Introduction
Wound healing is a tightly regulated, multiphase process involving an interplay between pro- and anti-inflammatory mediators, cellular and vascular growth factors, and mediators of phagocytosis and apoptosis. This complex system eliminates pathogens, promotes neovascularization, and regenerates new dermal and epidermal structures. The process involves a finely balanced remodeling phase, consisting of the consumption and deposition of cellular and extracellular materials. When this regulation is disturbed, or a genetic or environmental predisposition exists, suboptimal healing occurs, leading to atrophic, hypertrophic, or keloid scars.
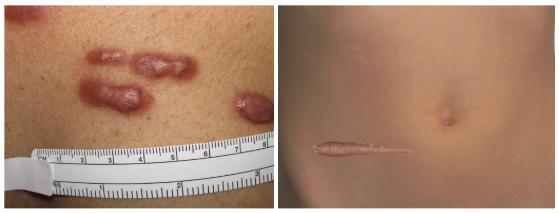
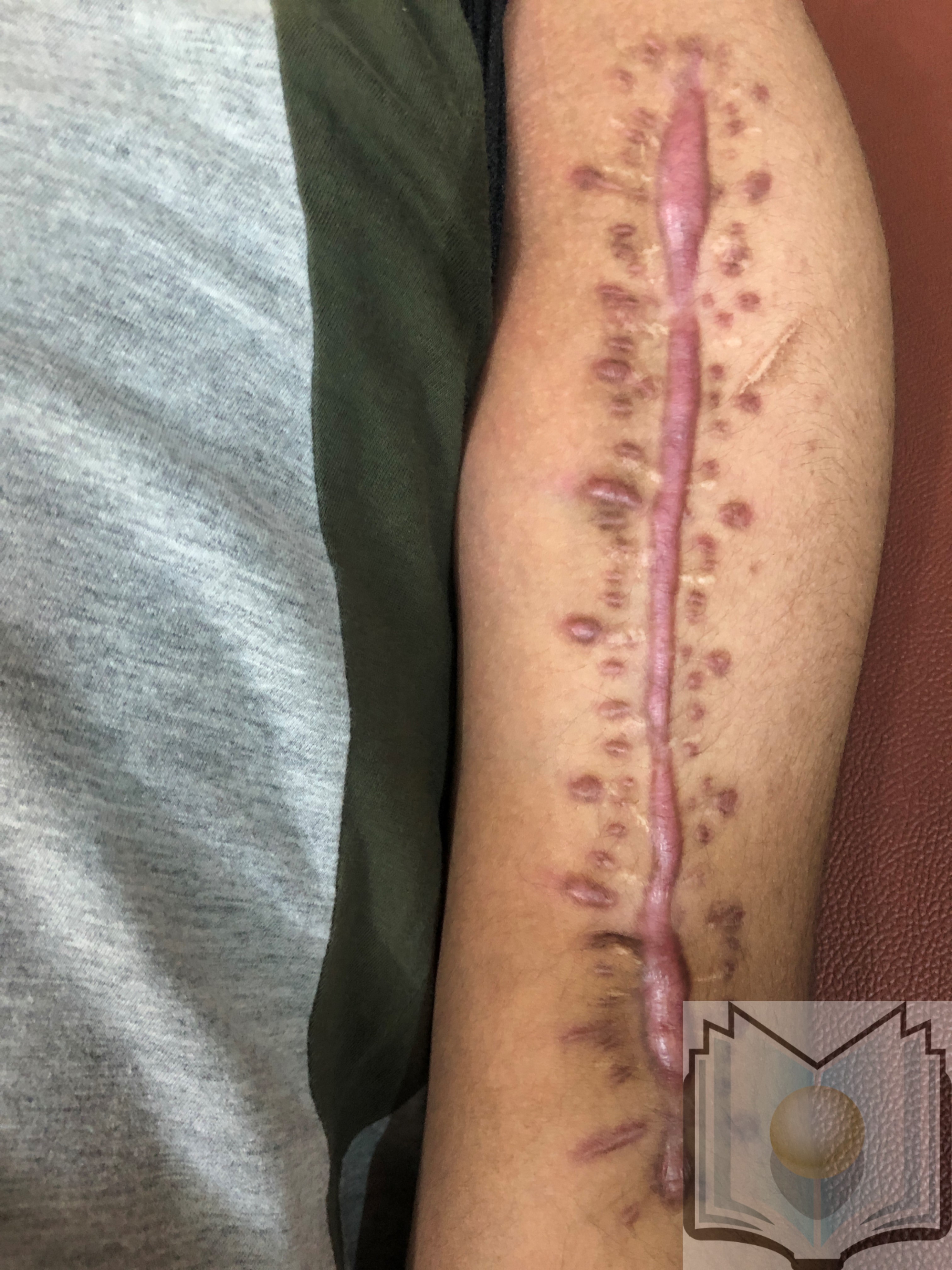
Hypertrophic scars and keloids are raised tissue formations resulting from excess fibrinogen and collagen production. Hypertrophic scars remain within the boundaries of the original wound and may regress over time (see Image. Hypertrophic Scars). In contrast, keloids extend beyond the borders of the initial injury and do not regress (see Image. Keloid Scars).[1] Both keloids and hypertrophic scars can be painful and pruritic, and these conditions can cause movement restrictions and disfigurement.[2] Histologically, hypertrophic scars show organized, wavy collagen, whereas keloids exhibit irregular and disorganized collagen deposition.[1] Recent studies have identified cellular, molecular, and genetic components that contribute to keloid formation, offering potential targets for preventative and therapeutic interventions. Individuals with known risk factors for keloid formation can benefit from preventative measures and the avoidance of unnecessary interventions.[3]
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
Keloids arise from dermal fibrosis and cutaneous scarring and are characterized by excess and chaotic extracellular matrix in the dermis of wound defects. This involves collagen fibroproliferation within the reticular dermis and heightened fibroblast activity.[4] Keloids occur due to increased production of collagen and extracellular matrix with insufficient degradation, following skin injuries that penetrate or damage the dermal layer. These injuries include burns, surgery, insect bites, tattoos, piercings, acne, or chickenpox.
Although the specific mechanisms are not fully understood, certain fibroblast subtypes and immune mediators have been identified as contributors to keloid formation by promoting excessive collagen deposition. Systemic proinflammatory conditions or genetic predispositions that increase collagen production may contribute to keloid formation, often accompanied by an increased presence of melanocytes.[5]
A genetic predisposition, combined with environmental factors, can trigger a keloid reaction. Sex hormones may elevate the risk by upregulating inflammation and collagen production, while hypertension has also been suggested as a risk factor, although its underlying mechanism remains unclear.[6] Additionally, mechanical forces, such as wound tension and stress, contribute to the risk of pathogenic scar formation.[7][8]
Epidemiology
Keloids may be genetically predisposed or occur sporadically, with a higher incidence during periods of hormonal fluctuation, such as puberty and pregnancy. They are more prevalent in Black, Hispanic, and Asian populations, with the incidence reaching as high as 16% in these groups.[9] The increased incidence of keloids in Black individuals may be associated with melanin-driven promotion of proinflammatory cytokines, including interleukins (ILs)-4 and IL13, which activate fibroblasts and the extracellular matrix, ultimately contributing to keloid formation.[10]
Keloids are more commonly observed in individuals aged 11 to 30, possibly due to increased cell turnover, greater skin tension, and a more active immune system. Familial cases of keloids have been identified, with incomplete penetrance and variable expression demonstrated.[11] Comorbidities associated with keloids include atopic dermatitis, rosacea, acne, and alopecia.[10][12][13]
Pathophysiology
The wound-healing process occurs in several phases—hemostasis, inflammation, proliferation, and remodeling. The inflammatory phase is crucial in determining the nature of the scar. During this phase, an imbalance between pro- and anti-inflammatory mediators leads to increased keloid formation, characterized by abnormal collagen deposition and disrupted extracellular matrix remodeling. Dysregulated fibroblasts contribute to the formation of irregular fibrotic lesions by promoting an imbalanced extracellular matrix.[14]
Fibrosis can develop during the early stages of wound healing if it is not corrected by subsequent remodeling. Disruptions affecting the dermis during healing, such as infection, additional injury, or an inflammatory response, can lead to excess tissue deposition.[1] Keloid tissue extends beyond the original wound, characterized by chaotic fibroblast and collagen proliferation within a milieu of proinflammatory cytokines.[3]
During the proliferative phase of healing, macrophages at the site of injury release growth factors, such as transforming growth factor-beta (TGF-β), which activate fibroblasts to produce collagen. TGF-β1 and TGF-β2 activate fibroblasts, whereas TGF-β3 inhibits them. Dysregulation of TGF-β signaling contributes to keloid formation. Overexpression of TGF-β1 and TGF-β2, coupled with decreased expression of TGF-β3, leads to increased fibroblast activity and excessive extracellular matrix production, resulting in abnormal scar formation. Collagen production increases by 20-fold in keloids.[1][15]
The conversion of fibroblasts into myofibroblasts is a key component of tissue repair, and dysregulation of this pathway contributes to excessive scarring. Fibroblast differentiation into myofibroblasts, triggered by TGF signaling, is essential for wound contraction. Myofibroblasts promote the expression of contractile alpha-smooth muscle actin, generate extracellular matrix secretion, and eventually undergo apoptosis during the remodeling phase. This process involves a delicate balance of collagen deposition and degradation, with any dysregulation leading to poor wound closure or excessive scarring.
Matrix metalloproteinases help minimize scarring by breaking down extracellular matrix components, including interstitial and denatured collagens, while facilitating the transition from collagen III to collagen I. The balance between matrix metalloproteinases and their inhibitors is partially regulated by fibroblasts in the dermal layers, which variably express matrix components.[14]
Keloids consist of disorganized collagen I and III with an excess of fibroblast proteins. Collagen deposition is exacerbated by hormonal influences and a proinflammatory state characterized by increased fibroblast activity, heightened proliferation of immune cells that promote fibrosis, and protease inhibition. Fibroblasts intrinsic to keloid formation are genetically distinct from normal fibroblasts and increase fibrosis, collagens I and III, and fibronectin. These fibroblasts are associated with reduced matrix metalloproteinases, increased growth factor receptors, and overexpression of TGF-β, which contributes to keloid angiogenesis.[14] The heterogeneity of macrophages contributes to keloid formation, influencing angiogenesis, collagen, and keratin deposition.[16]
In a study, the authors discovered bone marrow macrophage signaling led to the release of high levels of TGF-β from fibroblasts, resulting in increased scarring. Inhibiting this macrophage pathway reduced scarring. A serine protease that cleaves extracellular matrix proteins is postulated to promote TGF-β activation, leading to fibrosis and scarring. Mast cells containing the protease granzyme B were identified in individuals with hypertrophic scars and keloids but not in controls. When these mast cells degranulate, the protease is released extracellularly.[17][18] Another proposed mechanism of scar formation involves the expression of homeobox genes in response to mechanical forces on fibroblasts. The impact of these mechanical forces on gene expression may serve as a potential therapeutic target.[19]
Histopathology
Keloid histology is unique from other types of scars. These lesions contain a higher number of mast cells and an increased prevalence of epidermal keratinocytes, contributing to a thicker epidermal layer. The distinction between the reticular and papillary dermal layers is lost in keloid tissue, with an excess of fibroblasts and elevated glycosaminoglycans in the extracellular matrix.[14] The abundant, irregular collagen is arranged in whorls, in contrast to the parallel pattern seen in hypertrophic and normal scar tissue. Additionally, keloids exhibit both an increased ratio of type I to type III collagen and an overall higher collagen content compared to normal tissue.[20]
History and Physical
Keloid scars typically develop several weeks after an injury or surgery or may occur spontaneously, continuing to proliferate and often recurring after attempted excision.[21] The lesion may be associated with pain and pruritus. A family history may indicate a predisposition to keloid formation. The duration, change in size, and regression of the lesion help differentiate keloids from hypertrophic scars. Regression of the lesion suggests a hypertrophic scar, while prolonged duration and an increase in size indicate a keloid.[21]
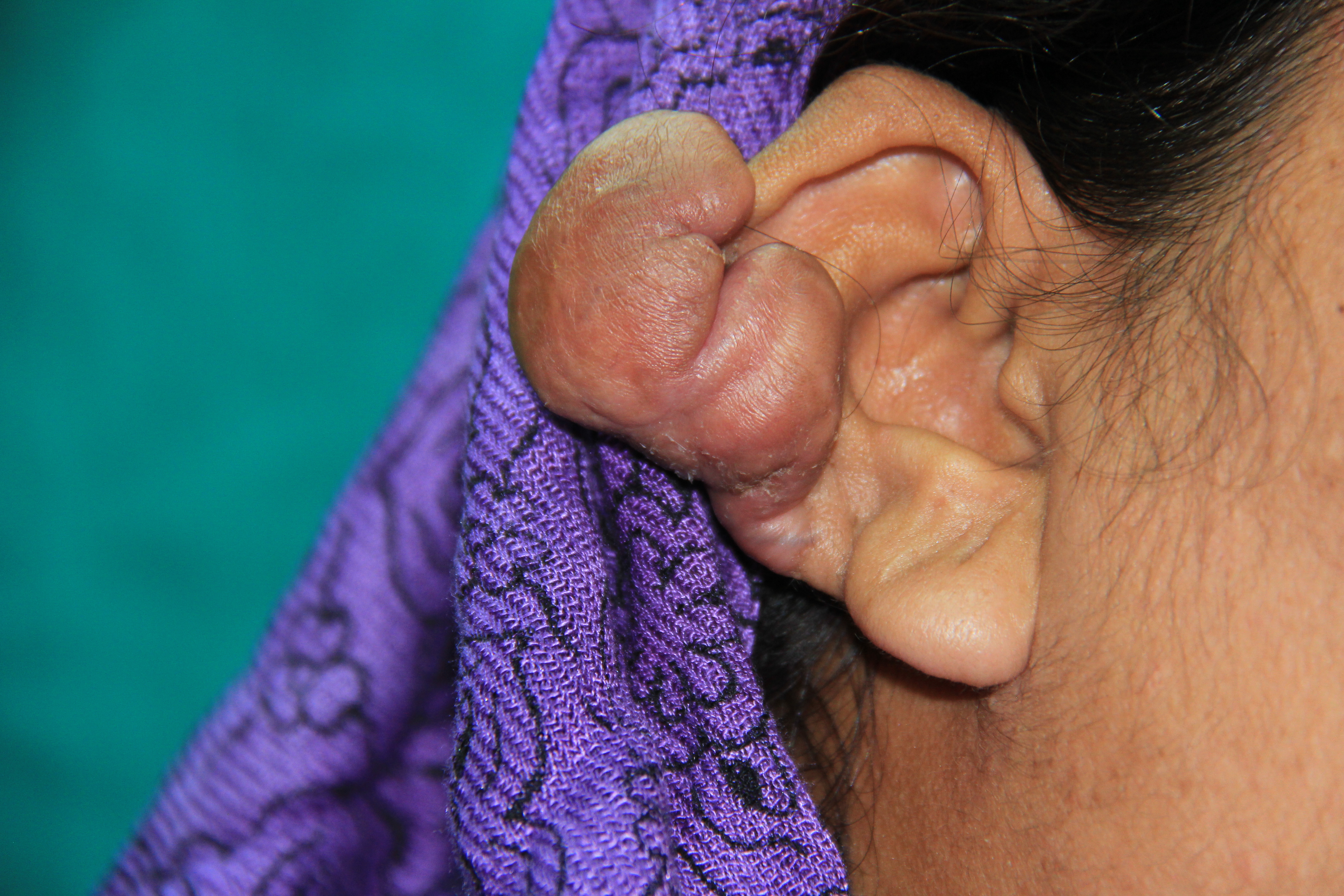
Keloids are commonly found on the earlobes, face, shoulders, chest, back, and knees, extending beyond the borders of the original injury (see Image. Earlobe Keloids). On examination, they present as a confluent, hairless, shiny area of raised, firm, or pliable scar tissue. The color may range from erythematous to dull red or pale, and the area may be indurated and tender. When crossing a joint, keloid tissue can restrict the full range of motion. Small projections into the neighboring skin may be observed. Keloid lesions on the face may be lobulated, while those on the trunk and extremities tend to be flattened or conical.[20][21]
Evaluation
The diagnosis of a keloid is often made through physical examination; however, a tissue biopsy may be performed if there is any uncertainty. Ultrasound or magnetic resonance imaging (MRI) may be used to further clarify the anatomy.[22] When diagnosing and planning treatment, laser speckle contrast imaging, which utilizes laser technology to visualize blood flow, can assess perfusion in the lesion and surrounding skin. High-frequency ultrasound provides full-thickness imaging of the skin, extending to the fascial layer, allowing for precise evaluation of lesion depth and vascularity. These imaging techniques can also track changes over time.
Other technologies that are not as commonly available include shear wave elastography, which measures tissue elasticity as a surrogate for stiffness using the dispersion of an acoustic radiation pulse. A 3-dimensional (3D) camera system can create high-definition images of scar morphology, which can be useful for monitoring changes in response to treatment. Although many scar assessment scales have been developed, their use is inconsistent, and they have not been standardized.[23][24]
Treatment / Management
Prevention is the cornerstone of treatment for individuals predisposed to keloid formation. When procedures are unavoidable, prophylactic therapies aimed at reducing aberrant fibroblast activity can decrease the likelihood of keloid development. Corticosteroid injections have been the primary method of preventive treatment. For individuals who sustain an injury or are diagnosed with a keloid following surgery, various options are available to reduce pain and pruritus, improve cosmesis, or fully excise the scar.[1][10] Treatment modalities range from noninvasive mechanical or topical approaches to pharmacotherapy injections, lasers, radiotherapy, and cryotherapy, as well as excision, either alone or in combination with other therapies. Most treatments focus on inhibiting fibroblast proliferation, collagen synthesis, or disrupting the blood supply to the keloid.[4](B2)
Occlusive dressings reduce collagen synthesis by limiting the delivery of oxygen and nutrients to the scar, making them most effective immediately after injury or surgery.[21] A recent meta-analysis of 15 studies revealed an unclear benefit of compression therapy on scar improvement, pain reduction, and quality of life.[25] Ultrasound therapy helps reduce the volume of keloid tissue by using mechanical vibrations to disrupt the keloid and promote tissue repair. The duration of ultrasound therapy required for clinical results varies based on the location and properties of the keloid. Cryotherapy increases the production of type III collagen and encourages the formation of more phenotypically normal fibroblasts. This therapy is particularly effective for smaller scars and is often part of a multimodal approach, which may include surgery or injections with corticosteroids or verapamil.[21] Liquid nitrogen, applied intralesionally or topically, is the standard agent, although some studies have used argon as a replacement.[4](A1)
Topical and injectable medications from various classes are commonly used to prevent and treat keloids. Pharmacotherapy can be used alone or in combination with other treatments. Topical corticosteroids and 5-fluorouracil creams are applied directly to the keloid, while interferon, bleomycin, and verapamil are injected intralesionally. Corticosteroids work by reducing collagen synthesis and proinflammatory mediators, promoting vasoconstriction, and inhibiting mitosis in keratinocytes and fibroblasts. Verapamil helps to break down collagen by inhibiting the extracellular matrix, while interferon reduces excessive collagen production and enhances collagenase activity by disrupting fibroblast proliferation.
Bleomycin, an antineoplastic agent, halts fibroblast proliferation, and 5-fluorouracil, an antimetabolite, interferes with DNA replication.[26] Topical formulations such as clobetasol (0.05%) are often applied daily, and intralesional corticosteroids such as triamcinolone (10-40 mg/mL) are used weekly or monthly for several months, either as monotherapy or in combination with other treatments. While most injectable therapies are administered with traditional syringes, innovations such as dissolving delivery systems are currently being researched.[1] (A1)
Surgical treatment involves excising the keloid scar with primary closure, ensuring a tension-free closure to minimize the risk of recurrence. While keloids have a high recurrence rate with excision alone, combining the procedure with steroids or other adjuvants, such as radiotherapy or interferon therapy, can significantly reduce the likelihood of recurrence.[1][21] Radiation therapy is commonly used after surgical excision, particularly for recurrent keloids. Standard external-beam radiation therapy is typically administered with a total dose of 20 to 25 Gy, divided into 3 fractions, starting the day after surgical resection. Recurrence rates following surgery combined with radiation vary widely. Although the radiation dose is relatively low, there is a risk of subsequent malignancy, which is a potential disadvantage. Brachytherapy has also been used postoperatively, although it is less commonly used.[21]
Emerging therapies for keloids include interferon, imiquimod, tacrolimus, retinoic acid, and botulinum toxin A. Interferon works in vitro by reducing collagen synthesis, while imiquimod induces apoptosis, and tacrolimus downregulates TGF-β receptors, thereby reducing fibroblast proliferation. Retinoids such as tretinoin and isotretinoin inhibit matrix metalloproteinases, which are overexpressed in keloids, and decrease collagen synthesis. Botulinum toxin A works by reducing wound tension, which leads to improved scarring.[1] Monoclonal antibodies, such as dupilumab, inhibit IL-4 and IL-13, which help in keloid formation. Many of these emerging therapies are used in conjunction with corticosteroids or excision and have been shown to reduce recurrence rates.
Additional treatments, such as extracorporeal shock wave therapy, have shown effectiveness comparable to intralesional corticosteroid injections in shrinking keloids. A trial combining radiofrequency with intralesional steroid injection demonstrated a significant reduction in lesion volume.[4][27] Colchicine was administered both before and after keloid excision in 10 subjects, with no reported recurrence. Additional post-excision treatments include topical imiquimod, porcine hydrogel, and hyperbaric oxygen. A novel technique involves creating micropunctures within the keloid to enhance drug penetration, while photobiomodulation has been used to improve the delivery and absorption of medications.[28](A1)
Laser therapy can be either ablative or non-ablative, targeting keloid blood vessels or fibroblasts while stimulating normal collagen deposition. This process helps reduce erythema, induration, and keloid growth. Different wavelengths of laser light inhibit various keloid components, such as fibroblasts or hemoglobin. Additionally, lasers can enhance the permeability of the epidermis, facilitating the delivery of topical medications. They are often combined with corticosteroids to improve drug delivery.[29] Lasers can be used as the primary treatment modality, as an adjunct to surgery to alleviate tension, or to assist in drug penetration into the lesion.
Carbon dioxide lasers, an ablative modality, are the most frequently used, while diode and pulsed dye lasers, which are non-ablative, are used less often. A study evaluating the efficacy of pulsed dye laser either alone or in combination with botulinum toxin A or triamcinolone found greater physician satisfaction in the treatment arms that included botulinum toxin or triamcinolone compared to pulsed dye laser alone.[4][30] Photodynamic therapy is another potential adjunctive treatment option.[27] (A1)
Differential Diagnosis
Differentiating malignancies from keloids is crucial, as steroids—the first-line therapy for keloids—are contraindicated in malignant tumors.[31] While most keloids are easily identifiable, some cases may present diagnostic uncertainty. For instance, malignant dermatofibrosarcoma protuberans can resemble a keloid phenotypically but is distinguishable through microscopy, which reveals pinwheel-shaped spindle cells and a honeycombed pattern of fibrous stroma. Immunohistochemistry will show the presence of vimentin, CD34, Bcl-2, HHF-35, and smooth muscle actin, markers that are absent in keloids.[20]
Trichilemmal carcinoma is a rare cutaneous adnexal malignant tumor originating from hair follicles, which can also resemble keloids. This condition is diagnosed by the presence of large polygonal keratinocytes with clear, glossy, periodic acid-Schiff-positive cytoplasm.[32] Keloidal basal cell carcinoma is characterized by prominent thick collagen bundles, and this can be mistaken for keloids as well. A case report by Nguyen et al described a child aged 9 who was initially diagnosed with a keloid and was later found to have a giant cell fibroblastoma, which is a malignant tumor.[32]
Cutaneous scleroderma presents with a thickened dermis and hyperpigmented, keloid-like lesions. A biopsy should be obtained if there is any diagnostic uncertainty. Microscopically, collagen bundles are parallel to the dermis, with fewer fibroblasts than in a keloid.[20] Sclerotic neurofibroma may also be mistaken for a keloid, but this lesion tests positive for the calcium-binding protein S100. Folliculitis can mimic the appearance of a keloid, and it is crucial to obtain a tissue diagnosis before initiating steroid therapy, as steroids may exacerbate the infection.[31]
Pertinent Studies and Ongoing Trials
An association between keloids and hypertension has been suggested in several studies.[33][34][35]
Prognosis
Keloids have a genetic predisposition, can continue to enlarge over an extended period, and do not typically regress. They also have a high rate of recurrence following treatment. The most effective therapeutic approach involves a combination of therapies that aim to minimize both appearance and symptoms. Adjuvant therapies should be incorporated after surgery to help prevent recurrence, and treatments often need to be repeated or managed chronically. The anatomical site of the keloid may also influence the prognosis.[36][37]
Complications
Keloids and hypertrophic scars can lead to prolonged pain and pruritus, are often disfiguring, and can be debilitating, leading to emotional and psychological distress for patients. Pruritus is the most common complication of keloids, followed by pain. When located over a joint, a keloid can restrict movement.[2] Many keloid treatments have adverse effects, including pigmentation changes, telangiectasia, and tissue atrophy, which are commonly seen with corticosteroids. Cryotherapy may lead to hyperpigmentation, blistering, and discomfort, whereas radiation therapy carries an increased risk of malignancy. Keloids are prone to recurrence, with the highest recurrence rate following surgical excision.[1]
Deterrence and Patient Education
The potential for functional disability and discomfort is significantly higher in keloids compared to normal scars. Individuals with a known genetic risk for keloid formation should be counseled before any surgical procedure about the risks of keloid development and strategies for prevention, including recommended preventative therapies and alternative management options.
Although numerous treatment modalities exist for keloids, no single therapy is entirely effective. Steroid injections up to 14 days before a planned surgery are standard prophylaxis. Affected individuals should collaborate closely with a knowledgeable provider to prevent, treat, and minimize the disability associated with keloids.[4][37]
Pearls and Other Issues
Research into the genetic susceptibility to keloids is ongoing.[38] A study examining the correlation between the skin microbiome and pathological scars found that certain microbes were associated with keloid formation.[39] Further investigations have also focused on distinguishing keloids from other lesions. A study of biomarkers for keloids found that insulin-like growth factor–binding protein-6 and tumor necrosis factor-alpha (TNF-α)–induced protein-6 levels were lower in keloids compared to hypertrophic scars.[40]
A correlation between keloids and skin cancer, potentially due to increased cytokine activity and upregulation of growth factors, highlights the need for enhanced surveillance in this population.[10] Additionally, a novel application of exosomes is being explored to expedite healing and reduce the recurrence of keloids.[41]
Enhancing Healthcare Team Outcomes
Keloid treatment options involve a variety of treatment options, including specialized technologies and equipment. Dermatologists and plastic surgeons are often key members of the interprofessional healthcare team.[31] In cases of diagnostic uncertainty, the expertise of a dermatopathologist may be necessary.[22][42] Wound specialists may be required to manage postsurgical dressings.[21] Radiation therapy treatments involve collaboration with a radiology team, while pharmacists ensure proper dosing for medication-based therapies. Interprofessional collaboration among healthcare providers is crucial for optimizing keloid management and improving patient outcomes.[43]
Media
(Click Image to Enlarge)
References
Berman B, Maderal A, Raphael B. Keloids and Hypertrophic Scars: Pathophysiology, Classification, and Treatment. Dermatologic surgery : official publication for American Society for Dermatologic Surgery [et al.]. 2017 Jan:43 Suppl 1():S3-S18. doi: 10.1097/DSS.0000000000000819. Epub [PubMed PMID: 27347634]
Trace AP, Enos CW, Mantel A, Harvey VM. Keloids and Hypertrophic Scars: A Spectrum of Clinical Challenges. American journal of clinical dermatology. 2016 Jun:17(3):201-23. doi: 10.1007/s40257-016-0175-7. Epub [PubMed PMID: 26894654]
Almadani YH, Vorstenbosch J, Davison PG, Murphy AM. Wound Healing: A Comprehensive Review. Seminars in plastic surgery. 2021 Aug:35(3):141-144. doi: 10.1055/s-0041-1731791. Epub 2021 Jul 15 [PubMed PMID: 34526860]
Bailey J, Schwehr M, Beattie A. Management of Keloids and Hypertrophic Scars. American family physician. 2024 Dec:110(6):605-611 [PubMed PMID: 39700364]
Wang ZC, Zhao WY, Cao Y, Liu YQ, Sun Q, Shi P, Cai JQ, Shen XZ, Tan WQ. The Roles of Inflammation in Keloid and Hypertrophic Scars. Frontiers in immunology. 2020:11():603187. doi: 10.3389/fimmu.2020.603187. Epub 2020 Dec 4 [PubMed PMID: 33343575]
Ibrahim NE, Shaharan S, Dheansa B. Adverse Effects of Pregnancy on Keloids and Hypertrophic Scars. Cureus. 2020 Dec 18:12(12):e12154. doi: 10.7759/cureus.12154. Epub 2020 Dec 18 [PubMed PMID: 33489566]
Ogawa R, Okai K, Tokumura F, Mori K, Ohmori Y, Huang C, Hyakusoku H, Akaishi S. The relationship between skin stretching/contraction and pathologic scarring: the important role of mechanical forces in keloid generation. Wound repair and regeneration : official publication of the Wound Healing Society [and] the European Tissue Repair Society. 2012 Mar-Apr:20(2):149-57. doi: 10.1111/j.1524-475X.2012.00766.x. Epub 2012 Feb 14 [PubMed PMID: 22332721]
Ogawa R. Keloid and Hypertrophic Scars Are the Result of Chronic Inflammation in the Reticular Dermis. International journal of molecular sciences. 2017 Mar 10:18(3):. doi: 10.3390/ijms18030606. Epub 2017 Mar 10 [PubMed PMID: 28287424]
Glass DA 2nd. Current Understanding of the Genetic Causes of Keloid Formation. The journal of investigative dermatology. Symposium proceedings. 2017 Oct:18(2):S50-S53. doi: 10.1016/j.jisp.2016.10.024. Epub [PubMed PMID: 28941494]
Level 3 (low-level) evidenceSharma AN, Birda A, Park M, Choe S, Salas J, Kincaid C, Mesinkovska NA. Insights Into Demographics, Comorbidities, and Risk Factors in Keloids and Hypertrophic Scars: A Retrospective Study. Journal of drugs in dermatology : JDD. 2025 Feb 1:24(2):212-215. doi: 10.36849/JDD.8401. Epub [PubMed PMID: 39913219]
Level 2 (mid-level) evidenceChike-Obi CJ, Cole PD, Brissett AE. Keloids: pathogenesis, clinical features, and management. Seminars in plastic surgery. 2009 Aug:23(3):178-84. doi: 10.1055/s-0029-1224797. Epub [PubMed PMID: 20676312]
Butzelaar L, Ulrich MM, Mink van der Molen AB, Niessen FB, Beelen RH. Currently known risk factors for hypertrophic skin scarring: A review. Journal of plastic, reconstructive & aesthetic surgery : JPRAS. 2016 Feb:69(2):163-9. doi: 10.1016/j.bjps.2015.11.015. Epub 2015 Dec 17 [PubMed PMID: 26776348]
Mahdavian Delavary B, van der Veer WM, Ferreira JA, Niessen FB. Formation of hypertrophic scars: evolution and susceptibility. Journal of plastic surgery and hand surgery. 2012 Apr:46(2):95-101. doi: 10.3109/2000656X.2012.669184. Epub [PubMed PMID: 22471257]
Level 2 (mid-level) evidenceKohlhauser M, Mayrhofer M, Kamolz LP, Smolle C. An Update on Molecular Mechanisms of Scarring-A Narrative Review. International journal of molecular sciences. 2024 Oct 28:25(21):. doi: 10.3390/ijms252111579. Epub 2024 Oct 28 [PubMed PMID: 39519131]
Level 3 (low-level) evidenceZang R, Xu CC, Fan Z, Wang QN, Guo ZJ, Liu L, Cui BN, Huang YY, Yang J. The Role of Fibroblasts in Chronic Inflammatory and Proliferative Skin Diseases. Experimental dermatology. 2025 Feb:34(2):e70066. doi: 10.1111/exd.70066. Epub [PubMed PMID: 39984797]
Wang X, Liu D. Macrophage Polarization: A Novel Target and Strategy for Pathological Scarring. Tissue engineering and regenerative medicine. 2024 Dec:21(8):1109-1124. doi: 10.1007/s13770-024-00669-7. Epub 2024 Oct 1 [PubMed PMID: 39352458]
Jia Y, Qin Y, Yuan FL, Shen JH. Macrophage-to-Myofibroblast Transition Contributes to Cutaneous Scarring Formation Through the TGF-β/Smad3 Signaling Pathways. Cell biology international. 2025 Feb 13:():. doi: 10.1002/cbin.70002. Epub 2025 Feb 13 [PubMed PMID: 39949174]
Level 2 (mid-level) evidenceAubert A, Goeres J, Liu A, Kao M, Richardson KC, Jung K, Hinz B, Crawford RI, Granville DJ. Potential implications of granzyme B in keloids and hypertrophic scars through extracellular matrix remodeling and latent TGF-β activation. Frontiers in immunology. 2024:15():1484462. doi: 10.3389/fimmu.2024.1484462. Epub 2025 Jan 16 [PubMed PMID: 39885984]
Kang M, Ko UH, Oh EJ, Kim HM, Chung HY, Shin JH. Tension-sensitive HOX gene expression in fibroblasts for differential scar formation. Journal of translational medicine. 2025 Feb 10:23(1):168. doi: 10.1186/s12967-025-06191-1. Epub 2025 Feb 10 [PubMed PMID: 39930512]
Jumper N, Paus R, Bayat A. Functional histopathology of keloid disease. Histology and histopathology. 2015 Sep:30(9):1033-57. doi: 10.14670/HH-11-624. Epub 2015 Apr 22 [PubMed PMID: 25900252]
Arno AI, Gauglitz GG, Barret JP, Jeschke MG. Up-to-date approach to manage keloids and hypertrophic scars: a useful guide. Burns : journal of the International Society for Burn Injuries. 2014 Nov:40(7):1255-66. doi: 10.1016/j.burns.2014.02.011. Epub 2014 Apr 24 [PubMed PMID: 24767715]
Liu Y, Xiong X, Cao N, Zhao Y. Diagnosis and Treatment of Keloid: Method Summary and Effect Evaluation. Clinical, cosmetic and investigational dermatology. 2023:16():3775-3783. doi: 10.2147/CCID.S446018. Epub 2023 Dec 29 [PubMed PMID: 38170138]
Quong WL, Dohi T, Miyama A, Ozeki S, Matsumura H, Bulstrode NW, Fish JS, Ogawa R. The efficacy of steroid tape for hypertrophic and keloid scars assessed using the SCAR-Q patient-reported outcome measure. Journal of plastic, reconstructive & aesthetic surgery : JPRAS. 2025 Jan 30:103():39-47. doi: 10.1016/j.bjps.2025.01.038. Epub 2025 Jan 30 [PubMed PMID: 39965439]
Cheruvu VPR, Khan MM, Devalla AV. Development of the AIIMS Bhopal Pre-sternal Keloid Scale (ABPSKS): A Tool for Assessing Severity and Comparing Treatment Outcomes. Cureus. 2024 Dec:16(12):e76255. doi: 10.7759/cureus.76255. Epub 2024 Dec 23 [PubMed PMID: 39845217]
Harris IM, Lee KC, Deeks JJ, Moore DJ, Moiemen NS, Dretzke J. Pressure-garment therapy for preventing hypertrophic scarring after burn injury. The Cochrane database of systematic reviews. 2024 Jan 8:1(1):CD013530. doi: 10.1002/14651858.CD013530.pub2. Epub 2024 Jan 8 [PubMed PMID: 38189494]
Level 1 (high-level) evidenceWang Q, Ren Z, Jin W, Jin Z. Real-world effectiveness and safety of bleomycin in patients with keloids and hypertrophic scars: a systematic review and meta-analysis. Archives of dermatological research. 2025 Jan 6:317(1):170. doi: 10.1007/s00403-024-03687-6. Epub 2025 Jan 6 [PubMed PMID: 39760851]
Level 1 (high-level) evidenceWalsh LA, Wu E, Pontes D, Kwan KR, Poondru S, Miller CH, Kundu RV. Keloid treatments: an evidence-based systematic review of recent advances. Systematic reviews. 2023 Mar 14:12(1):42. doi: 10.1186/s13643-023-02192-7. Epub 2023 Mar 14 [PubMed PMID: 36918908]
Level 1 (high-level) evidenceVettorato E, Volonté P, Musazzi UM, Cilurzo F, Casiraghi A. Skin microincision technique to enhance drug penetration for the treatment of keloid and hypertrophic scars. International journal of pharmaceutics. 2025 Feb 25:671():125259. doi: 10.1016/j.ijpharm.2025.125259. Epub 2025 Jan 30 [PubMed PMID: 39892674]
Mishra T, Wairkar S. Pathogenesis, attenuation, and treatment strategies for keloid management. Tissue & cell. 2025 Jun:94():102800. doi: 10.1016/j.tice.2025.102800. Epub 2025 Feb 17 [PubMed PMID: 39999656]
Atefi N, Jafari MA, Roohaninasab M, Dehghani A, Jafarzadeh A, Peighambari SB, Peighambari A, Goodarzi A. Evaluating the effectiveness and safety of pulsed dye laser alone, the combination of pulsed dye laser and botulinum toxin type A, and the combination of pulsed dye laser and triamcinolone injection in the treatment of hypertrophic and keloid scars: a three-arm randomized controlled clinical trial. Lasers in medical science. 2025 Feb 15:40(1):92. doi: 10.1007/s10103-025-04338-6. Epub 2025 Feb 15 [PubMed PMID: 39953344]
Level 1 (high-level) evidenceOgawa R, Akaishi S, Hyakusoku H. Differential and exclusive diagnosis of diseases that resemble keloids and hypertrophic scars. Annals of plastic surgery. 2009 Jun:62(6):660-4. doi: 10.1097/SAP.0b013e31817e9d67. Epub [PubMed PMID: 19461281]
Nguyen CM, Burch JM, Fitzpatrick JE, Peterson SL, Weston WL. Giant cell fibroblastoma in a child misdiagnosed as a dermatofibroma. Pediatric dermatology. 2002 Jan-Feb:19(1):28-32 [PubMed PMID: 11860566]
Level 3 (low-level) evidenceRutherford A, Glass DA 2nd. A case-control study analyzing the association of keloids with hypertension and obesity. International journal of dermatology. 2017 Sep:56(9):e187-e189. doi: 10.1111/ijd.13618. Epub 2017 May 11 [PubMed PMID: 28497468]
Level 2 (mid-level) evidenceDu D, Li J, Jiang X. Evidence of a causal relationship between blood pressure and pathological scars: a bidirectional Mendelian randomization study. Frontiers in medicine. 2024:11():1405079. doi: 10.3389/fmed.2024.1405079. Epub 2024 Jul 24 [PubMed PMID: 39114830]
Li X, Jiang B, Yao C, Li S, Zuo Y, Yan H. Association between pathological scar and hypertension: A two-sample Mendelian randomization study. Medicine. 2024 Dec 27:103(52):e40977. doi: 10.1097/MD.0000000000040977. Epub [PubMed PMID: 39969358]
Chen F, Kuo YR, Huang CJ, Tang JY, Chiang CH, Huang MY. Lesion Site Is the Key Prognostic Factor for Keloid Patients Receiving Surgery With Adjuvant Radiotherapy. Annals of plastic surgery. 2022 Dec 1:89(6):626-630. doi: 10.1097/SAP.0000000000003315. Epub 2022 Oct 28 [PubMed PMID: 36416688]
Ud-Din S, Bayat A. New insights on keloids, hypertrophic scars, and striae. Dermatologic clinics. 2014 Apr:32(2):193-209. doi: 10.1016/j.det.2013.11.002. Epub [PubMed PMID: 24680006]
Dand N, Ung CY, Saklatvala JR, Simpson MA, Barker JN, Shaw TJ, McGrath JA, Onoufriadis A. GWAS Meta-Analysis Identifies Susceptibility Loci for Keloids and Hypertrophic Scarring in Europeans. The Journal of investigative dermatology. 2024 Dec 31:():. pii: S0022-202X(24)03049-5. doi: 10.1016/j.jid.2024.12.011. Epub 2024 Dec 31 [PubMed PMID: 39746571]
Level 1 (high-level) evidenceZhu W, Wang X, He D, Chen B. Causal relationships between skin microbiome and pathological scars: a bidirectional mendelian randomization study. Archives of dermatological research. 2024 Dec 20:317(1):139. doi: 10.1007/s00403-024-03576-y. Epub 2024 Dec 20 [PubMed PMID: 39704837]
Zhong C, Shi K, Li P, Qiu X, Wu X, Chen S, Liu Y, Li F, Zhao Z, Zhou J, Liang G, Xu D. Single-cell sequencing analysis and bulk-seq identify IGFBP6 and TNFAIP6 as novel differential diagnosis markers for postburn pathological scarring. Burns : journal of the International Society for Burn Injuries. 2024 Dec:50(9):107255. doi: 10.1016/j.burns.2024.08.021. Epub 2024 Sep 3 [PubMed PMID: 39317554]
Level 2 (mid-level) evidencePark SY, Yi KH. Exosome-mediated Advancements in Plastic Surgery: Navigating Therapeutic Potential in Skin Rejuvenation and Wound Healing. Plastic and reconstructive surgery. Global open. 2024 Aug:12(8):e6021. doi: 10.1097/GOX.0000000000006021. Epub 2024 Aug 1 [PubMed PMID: 39534068]
Tan S, Khumalo N, Bayat A. Understanding Keloid Pathobiology From a Quasi-Neoplastic Perspective: Less of a Scar and More of a Chronic Inflammatory Disease With Cancer-Like Tendencies. Frontiers in immunology. 2019:10():1810. doi: 10.3389/fimmu.2019.01810. Epub 2019 Aug 7 [PubMed PMID: 31440236]
Level 3 (low-level) evidenceKim SW. Management of keloid scars: noninvasive and invasive treatments. Archives of plastic surgery. 2021 Mar:48(2):149-157. doi: 10.5999/aps.2020.01914. Epub 2021 Mar 15 [PubMed PMID: 33765731]