaging Technique Applications and Risk Factors in the Medical Field: A Review. BioMed research international. 2022:2022():5164970. doi: 10.1155/2022/5164970. Epub 2022 Jun 6 [PubMed PMID: 35707373]
Cruzat A, Qazi Y, Hamrah P. In Vivo Confocal Microscopy of Corneal Nerves in Health and Disease. The ocular surface. 2017 Jan:15(1):15-47. doi: 10.1016/j.jtos.2016.09.004. Epub 2016 Oct 19 [PubMed PMID: 27771327]
Hagan S. Biomarkers of ocular surface disease using impression cytology. Biomarkers in medicine. 2017 Dec:11(12):1135-1147. doi: 10.2217/bmm-2017-0124. Epub 2017 Nov 28 [PubMed PMID: 29182030]
Lee YC, Kim SY. Treatment of neurotrophic keratopathy with nicergoline. Cornea. 2015 Mar:34(3):303-7. doi: 10.1097/ICO.0000000000000348. Epub [PubMed PMID: 25625360]
Pérez-Bartolomé F, Mingo Botín D, de Dompablo E, de Arriba P, Arnalich Montiel F, Muñoz Negrete FJ. Post-herpes neurotrophic keratopathy: Aetiopathogenesis, clinical signs and current therapies. Archivos de la Sociedad Espanola de Oftalmologia. 2019 Apr:94(4):171-183. doi: 10.1016/j.oftal.2019.01.002. Epub 2019 Feb 2 [PubMed PMID: 30718014]
Voelker R. New Drug Treats Rare, Debilitating Neurotrophic Keratitis. JAMA. 2018 Oct 2:320(13):1309. doi: 10.1001/jama.2018.14132. Epub [PubMed PMID: 30285163]
Scanzera AC, Shorter E. Case Series: Management of Neurotrophic Keratitis from Familial Dysautonomia. Optometry and vision science : official publication of the American Academy of Optometry. 2018 Aug:95(8):678-681. doi: 10.1097/OPX.0000000000001255. Epub [PubMed PMID: 30063663]
Level 2 (mid-level) evidenceLacorzana J, Neo YN, Maubon L, Sibley D, Ahmad S. Current and Emerging Therapeutic Strategies for the Management of Neurotrophic Keratitis. Drugs. 2025 Jan 23:():. doi: 10.1007/s40265-025-02147-3. Epub 2025 Jan 23 [PubMed PMID: 39843756]
Yao Y, Wang L, Ding J, Pan X, Yang L, Guo C, Wang Y, Gruber R, Nan K, Li L. Nerve growth factor loaded hypotonic eye drops for corneal nerve repair. Journal of controlled release : official journal of the Controlled Release Society. 2025 Apr 10:380():71-84. doi: 10.1016/j.jconrel.2025.01.080. Epub 2025 Feb 4 [PubMed PMID: 39884437]
Mohamed MT, Inoue D, Yoshimura S, Uematsu M, Mohamed YH, Kusano M, Tang D, Oishi A, Kitaoka T, Takeo G, Ohira A. Neuronal Intranuclear Inclusion Disease with a Corneal Disorder: A Case Report. Medicina (Kaunas, Lithuania). 2024 Oct 22:60(11):. doi: 10.3390/medicina60111730. Epub 2024 Oct 22 [PubMed PMID: 39596915]
Level 3 (low-level) evidenceMurugan SRB, Sanjay S, Somanath A, Mahendradas P, Patil A, Kaur K, Gurnani B. Artificial Intelligence in Uveitis: Innovations in Diagnosis and Therapeutic Strategies. Clinical ophthalmology (Auckland, N.Z.). 2024:18():3753-3766. doi: 10.2147/OPTH.S495307. Epub 2024 Dec 14 [PubMed PMID: 39703602]
Gurnani B, Kaur K. Navigating the challenges of infective keratitis: A critical analysis of treatment and diagnostic approaches. Indian journal of ophthalmology. 2024 Aug 1:72(8):1227-1228. doi: 10.4103/IJO.IJO_706_24. Epub 2024 Jul 29 [PubMed PMID: 39078975]
Golhait P, Peseyie R. Persistent Epithelial Defect. StatPearls. 2025 Jan:(): [PubMed PMID: 34424633]
Adams BS, Patel AR. Cenegermin. StatPearls. 2025 Jan:(): [PubMed PMID: 34424642]
Xia HQ, Jiang XD, Song YF, Li XM, Tian YJ. Hydrogel dressings on neurotrophic keratitis in an experimental animal model. International journal of ophthalmology. 2024:17(8):1396-1402. doi: 10.18240/ijo.2024.08.02. Epub 2024 Aug 18 [PubMed PMID: 39156779]
Level 3 (low-level) evidenceKumar A, Yun H, Funderburgh ML, Du Y. Regenerative therapy for the Cornea. Progress in retinal and eye research. 2022 Mar:87():101011. doi: 10.1016/j.preteyeres.2021.101011. Epub 2021 Sep 14 [PubMed PMID: 34530154]
Van Bentum RE, Van den Berg JM, Wolf SE, Van der Bijl J, Tan HS, Verbraak FD, van der Horst-Bruinsma IE. Multidisciplinary management of auto-immune ocular diseases in adult patients by ophthalmologists and rheumatologists. Acta ophthalmologica. 2021 Mar:99(2):e164-e170. doi: 10.1111/aos.14548. Epub 2020 Aug 4 [PubMed PMID: 32749781]
Gurnani B, Kaur K. Bacterial Keratitis. StatPearls. 2025 Jan:(): [PubMed PMID: 34662023]
Gurnani B, Narayana S, Christy J, Rajkumar P, Kaur K, Gubert J. Successful management of pediatric pythium insidiosum keratitis with cyanoacrylate glue, linezolid, and azithromycin: Rare case report. European journal of ophthalmology. 2022 Sep:32(5):NP87-NP91. doi: 10.1177/11206721211006564. Epub 2021 Mar 28 [PubMed PMID: 33779337]
Level 3 (low-level) evidenceGoyal S, Hamrah P. Understanding Neuropathic Corneal Pain--Gaps and Current Therapeutic Approaches. Seminars in ophthalmology. 2016:31(1-2):59-70. doi: 10.3109/08820538.2015.1114853. Epub [PubMed PMID: 26959131]
Level 3 (low-level) evidenceBonini S, Lambiase A, Rama P, Sinigaglia F, Allegretti M, Chao W, Mantelli F, REPARO Study Group. Phase II Randomized, Double-Masked, Vehicle-Controlled Trial of Recombinant Human Nerve Growth Factor for Neurotrophic Keratitis. Ophthalmology. 2018 Sep:125(9):1332-1343. doi: 10.1016/j.ophtha.2018.02.022. Epub 2018 Apr 10 [PubMed PMID: 29653858]
Level 1 (high-level) evidenceKanu LN, Ciolino JB. Nerve Growth Factor as an Ocular Therapy: Applications, Challenges, and Future Directions. Seminars in ophthalmology. 2021 May 19:36(4):224-231. doi: 10.1080/08820538.2021.1890793. Epub 2021 Feb 27 [PubMed PMID: 33641595]
Level 3 (low-level) evidenceMahmood AH, Alharbi AS, Almanea BA, Alsaati AF. Sutureless Amniotic Membrane (ProKera®) and Intravenous Immunoglobulin in the Management of Ocular Complications of Stevens-Johnson Syndrome-Toxic Epidermal Necrolysis Overlap. Cureus. 2021 Aug:13(8):e16989. doi: 10.7759/cureus.16989. Epub 2021 Aug 8 [PubMed PMID: 34540392]
Rashad R, Kwan JT, Shanbhag SS, Ngowyutagon P, Saeed M, Tahboub MA, Haseeb A, Chodosh J, Saeed HN. Long-term outcomes of glued (sutureless) amniotic membrane transplantation in acute Stevens-Johnson syndrome/toxic epidermal necrolysis: a comparative study. The British journal of ophthalmology. 2024 Oct 22:108(11):1508-1513. doi: 10.1136/bjo-2023-324076. Epub 2024 Oct 22 [PubMed PMID: 38490716]
Level 2 (mid-level) evidenceKhan L, Batavia E. Medroxyprogesterone to treat corneal thinning postcurvularia keratitis. Oman journal of ophthalmology. 2019 Sep-Dec:12(3):194-196. doi: 10.4103/ojo.OJO_228_2018. Epub 2019 Oct 11 [PubMed PMID: 31902997]
Sacchetti M, Lambiase A. Neurotrophic factors and corneal nerve regeneration. Neural regeneration research. 2017 Aug:12(8):1220-1224. doi: 10.4103/1673-5374.213534. Epub [PubMed PMID: 28966630]
Lee SH, Tseng SC. Amniotic membrane transplantation for persistent epithelial defects with ulceration. American journal of ophthalmology. 1997 Mar:123(3):303-12 [PubMed PMID: 9063239]
Wu A, Khawaja AP, Pasquale LR, Stein JD. A review of systemic medications that may modulate the risk of glaucoma. Eye (London, England). 2020 Jan:34(1):12-28. doi: 10.1038/s41433-019-0603-z. Epub 2019 Oct 8 [PubMed PMID: 31595027]
Saad J, Mathew D. Nonsteroidal Anti-Inflammatory Drugs Toxicity. StatPearls. 2024 Jan:(): [PubMed PMID: 30252262]
Harthan JS, Shorter E. Therapeutic uses of scleral contact lenses for ocular surface disease: patient selection and special considerations. Clinical optometry. 2018:10():65-74. doi: 10.2147/OPTO.S144357. Epub 2018 Jul 11 [PubMed PMID: 30319297]
Mehuys E, Delaey C, Christiaens T, Van Bortel L, Van Tongelen I, Remon JP, Boussery K. Eye drop technique and patient-reported problems in a real-world population of eye drop users. Eye (London, England). 2020 Aug:34(8):1392-1398. doi: 10.1038/s41433-019-0665-y. Epub 2019 Nov 5 [PubMed PMID: 31690823]
Dana R, Farid M, Gupta PK, Hamrah P, Karpecki P, McCabe CM, Nijm L, Pepose JS, Pflugfelder S, Rapuano CJ, Saini A, Gibbs SN, Broder MS. Expert consensus on the identification, diagnosis, and treatment of neurotrophic keratopathy. BMC ophthalmology. 2021 Sep 8:21(1):327. doi: 10.1186/s12886-021-02092-1. Epub 2021 Sep 8 [PubMed PMID: 34493256]
Level 3 (low-level) evidenceLinsey K. Use of an Eyelid Pressure Patch Concomitantly with a Decellularized Dehydrated Amniotic Membrane for Ocular Surface Disease Management. Ophthalmology and therapy. 2025 Mar:14(3):573-584. doi: 10.1007/s40123-025-01094-2. Epub 2025 Feb 3 [PubMed PMID: 39899264]
Qu Y, Peng R, Hu B, Ma S, Liu Z, Gu S, Hong J. A New Treatment for Recalcitrant Neurotrophic Keratopathy of Ocular Graft-Versus-Host Disease with Virus Infection. Ophthalmology and therapy. 2024 Feb:13(2):469-479. doi: 10.1007/s40123-023-00857-z. Epub 2023 Dec 6 [PubMed PMID: 38057555]
Feroze KB, Gurnani B, O'Rourke MC. Transient Loss of Vision. StatPearls. 2025 Jan:(): [PubMed PMID: 28613595]
Green SM, Tomaszewski C, Valente JH, Lo B, Milne K. Use of Topical Anesthetics in the Management of Patients With Simple Corneal Abrasions: Consensus Guidelines From the American College of Emergency Physicians. Annals of emergency medicine. 2024 May:83(5):477-489. doi: 10.1016/j.annemergmed.2024.01.004. Epub 2024 Feb 6 [PubMed PMID: 38323950]
Level 3 (low-level) evidenceGheorghe A, Rosoga AT, Mrini F, Vărgău I, Gherghiceanu F. Various therapies for ocular surface diseases. Romanian journal of ophthalmology. 2018 Jan-Mar:62(1):83-87 [PubMed PMID: 29796439]
Gurnani B, Christy J, Narayana S, Kaur K, Moutappa F. Corneal Perforation Secondary to Rosacea Keratitis Managed with Excellent Visual Outcome. Nepalese journal of ophthalmology : a biannual peer-reviewed academic journal of the Nepal Ophthalmic Society : NEPJOPH. 2022 Jan:14(27):162-167. doi: 10.3126/nepjoph.v14i1.36454. Epub [PubMed PMID: 35996914]
Ashena Z, Nanavaty MA, Bardan AS, Thaker R, Bascaran L. Prophylactic Use of Nonsteroidal Anti-Inflammatory Drugs after Cataract Surgery and Corneal Melt. Journal of current ophthalmology. 2021 Oct-Dec:33(4):485-491. doi: 10.4103/joco.joco_107_21. Epub 2022 Jan 6 [PubMed PMID: 35128199]
Levin MJ, Gershon AA, Dworkin RH, Brisson M, Stanberry L. Prevention strategies for herpes zoster and post-herpetic neuralgia. Journal of clinical virology : the official publication of the Pan American Society for Clinical Virology. 2010 May:48 Suppl 1(Suppl 1):S14-9. doi: 10.1016/S1386-6532(10)70004-4. Epub [PubMed PMID: 20510262]
DeMill DL, Hussain M, Pop-Busui R, Shtein RM. Ocular surface disease in patients with diabetic peripheral neuropathy. The British journal of ophthalmology. 2016 Jul:100(7):924-928. doi: 10.1136/bjophthalmol-2015-307369. Epub 2015 Oct 23 [PubMed PMID: 26500330]